practice-problems-2013-11-17

Energy Processing
Metabolism and ATP
Classwork
1. What is metabolism?
2. What role do enzymes play in metabolic pathways?
3. How are catabolic pathways related to energy production?
4.
‘Anabolic’ steroids were/are occasionally utilized by bodybuilders and athletes to enhance performance. Relate what you know about anabolic pathways to explain why these specific groups of people used these drugs.
5. Are catabolic pathways endergonic or exergonic reactions? Compare the amount of free energy in the reactants to the amount of free energy in the products of a catabolic pathway.
6. Explain the relationship between catabolic and anabolic pathways.
7. Which type of pathway may occur spontaneously? Explain this in terms of free energy availability.
8. What energy-storing molecule, created through catabolic pathways, can be utilized later as a source of energy currency?
9. Which bond, in an ATP molecule, contains the most energy? Why is this true?
10. What has happened to a molecule that has been ‘phosphorylated?’ How do you know energy has been released in this process?
11. Why is ATP considered a recyclable molecule?
12. What type of cellular process may be powered by the input of ATP molecules?
Homework
13. Suppose you were able to remove enzymes from their involvement in metabolic pathways. What changes, if any, would you be able to observe in the metabolic processes?
14. What are the differences between catabolic and anabolic pathways?
15. Would a bodybuilder elect to use a catabolic steroid or an anabolic steroid?
Explain your answer.
16. Explain why it makes sense, in terms of energy content, that a catabolic pathway is utilized in obtaining energy from food.
17. Which type of a pathway (catabolic or anabolic) needs to occur first, in order to supply energy for the other one?
18. When coupled, do metabolic pathways result in a net endergonic or exergonic reaction? Explain your answer.
19. Which molecule contains more energy, adenosine diphosphate or adenosine triphosphate? Explain your answer.
20. Which bond, in a molecule of ATP, is broken to obtain energy from the molecule? Why is this the case?
21. Why does adding phosphate groups to the tail of adenosine monophosphate store energy within the molecule? www.njctl.org PSI Biology Energy Processing
22. After providing a reaction with energy through phosphorylation, why does an
ADP molecule get recycled back to ATP, instead of supporting an additional reaction?
Cellular Respiration
Classwork
23. If cellular respiration is a process that yields ATP molecules, do you think this is a catabolic or anabolic pathway? Support your answer.
24. What role do the molecules NAD + and FAD play in the process of cellular respiration?
25. Why are electron acceptors necessary in order for NADH+H + and FADH 2 to release stored energy?
26. Is oxygen a good agent for reduction or oxidation processes? Why?
27. Suppose you are a scientist studying a newly discovered organism. You are asked to determine the type of cellular respiration utilized by this organism and determine whether it is an a)obligate anaerobe, b) obligate aerobe, or c) facultative anaerobe. Design an experiment to test this, and explain what results you would expect for each type of organism.
28. What is the net yield of ATP through glycolysis?
29. Why does the citric acid cycle need to convert pyruvate to a different carbon compound in order complete the cycle? What is the scientific name for this process?
30. What relationship exists between the CO
2
molecules produced through the citric acid cycle and the pyruvates that begin the cycle?
31. Why does the citric acid cycle need to complete two rounds in order to completely process a molecule of glucose?
32. Why is the reduction of FAD to FADH
2
a significant result of the citric acid cycle?
33. When analyzing the process of cellular respiration, it may help to compare the process to volleyball, with the first three stages being the ‘set,’ and the final stage the ‘spike.’ Explain why this analogy makes sense.
34. Considering anaerobic respiration, explain why primordial Earth had a higher concentration of CH
4
, H
2
, and N
2
S than our current atmosphere.
35. Why is the creation of a proton gradient, through the electron transport chain, essential for the ultimate goal of energy production through oxidative phosphorylation?
