Heat Notes
advertisement

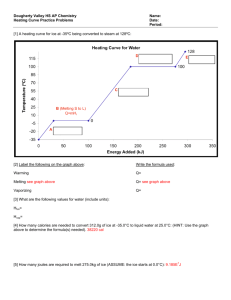
The three laws of thermodynamics - The first law of thermodynamics, also called conservation of energy, states that the total amount of energy in the universe is constant. This means that all of the energy has to end up somewhere, either in the original form or in a different from. We can use this knowledge to determine the amount of energy in a system, the amount lost as waste heat, and the efficiency of the system. -The second law of thermodynamics states that the disorder in the universe always increases. After cleaning your room, it always has a tendency to become messy again. This is a result of the second law. As the disorder in the universe increases, the energy is transformed into less usable forms. Thus, the efficiency of any process will always be less than 100%. - The third law of thermodynamics tells us that all molecular movement stops at a temperature we call absolute zero, or 0 Kelvin (-273oC). Since temperature is a measure of molecular movement, there can be no temperature lower than absolute zero. At this temperature, a perfect crystal has no disorder. When put together, these laws state that a concentrated energy supply must be used to accomplish useful work. Remember: Heat flow from where it’s HOT to where it’s NOT (hot) Heat is measure in calories (Calorie = 1000 calories; a calorie is the amount of heat needed to raise 1 grams of water 1 oC), joules or in the USA B.T.U.’s (British thermal unit; the energy needed to raise one pound of water 1 oF) Temperature is a measure of the average kinetic energy of the material measured in degrees; oF, oC or Kelvin (K) Specific heat is the energy needed to change the temperature of one grams of the specific material 1oC; or the heat Capacity (specifically); we use the symbol capital “C” with a subscript to denote the material Example: CFe is the specific heat of iron, CFe = 0416 j/goC; CPb is the specific heat of lead, CPb = 0.16 j/goC q = mC T Heat equations: ; read as: xx “quantity of heat” equals “mass” times “specific heat” times the “change in temperature” Use your algebra skills to get: Cxx = q/m T or m=q/ Cxx T or T = q/mCxx Remember: Q has units of joules, m has units of grams, DT has units of oC, Cxx has units of j/goC Understanding “The Heating Curve” and phase changes as they relate to thermodynamics The diagram below is a plot of temperature vs. energy input. It represents the heating of what is initially ice at -50oC at a near constant rate of heat transfer. 1) Identify the phase of matter at each section A,B, C, D, & E (write right on the graph) 2) What is happening at the flat spots; energy is going into the material but the temperature is not changing. What’s up with that? _________________________________________________ 3) How much energy was required to heat the ice to its melting point? _______________ 4) How much energy did it take to melt the ice? _______________ 5) How much energy did it require to heat the now liquid water to its boiling point? ____________ 6) How much energy did the 100oC water absorb to vaporize? _________________ 7) Why do you think it takes so much more energy to vaporize water than to melt ice? _________________________________________________________________________ For phase Changes: q = m Hfus or q = mHvap The heat of vaporization for water is 2260 joules/ gram and the heat of fusion of water is 334 j/g Using this information is the graph correct and how much (in grams) water must have been present? For the smarty pants use the heat equation: q=mCxx T to calculate (or confirm) the specific heat of Ice, water and steam; Is the graph drawn correctly to show that the specific heat of water is greatest (about double that of ice? Heat Lecture Notes (revised 2016) (Chapter 12 and 15 of Glencoe Chemistry) (Chapter 13 and 17 of Prentice Hall Chemistry Chapter 9 and 10 of Addison-Wesley Chemistry) What Is Heat? Heat is a form of ENERGY Remember that ENERGY is the ability to cause change or do WORK (we will talk more about work later on in Physics) Temperature is just a measure of the AVERAGE kinetic energy of a sample. (that’s why all liquids evaporate (have a vapor pressure); some molecules will have enough energy to leave the system as a gas) The units of heat are calories or Joules ; or kcal or kJ (we still sometimes use B.T.U.’s here in the USA) One calorie is the energy necessary to change one gram of water one degree Celsius 1 calorie = 4.184 joules Heat capacity is different for different things. Larger things have more heat capacity than smaller pieces of the same stuff. Water is a material that can hold a lot of heat, it has a very large heat capacity. The specific heat of liquid water is 4.18 joules per gram×oC The heat Equation is: Q = m× C×T Where: Q = the amount of heat (usually measured in calories or joules) m = the mass of the material (measured in grams or kilograms) C = the specific heat of the material (weird units: j/goC) T = the change in temperature of the material (usually oC) T= Tfinal − Tinitial So…. The amount of heat is equal to the mass times the specific heat times the change in temperature For a change of phase (state). The heat equation is: Q = m×Hfus Where: or Q = m×Hvap Q = the amount of heat m = the mass of the material Hfus = the heat of fusion of the material Hvap = the heat of vaporization of the material You need to know these constants for water Hfus = the heat of fusion of water is 334 J/g Hvap = the heat of vaporization of water 2260 J/g You will need to know (or look up). (Remember K = oC; you can use them interchangeably) C = the specific heat of the material Cwater = 4.18 J/g× oC Cice = 2.1 J/g× oC Csteam = 1.7 J/g× oC