Fabrication II - Deposition-1
advertisement

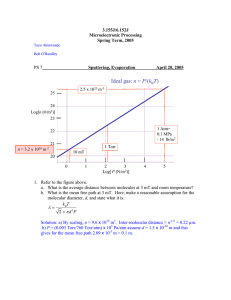
II. Thin Film Deposition Physical Vapor Deposition (PVD) - Film is formed by atoms directly transported from source to the substrate through gas phase • Evaporation • Thermal evaporation « • E-beam evaporation « • Sputtering • DC sputtering « • DC Magnetron sputtering « • RF sputtering « • Reactive PVD Chemical Vapor Deposition (CVD) - Film is formed by chemical reaction on the surface of substrate • Low-Pressure CVD (LPCVD) « • Plasma-Enhanced CVD (PECVD) « • Atmosphere-Pressure CVD (APCVD) • Metal-Organic CVD (MOCVD) Oxidation Spin Coating Platting Applied Physics 298r 1 E. Chen (4-12-2004) General Characteristics of Thin Film Deposition • Deposition Rate • Film Uniformity • Across wafer uniformity • Run-to-run uniformity • Materials that can be deposited • Metal • Dielectric • Polymer • Quality of Film – Physical and Chemical Properties • Stress • Adhesion • Stoichiometry • Film density, pinhole density • Grain size, boundary property, and orientation • Breakdown voltage • Impurity level • Deposition Directionality • Directional: good for lift-off, trench filling • Non-directional: good for step coverage • Cost of ownership and operation Applied Physics 298r 2 E. Chen (4-12-2004) Evaporation ¨ Load the source material-to-bedeposited (evaporant) into the container (crucible) ¨ Heat the source to high temperature ¨ Source material evaporates ¨ Evaporant vapor transports to and Impinges on the surface of the substrate ¨ Evaporant condenses on and is adsorbed by the surface Applied Physics 298r 3 Substrate Film Evaporant Vapor Current Crucible (energy source) E. Chen (4-12-2004) Langmuire-Knudsen Relation Mass Deposition Rate per unit area of source surface: Substrate 1 2 1 M Rm = Cm cos θ cos ϕ 2 (Pe (T ) − P ) r T r θ Cm = 1.85x10-2 r: source-substrate distance (cm) T: source temperature (K) Pe: evaporant vapor pressure (torr), function of T P: chamber pressure (torr) M: evaporant gram-molecular mass (g) ¬ Maximum deposition rate reaches at high chamber vacuum (P ~ 0) Applied Physics 298r 4 ϕ P Pe Source (K-Cell) E. Chen (4-12-2004) Uniform Coating Spherical surface with source on its edge: Spherical Surface r cos θ = cos ϕ = 2r0 ϕ 1 2 M Pe Rm = Cm 2 T 4r0 r0 θ P ¨ Angle Independent – uniform coating! ¬ Used to coat instruments with spherical surfaces Applied Physics 298r r 5 Pe Source (K-Cell) E. Chen (4-12-2004) Uniformity on a Flat Surface Consider the deposition rate difference between wafer center and edge: R1 ∝ W /2 1 2 r1 2 1 r R2 ∝ 2 cos 2 θ = 1 4 r2 r2 ϕ r1 θ r2 Define Uniformity: σ (% ) = P R1 − R2 (% ) R1 Pe −2 2 W 2 W σ = 1 − 1 + ≈ 2 2r1 2r1 Applied Physics 298r or W = 2σ r1 6 Source (K-Cell) E. Chen (4-12-2004) Wafer Uniformity Requirement on a Flat Surface Source-substrate distance requirement: W 2σ In practice, it is typical to double this number to give some process margin: r >W 2 σ Source-Sample Distance (r) r> 160 Larger r Means: ¬ bigger chamber ¬ higher capacity vacuum pump ¬ lower deposition rate ¬ higher evaporant waste Applied Physics 298r 140 1% 2% 120 5% 100 10% 80 60 40 20 0 0 2 4 6 8 Sample Size (W) Another Common Solution: off-axis rotation of the sample 7 E. Chen (4-12-2004) 10 Thickness Deposition Rate vs. Source Vapor Pressure dh Rm = Ae dt ρ Thickness deposition rate Substrate Film dh 1 2 dh Ae 1 M = Cm cos θ cos ϕ 2 Pe (T ) dt ρ r T T: Ae: ρ: θ source temperature (K) source surface area (cm2) evaporant density (g/cm3) Ae Pe is function of source Temperature! (A/s) Applied Physics 298r ¬ ϕ P Pe T Example: Al M ~ 27, ρ ~ 2.7, Ae ~ 10-2 cm2, T ~ 900 K R ~ 50 cm (uniformity requirement) dh = 50 Pe dt r Source (K-Cell) The higher the vapor pressure, the higher the material’s deposition rate 8 E. Chen (4-12-2004) Deposition Rate vs. Source Temperature Typically for different material: dh = (10 ~ 100) Pe (T ) dt • • • • ( A / s) For deposition rate > 1 A/s: Pe > ~ 100 mtorr Pe depends on: 1) materila and 2) temperature Deposition rates are significantly different for different materials Hard to deposit multicomponent (alloy) film without losing stoichiometry Applied Physics 298r Example: for Pe > 100 mtoor T(Al) > 1400K, T(Ta) > 2500K 9 E. Chen (4-12-2004) Heating Method – Thermal (Resist Heater) Source Material Resistive Wire Current Foil Dimple Boat Crucible Alumina Coated Foil Dimple Boat Contamination Problem with Thermal Evaporation Container material also evaporates, which contaminates the deposited film Cr Coated Tungsten Rod Applied Physics 298r 10 E. Chen (4-12-2004) CIMS’ Sharon Thermal Evaporator Applied Physics 298r 11 E. Chen (4-12-2004) Heating Method – e-Beam Heater e- Electron Beam Crucible Magnetic Field Evaporant (beam focusing & positioning) Evaporant Focusing Aperture Cathode Filament Water Cooled Rotary Copper Hearth (Sequential Deposition) Applied Physics 298r 12 Advantage of E-Beam Evaporation: Very low container contamination E. Chen (4-12-2004) CIMS’ Sharon E-Beam Evaporator Applied Physics 298r 13 E. Chen (4-12-2004) Comparison Deposition Thermal E-Beam • • Material Typical Evaporant Impurity Deposition Rate Temperature Range Cost Metal or low melt-point materials Au, Ag, Al, Cr, Sn, Sb, Ge, In, Mg, Ga CdS, PbS, CdSe, NaCl, KCl, AgCl, MgF2, CaF2, PbCl2 High 1 ~ 20 A/s ~ 1800 ºC Low Both metal and dielectrics Everything above, plus: Ni, Pt, Ir, Rh, Ti, V, Zr, W, Ta, Mo Al2O3, SiO, SiO2, SnO2, TiO2, ZrO2 Low 10 ~ 100 A/s ~ 3000 ºC High Stoichiometrical Problem of Evaporation Compound material breaks down at high temperature Each component has different vapor pressure, therefore different deposition rate, resulting in a film with different stoichiometry compared to the source Applied Physics 298r 14 E. Chen (4-12-2004) Typical Boat/Crucible Material Refractory Metals Material Melting Point (ºC) Temperature for 10-mtorr Vapor Pressure (Pe) (ºC) Tungsten (W) 3380 3230 Tantalum (Ta) 3000 3060 Molybdenum (Mo) 2620 2530 Refractory Ceramics Graphitic Carbon (C) 3799 2600 Alumina (Al2O3) 2030 1900 Boron Nitride (BN) 2500 1600 Applied Physics 298r 15 E. Chen (4-12-2004) DC Diode Sputtering Deposition 2 – 5kV • Target (source) and substrate are placed on two parallel electrodes (diode) • They are placed inside a chamber filled with inert gas (Ar) • DC voltage (~ kV) is applied to the diode Target (Cathode) • Free electron in the chamber are e- e- accelerated by the e-field • These energetic free electrons inelastically Ar γ Ar+ e- Ar collide with Ar atoms excitation of Ar ¨ gas glows Substrate (Anode) ionization of Ar ¨ Ar+ + 2nd electron • 2nd electrons repeat above process ¨ “gas breakdown” ¨ discharge glow (plasma) Applied Physics 298r 16 E. Chen (4-12-2004) Self-Sustained Discharge • Near the cathode, electrons move much faster than ions because of smaller mass ¬ positive charge build up near the cathode, raising the potential of plasma ¬ less electrons collide with Ar ¬ few collision with these high energetic electrons results in mostly ionization, rather than excitation ¬ dark zone (Crookes Dark Space) • Discharge causes voltage between the electrodes reduced from ~103 V to ~102V, mainly across the dark space • Electrical field in other area is significantly reduced by screening effect of the position charge in front of cathode • Positive ions entering the dark space are accelerated toward the cathode (target), bombarding (sputtering) the target ¬ atoms locked out from the target transport to the substrate (momentum transfer, not evaporation!) ¬ generate 2nd electrons that sustains the discharge (plasma) Applied Physics 298r 17 2 – 5kV Target (Cathode) + + + + + Ar+ Crookes Dark Space Ar+ t t Substrate (Anode) E. Chen (4-12-2004) Requirement for Self-Sustained Discharge • If the cathode-anode space (L) is less than the dark space length ¬ ionization, few excitation ¬ cannot sustain discharge • On the other hand, if the Ar pressure in the chamber is too low ¬ Large electron mean-free path ¬ 2nd electrons reach anode before colliding with Ar atoms ¬ cannot sustain discharge either L ⋅ P > 0.5 (cm ⋅ torr ) Condition for Sustain Plasma: L: electrode spacing, P: chamber pressure For example: Typical target-substrate spacing: L ~ 10cm ¨ P > 50 mtorr Applied Physics 298r 18 E. Chen (4-12-2004) Deposition Rate vs. Chamber Pressure High chamber pressure results in low deposition rate Mean-free path of an atom in a gas ambient: In fact, sputtering deposition rate R: 5 × 10 −3 λ~ (cm) P (torr ) R∝ Use previous example: L = 10 cm, P = 50 mtorr ¨ λ ~ 0.1 cm ¨ sputtered atoms have to go through hundreds of collisions before reaching the substrate ¨ significantly reduces deposition rate ¨ also causes source to deposit on chamber wall and redeposit back to the target Applied Physics 298r 1 L⋅P Large LP to sustain plasma small LP to maintain good deposition rate and reduce random scattering 19 E. Chen (4-12-2004) ? DC Magnetron Sputtering • Using low chamber pressure to maintain high deposition rate • Using magnetic field to confine electrons near the target to sustain plasma E e- + B + S Cathode (Target) N S Apply magnetic field parallel to the cathode surface Target ¨ electrons will hope (cycloid) near the S surface (trapped) Applied Physics 298r 20 N E. Chen (4-12-2004) S Impact of Magnetic Field on Ions Hoping radius r: r~ 1 B 2m Vd e Ar+ Vd – voltage drop across dark space (~ 100 V) B – Magnetic field (~ 100 G) e- E + B r Cathode (Target) For electron r ~ 0.3 cm For Ar+ ion: r ~ 81 cm Applied Physics 298r 21 E. Chen (4-12-2004) As A Result … ¬ current density (proportional to ionization rate) increases by 100 times ¬ required discharge pressure drops 100 times ¬ deposition rate increases 100 times Deposition Rate (R) Magnetron Non-Magnetron ~ 1mT ~ 100mT Chamber Pressure (P) Applied Physics 298r 22 E. Chen (4-12-2004) RF (Radio Frequency) Sputtering DC sputtering cannot be used for depositing dielectrics because insulating