36. In what way is ATP synthase like a turnstile? What object would represent people in this analogy?
37. How are electron carriers NADH and FADH 2 utilized in the electron transport chain process?
38. How do you think the oxidative phosphorylation process would be affected by the lack of an electron acceptor at the end of the ETC? For example, what would happen if all of the oxygen were consumed during the process of phosphorylation, but reduced electron carriers were still available?
39. Explain why the versatility of catabolism through cellular respiration allows organisms to survive in many different conditions. www.njctl.org PSI Biology Energy Processing
40. Summarize the process of cellular respiration to describe how organisms produce ATP through catabolic pathways.
Homework
41. Life on Earth is sometimes described as a cycle by biologists. Explain why this ‘cycle’ can never reach a point of equilibrium.
42. Which molecule contains more energy: FAD or FADH
2
? Explain your response.
43. Is the conversion of FADH
2
to FAD an example of oxidation or reduction?
Why?
44. Suppose you are experimenting in a lab with an organism that dies in the presence of oxygen. Considering metabolic pathways, how would you categorize this organism?
45. Glycolysis is considered an anaerobic process. What information can you determine from this description?
46. What carbon compound is created as a result of glycolysis?
47. What three energy storing compounds are created through the citric acid cycle?
48. What is the net gain of ATPs from the breakdown of one glucose molecule at the end of the citric acid cycle?
49. When oxygen is present, oxidative phosphorylation can yield 3 ATPs per
NADH and 2 ATPs per FADH
2
. After the first three steps of cellular respiration, we have 10 NADH molecules and 2 FADH
2
molecules. How many ATP molecules can these electron carriers yield?
50. Why does the valence number of an atom of oxygen allow it to be a strong electron acceptor, enabling more copious energy production?
51. Why is the creation of a concentration gradient essential for the process of oxidative phosphorylation?
52. How would a condition that caused the inability of the previous steps of cellular respiration to create NADH and FADH
2
affect production of ATP through oxidative phosphorylation?
53. Compare the process of oxidative phosphorylation to the synthesis or hydroelectric power through using a dam.
54. Which carbon compound yields more energy through respiration, fats or carbohydrates?
Fermentation
Classwork
55. Why must organisms resort to the process of fermentation when electron acceptors are unavailable?
56. Do obligate aerobes engage in the process of fermentation? Why or why not?
57. Would fermentation occur in an organism that had an ample supply of electron acceptors? Why or why not?
58. It may be said that in an absence of electron acceptors, fermentation allows glycolysis to occur. Why is this true? www.njctl.org PSI Biology Energy Processing
59. Swiss cheese is produced partially by the process of fermentation.
Hypothesize why Swiss cheese has holes in it when completed.
60. Imagine you are working as a sports medicine physician. You are asked to devise a way that athletes could perform strenuous activity without the associated muscle burn. Provide a possible solution for this problem.
61. Does the process of fermentation itself produce any ATP? Explain.
Homework
62. In what way is glycolysis related to fermentation?
63. What is the difference between lactic acid fermentation and ethanol fermentation?
64. What role does anaerobic respiration play in the creation of alcoholic beverages, such as beer or wine?
65. Why do your muscles burn during a strenuous physical activity?
66. Suppose you are sitting in the lunch room when a neighboring student makes the claim that ‘eating bacteria would be so gross.’ You notice the student has just eaten a yogurt. How do you reply to this statement?
Photosynthesis
Classwork
67. Based on the chemical equation, what relationship exists between photosynthesis and cellular respiration?
68. Even though humans do not conduct photosynthesis, our existence relies in this process in two ways. Identify these two ways.
69. In what way might photosynthesis and cellular respiration be described as a cycle?
70. Why might the addition of oxygen to Earth’s atmosphere 2.5 BYA have been problematic for the obligate anaerobes in existence at that time?
71. Identify the primary electron carrier utilized in the process of photosynthesis.
72. Suppose you placed a plant in a container that allowed only green, blue and indigo wavelengths of light through. Would this plant be able to conduct photosynthesis at a normal rate? Why or why not?
