DrBs Acids and Bases Lab
advertisement

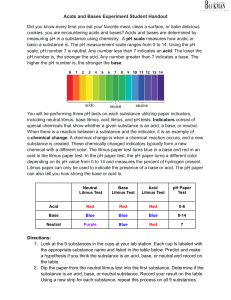
Acids and Bases Lab Today you will testing whether or not certain substances are acids or bases. You will be doing three tests on each substance to determine this: litmus tests and pH paper. 1. You have 18 substances stationed around the room. Each are labeled with the appropriate name and listed in the table below. 2. Predict if the substance is an acid, base, or neutral substance. Record prediction in the correct column. 3. The next test is the litmus test. This will serve as a prediction test. If your prediction indicated the substance was an acid, use the blue litmus paper. It should turn red. If you think the substance was a base, use the red litmus paper, it should turn blue. If you think the substance is neutral, use the both pink & blue paper. Record your result under the litmus column 5. The final test is to determine the number of the substance on the pH scale. Use the pH Hydrion Paper. Dip a strip into each substance. Wait 30 seconds for the paper to change and compare with the scale on the tube. Record the number that the color matches in the correct column on the data table. 6. Discard strips in the receptacle provided. Substance Tap Water Coffee Salt Water Soda Peroxide Lemon Juice Vinegar Hand Soap Water Mouthwash HCl Liquid Plummer Baking Soda in Water Apple Juice Milk Shampoo Ammonia Distilled Water Prediction Litmus Paper pH Paper Acid, Base, or Neutral Watch this video: https://ed.ted.com/lessons/the-strengths-and-weaknesses-of-acids-andbases-george-zaidan-and-charles-morton Add new knowledge from the video to your IN notes. Analysis Questions: 1. Explain why some substances are acids and some substances are bases. 2. What does it mean to say that distilled water is neutral? What pH should it have? 3. Consider the food items tested. What conclusions, if any, can you make concerning their pH? What conclusion can be made about the cleaning products in general? 4. From the data collected, what generalization can you make about the pH of a solution and the effect of a solution on litmus paper? 5. Using the data from this investigation, design a pH scale by listing the name and the pH of the substances tested.