73. What role does chlorophyll play in the initiation of the photosynthesis process?
74. In what way does a concentration gradient play a role in the light reaction process?
75. What happens to an electron that has been through photosystems II and I?
76. What is the source of the oxygen produced in non-cyclic energy transport?
77. At what point in non-cyclic energy transport is oxygen produced?
78. In what way are the light-independent reactions reliant upon the lightdependent reactions?
79. What role did the Calvin Cycle play in the origination of oxygen in our
Earth’s atmosphere?
80. When looking at a tree, you might realize the solid form of the bark, tissue, roots and branches were once another form of carbon. Explain this thought process. www.njctl.org PSI Biology Energy Processing
81. Can the Calvin Cycle occur at nighttime? Explain your response.
82. How does burning fossil fuels add more carbon to the carbon cycle?
83. How is the carbon cycle related to the greenhouse effect?
84. Why is it important to consider long term, holistic studies when analyzing climate change, as opposed to isolated years and events?
85. In what way does the existence of large, forested areas such as the
Amazon rainforest, help to moderate Earth’s climate?
86. Explain the relationship between the burning of fossil fuels and climate change.
Homework
87. Write the balanced chemical equations for photosynthesis and cellular respiration.
88. How are the products of cellular respiration related to the reactants of photosynthesis?
89. In photosynthesis, the reduction of NADP+ yields what molecule?
90. What is the name of the pigment that allows plant cells to absorb sunlight and power photosynthesis?
91. Explain the ‘cyclic’ nature of cyclic energy transport in photosynthesis?
92. Sketch a thylakoid and label the following: stroma, membrane, lumen.
93. How is the production of ATP in the process of cellular respiration similar to the production of ATP in photosystems II and I?
94. Would it be true to say that oxygen is a byproduct of photosynthesis? Why or why not?
95. What is the role of NADP+ in photosystem I?
96. What role does CO
2
play in the light-independent reactions?
97. Explain the term ‘carbon fixation.’
98. Explain how the light-independent reactions are responsible for the existence of carbohydrates on Earth.
99. Why are greenhouse gases necessary for the maintenance of temperatures adequate to support life on Earth?
100. How did the death and preservation of prehistoric plants and animals affect global temperature trends?
101. Explain the concept of ‘anthropogenic climate change.’ www.njctl.org PSI Biology Energy Processing
Free Response
1. Anabolic pathways of metabolism and catabolic pathways of metabolism are often linked. a. Describe each type of pathway. b. Identify each pathway as either endergonic or exergonic. c. Explain the characteristics of an endergonic reaction. d. Explain the characteristics of an exergonic reaction.
2. The table of data below shows the net molecules produced as the result of glycolysis, pyruvate decarboxylation and the citric acid (Krebs) cycle. Use this table of data to respond to the prompts below :
ATP Stage of cell respiration
Glycolysis
Pyruvate decarboxylation
Citric acid (Krebs) cycle
NADH produced
2
2
6
FADH
2 produced
0
0
2
2
0
2 a. What is the net amount of each molecule, NADH, FADH and ATP, produced by the end of the Citric acid (Krebs) cycle? b. Oxidative phosphorylation, the next step in cellular respiration, will convert all of the NADH and FADH
2
into ATP. How many total ATP molecules will have been produced once oxidative phosphorylation has occurred? (show your work)
3. Compare and contrast the equations, processes, reactants and products of aerobic respiration and photosynthesis by responding to the prompts and questions below: a. Write the balanced equation for aerobic respiration. b. Write the balanced equation for photosynthesis. c. Compare the role of glucose, oxygen, carbon dioxide and water in the process of aerobic respiration with the role of the same molecule in the process of photosynthesis.
4. For photosystems II to occur, there must be two distinct environments. One environment is “inside”, in the lumen, of the thylakoid and one environment is
“outside” the thylakoid, in the stroma of the chloroplast. a.
Explain why this “inside” and “outside” condition must exist in order for these photosystems to function. www.njctl.org PSI Biology Energy Processing
5. The following image of a tree illustrates two processes; one requiring carbon dioxide and one requiring oxygen. Respond to the prompts below based upon this information. http://www.bbc.co.uk/schools/gcsebitesize/science/add_ocr_gateway/green_worl d/photosynthesisrev1.shtml a. Identify the process requiring carbon dioxide. b. Identify the process requiring oxygen. c. Explain why both processes are shown as occurring within one organism, the tree.
Energy Processing
Answer Key
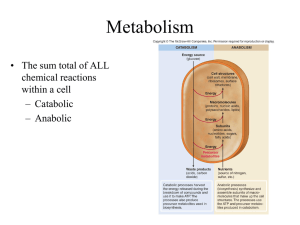
1. Metabolism is the sum of all chemical reactions that occur within an organism.
2. Enzymes are catalysts that support the reactions that occur in metabolic pathways. They allow the reactions to occur at a rate that can support life.
3. Catabolic pathways involve the breakdown of organic molecules into usable energy forms that drive life functions.
4. Anabolic pathways use energy to synthesize complex organic molecules, such as building bone or building muscle. The goal of using anabolic steroids is to synthesize larger muscles at a faster than normal rate.
5. Catabolic pathways are exergonic. There is more free energy in the reactant than the products involved in a catabolic pathway.
6. Catabolic and anabolic pathways are coupled. Anabolic pathways utilize the energy released by catabolic pathways to accomplish a task or synthesize a molecule.
7. Catabolic pathways may occur spontaneously. Since the change in free energy over the course of the reaction is negative, the reaction can occur spontaneously.
8. ATP
9. The bond between the second and third phosphate groups. Each phosphate group has a negative charge. In order to add a new phosphate, the negative charge must be overcome, like compressing a www.njctl.org PSI Biology Energy Processing
spring. Because of this, adding each phosphate requires work, and adding the third phosphate requires the most work, because you must overcome the added negative charges of the first two phosphates.
10. A phosphate group is added to a molecule in the process of phosphorylation. This process allows mechanical work, chemical work and transport work to be accomplished.
11. ATP is considered recyclable because once it is dephosphorylated to
ADP, it can be rephosphorylated to ATP and used again.
12. Mechanical, chemical and transport work are all made possible by ATP.
13. The metabolic processes would occur at a much slower rate than in the presence of enzymes. Some may not occur at all.
14. Catabolic pathways are exergonic, may occur spontaneously, and involve breaking down molecules to release energy. Anabolic pathways are endergonic, are not spontaneous, and involve synthesizing more complex organic molecules.
15. An anabolic steroid. Anabolic pathways synthesize more complex organic molecules, and are involved in creating body tissues such as muscle and bone. Catabolic pathways release energy to be used in anabolic pathways.
16. Catabolic pathways break down molecules, releasing energy in the process.
17. A catabolic pathway releases energy that is used to initiate and perpetuate an anabolic pathway.
18. Exergonic. When coupled, more energy is released in an exergonic reaction than is stored in an endergonic reaction, resulting in a net exergonic system.
19. Adenosine triphosphate. Adding the third phosphate group stores more energy in ATP than is available in ADP.
20. The bond between the second and third phosphate groups is broken to obtain energy. There is more energy stored in this bond than in the other phosphate bonds in the molecule. Breaking this bond also allows for the rephosphorylation of ADP in the future.
21. The phosphate groups carry a negative charge, so adding more phosphates stores more energy in the bonds, similar to compressing a spring.
22. There is more energy stored in the last phosphate bond than in the other two, so it makes more sense to regenerate ATP and obtain a higher energy yield.
23. Cellular respiration is catabolic. The process breaks down organic molecules to yield energy in the form of ATP.
24. NAD + and FAD are electron carrier molecules that can transport high energy electrons from one area to another and release them in order to harness their energy.
25. Electron acceptors are necessary to gather the electrons at the end of the energy harnessing process. www.njctl.org PSI Biology Energy Processing
26. Because oxygen has a high affinity for electrons, it is a good oxidizing agent. This is related to the valence level of oxygen atoms.
27. You would use the presence of oxygen as your independent variable, as the three different categories are related to oxygen tolerance. Your potential solutions would be: a. organism dies in presence of oxygen: obligate anaerobe b. organism dies in absence of oxygen: obligate aerobe c. organism survives whether oxygen is present or absent: facultative anaerobe
28. 2 ATP
29. The citric acid cycle can only process 2-carbon molecules, pyruvate is a 3carbon molecule. The process is called pyruvate decarboxylation.
30. The excess carbon atom from the pyruvate that is removed during pyruvate decarboxylation is released as CO
2
.
31. A molecule of glucose yields two molecules of pyruvate. The citric acid cycle processes one pyruvate molecule per round.
32. The FADH
2 carries energy that will be released and converted to ATP in the final stages of cellular respiration.
33. Just as the ‘set’ doesn’t yield any points in volleyball, the first three stages of cellular respiration does not yield a lot of ATP. However, the set allows the spike to occur, which does yield points. Likewise, the first three stages of cellular respiration allow the electron transport chain and oxidative phosphorylation to occur, which yields a large amount of ATP.
34. These three gases were created as byproducts of anaerobic respiration, as a result of combining with the electron acceptors that were available in the atmosphere at that time.
35. The proton gradient causes the protons to flow across the membrane due to the nature of diffusion, and in doing so, the energy is harnessed through
ATP synthase. Without the gradient, the protons would not flow in a particular direction.
36. Just as people walk through a turnstile to enter a building, protons enter
ATP synthase to cross the membrane. As ATP synthase turns, it phosphorylates a molecule of ATP from the energy released by the proton. Likewise, a turnstile uses the movement of a person to change the number on the meter as the person enters.
37. The electron carriers release their electrons into the electron transport chain, which harnesses the energy and uses it to pump protons across the membrane and create a gradient.
38. The lack of an electron acceptor would halt the phosphorylation process.
If the spent electrons were not removed from the conclusion of the chain, they would essentially create a ‘traffic jam,’ and the process would cease until the electrons were removed. Oxygen readily accepts the electrons, creating H
2
O and allowing the process to continue.
39. Glycolysis, the beginning of the cellular respiration process, can use a variety of different organic molecules as a source of carbon to initiate the process. www.njctl.org PSI Biology Energy Processing
40. The energy stored in organic molecules is systematically broken down through glycolysis and the Krebs cycle until it is used to synthesize a usable form of energy currency, ATP through the electron transport chain and oxidative phosphorylation.
41. Life on Earth is an open system, and thus cannot reach equilibrium.
Energy is constantly being exchanged between organisms and the environment.
42. FADH
2
. The creation of FADH
2
from FAD stores electrons in the molecule, which can be released later, yielding energy.
43. Oxidation. Reduction adds electrons, oxidation removes them.
44. This organism is likely an obligate anaerobe.
45. Glycolysis may occur in the absence of oxygen.
46. Pyruvate.
47. NADH, FADH
2
, ATP
48. 2 ATP
49. 34 ATP
50. Oxygen has a valence number of 6, so it has a high affinity for two additional electrons in order to fill its outer shell and become more stable.
51. The concentration gradient will cause the protons to flow back into the cell, through the ATP synthase, which will use their movement to generate energy and phosphorylate ATP molecules.
52. Oxidative phosphorylation would likely not occur. The electrons donated by NADH and FADH
2
are necessary to pump the protons outside of the cell and create the concentration gradient.
53. Creating a dam allows us to harness the power of water as it moves from an area of artificially created high concentration to an area of low concentration. In a similar way, oxidative phosphorylation harnesses energy through the movement of protons from an area of high concentration to an area of low concentration.
54. Fats yield double the energy of carbohydrates.
55. If electron acceptors are unavailable, oxidative phosphorylation and the electron transport chain cannot proceed. Fermentation recycles NADH and allows glycolysis to continue.
56. No. Obligate aerobes cannot survive in the absence of oxygen.
57. No. If electron acceptors were readily available, the citric acid cycle and electron transport chain would proceed as normal.
58. Fermentation breaks down the pyruvates produced through glycolysis and recycles the NADH back to NAD + . Without this process, the pyruvate would accumulate and there would not be enough NAD+ molecules available for glycolysis to continue.
59. One of the fermentation pathways produces CO
2
as a byproduct. This
CO
2
creates the bubbles in Swiss cheese that we see as holes when the cheese is cut.
60. There are multiple answers to this question. You could find a way to deliver more oxygen to the body, which would prevent the body from entering fermentation. Or, you could find a way to break down the lactic www.njctl.org PSI Biology Energy Processing
acid produced through fermentation, which produces the associated muscle burn of strenuous exercise.
61. No. Fermentation allows for the continuation of glycolysis, which nets 2
ATP, but fermentation itself does not produce ATP.
62. Fermentation uses the products of glycolysis in the absence of oxygen during cellular respiration.
63. Lactic acid fermentation produces lactic acid, ethanol fermentation produces ethanol and CO 2 .
64. Yeast is used in the brewing or fermenting process, which uses a carbohydrate source (grain or grapes) to conduct ethanol fermentation, producing alcohol and CO 2 in the process.
65. During strenuous activity you use up the oxygen available in your body, and some of your cells begin to enter anaerobic respiration to produce
ATP through glycolysis. Since electron acceptors are not available, lactic acid fermentation occurs, and the burn in your muscles is a result of the accumulation of lactic acid.
66. The yogurt consumed is produced through fermentation, which is conducted by bacteria strains, such as Lactobacillus. Most yogurt containers will say this right on the label.
67. The reactants of photosynthesis are the products of respiration, the reactants of respiration are the products of photosynthesis.
68. We use the sugars produced in photosynthesis as our primary energy source, we require the oxygen produced in photosynthesis to breathe.
69. Since the products of one are the reactants of the other, in some ways they allow each other to keep going.
70. Obligate anaerobes cannot survive in the presence of oxygen, so the addition of oxygen to the atmosphere would have been detrimental for their existence. This is where the term ‘oxygen catastrophe’ originates.
71. NADP+
72. Probably not. Most plants use chlorophyll as their primary photosynthetic pigment. Chlorophyll absorbs red and violet-blue light. While some plants contain other pigments in addition to chlorophyll, the presence of red light is what allows chlorophyll to absorb light and initiate photosynthesis.
73. Chlorophyll absorbs sunlight, which will then energize electrons to begin the light reactions. Chlorophyll absorbs the light energy which will ultimately be converted to chemical energy in the process.
74. The light reactions use energized electrons to transport protons across the thylakoid membrane, which will then eventually flow back in, down their gradient, through ATP synthase to produce ATP.
75. They are used to store energy by reducing NADP+ to NADPH.
76. The oxygen is a byproduct of splitting H
2
O molecules to obtain electrons.
77. At the beginning of photosystem II.
78. The light-independent reactions use the NADPH and ATP to energize their sugar production.
79. The Calvin Cycle adds oxygen to the atmosphere as it breaks down CO
2 molecules and releases oxygen as a byproduct. www.njctl.org PSI Biology Energy Processing
80. The solid tissue of a tree is created from carbon. This carbon was obtained through carbon fixation, in which plants convert carbon dioxide gas into carbohydrates. The solid carbon that now makes up a tree was at one time an atmospheric gas.
81. Yes, the Calvin Cycle does not directly depend on the presence of light, but instead the products of the light-dependent reactions to occur.
82. Burning fossil fuels releases carbon that had been locked in preserved plants and animals as CO
2
back into the atmosphere.
83. As carbon compounds are burned, they release their carbon as carbon dioxide, which returns to the atmosphere and contributes the greenhouse effect.
84. Climate change needs to be analyzed in long-term trends, not isolated events. Monthly and yearly swings in temperature do not reflect planetary temperature trends and are not a good reflection of the effects of climate change.
85. Large, forested areas are referred to as a ‘carbon sink,’ because the large amount of carbon locked in the trees is not available to contribute to the greenhouse effect in the atmosphere. These trees can also remove massive amounts of carbon dioxide from the air in the process of photosynthesis.
86. Burning fossil fuels increases the amount of greenhouse gases in the atmosphere, such as carbon dioxide and methane. Increased greenhouse gases are responsible for global warming.
87. Photosynthesis: 6CO
2
+ 6H
2
O + Light 6O
2
+ C
6
H
12
O
6
Respiration: 6O
2
+ C
6
H
12
O
6
6CO
2
+ 6H
2
O + ATP
88. With the exception of ATP and Light, the products of one process are the reactants of the other.
89. NADPH
90. Chlorophyll
91. Cyclic energy transport recycles the electrons used back to the chlorophyll to restart the process.
92.
93. Both processes utilize an electron transport chain to harness the energy of an electron to pump protons against their gradient and allow them to flow through ATP synthase to phosphorylate ADP to ATP. www.njctl.org PSI Biology Energy Processing
94. Yes, oxygen is produced at two different points in photosynthesis, and is not utilized during the process.
95. NADP+ is utilized as an electron carrier.
96. CO
2
is the source of carbon to synthesize organic molecules.
97. Carbon fixation is the process of converting atmospheric CO
2
into an organic molecule such as carbohydrates.
98. The light-independent reactions utilize energy harnessed in the lightdependent reactions to convert atmospheric CO
2
into carbohydrates. This process is responsible for the origination and perpetuation of carbohydrates on Earth.
99. The greenhouse effect warms the Earth to temperatures that support complex life as we know it.
100. The death and preservation of prehistoric plants and animals essentially removed carbon from the carbon cycle, reducing the amount of
CO
2
in the atmosphere and decreasing the greenhouse effect, lowering
Earth’s temperature to more moderate levels.
101. Anthropogenic climate change is climate changed that is induced by human action. For example, burning fossil fuels releases trapped carbon into the atmosphere as CO
2
, where it accumulates and contributes to the greenhouse effect. The greenhouse effect will occur regardless of our actions, but the degree to which it occurs is linked to our consumption of fossil fuels.
1. Question 1 a. Anabolic pathways synthesize molecules and power cell processes.
Catabolic functions break down molecules. b. Anabolic pathways are endergonic. Catabolic pathways are exergonic, c. Endergonic reactions require an input of energy and are not spontaneous. d. Exergonic reactions release energy and are spontaneous.
2. Question 2 a. 10 NADH, 2 FADH
2
, and 4ATP are produced by the end of the citric acid or Krebs cycle b. 38 ATP
3. Question3 a. C
6
H
12
O
6
+ 6O
2
6CO
2
+ 6 H
2
O + ATP b. 6CO
2
+ 6 H
2
O + ATP C
6
H
12
O
6
+ 6O
2 c. In aerobic respiration, glucose and oxygen are reactants for the reaction while in photosynthesis the glucose and oxygen are products. The carbon dioxide and water are products (considered by products) of aerobic respiration while they act as reactants in photosynthesis.
4. Question 4 www.njctl.org PSI Biology Energy Processing
a. Photosystem II is embedded within a membrane of the thylakoid. A proton gradient, or difference in concentration, is produced between the inside (lumen) and the outside (stroma). The protons must move through ATP synthase. This is the energy that produces ATP
5. Question 5 a. The carbon dioxide is taken in by the tree in order to perform photosynthesis. b. The oxygen is taken in order for the tree to perform aerobic
(cellular) respiration. c. Plants produce glucose and oxygen as the result of photosynthesis.
The tree then utilizes the glucose produced as the reactant to perform its own aerobic respiration. Plants such as trees must perform aerobic respiration in order to grow and to perform cellular functions as an organism. www.njctl.org PSI Biology Energy Processing