cathode will cause charge build up during Ar+ bombarding 13.56 MHz ~ ¨ reduce the voltage between electrodes ¨ discharge distinguishes Target Solution: use AC power • at low frequency (< 100 KHz), both electrons and ions can follow the switching of the voltage – ¨ DC sputtering • at high frequency (> 1 MHz), heave ions cannot no long follow the switching ¨ ions are accelerated by dark-space (sheath) voltage ¨ electron neutralizes the positive charge buildup on both electrodes • However, there are two dark spaces ¨ sputter both target and substrate at different cycle Applied Physics 298r 23 Target Sheath e- eAr Ar+ t Substrate Sheath Substrate E. Chen (4-12-2004) RF (Radio Frequency) Sputtering 13.56 MHz A VT ∝ S VS AT VT Vs AT As – – – – n ~ (n ~ 2) AT voltage across target sheath voltage across substrate sheath area of target electrode area of substrate electrode Target VT AS VS Substrate Larger dark-space voltage develops at the electrode with smaller area ¨ make target electrode small Applied Physics 298r 24 E. Chen (4-12-2004) Comparison between Evaporation and Sputtering Evaporation Low energy atoms (~ 0.1 eV) Sputtering High energy atoms / ions (1 – 10 eV) • denser film • smaller grain size • better adhesion High Vacuum • directional, good for lift-off • lower impurity Low Vacuum • poor directionality, better step coverage • gas atom implanted in the film Point Source • poor uniformity Parallel Plate Source • better uniformity Component Evaporate at Different Rate • poor stoichiometry All Component Sputtered with Similar Rate • maintain stoichiometry Applied Physics 298r 25 E. Chen (4-12-2004) Chemical Vapor Deposition (CVD) Deposit film through chemical reaction and surface absorption • Introduce reactive gases to the chamber • Activate gases (decomposition) A B ¬ heat A B ¬ plasma • Gas absorption by substrate surface B A • Reaction take place on substrate surface; A W film firmed B W Substrate • Transport of volatile byproducts away form substrate • Exhaust waste Applied Physics 298r 26 E. Chen (4-12-2004) Types of CVD Reactions Pyrolysis (Thermal Decomposition) AB ( gas ) → A ( solid ) + B ( gas ) Example α-Si deposited at 580 - 650 ºC: SiH 4 ( gas ) = Si ( solid ) + 2 H 2 ( gas ) Reduction (lower temperature than Pyrolysis) AB ( gas ) + H 2 ( gas, commonly used ) ↔ A ( solid ) + HB ( gas ) Example W deposited at 300 ºC: WF6 ( gas ) + 3H 2 ( gas ) = W ( solid ) + 6 HF ( gas ) Reversible process, can be used for chamber cleaning Applied Physics 298r 27 E. Chen (4-12-2004) Types of CVD Reactions (Cont.) Oxidation AB ( gas or solid ) + O2 ( gas, commonly used ) ↔ AO ( solid ) + [O ]B ( gas ) Example Low-temperature SiO2 deposited at 450 ºC: SiH 4 ( gas ) + O2 ( gas ) = SiO2 ( solid ) + 2 H 2 ( gas ) Example SiO2 formed through dry oxidation at 900 - 1100 ºC: Si ( Solid ) + O2 ( gas ) = SiO2 ( solid ) Applied Physics 298r 28 E. Chen (4-12-2004) Types of CVD Reactions (Cont.) Compound Formation AB ( gas or solid ) + XY ( gas or solid ) ↔ AX ( solid ) + BY ( gas ) Example SiO2 formed through wet oxidation at 900 - 1100 ºC: Si ( Solid ) + 2 H 2O(vapor ) = SiO2 ( solid ) + 2 H 2 Example SiO2 formed through PECVD at 200 - 400 ºC: Si H 4 ( gas ) + 2 N 2O( gas ) = SiO2 ( solid ) + 2 N 2 + 2 H 2 Example Si3N4 formed through LPCVD at 700 - 800 ºC: 3Si H 2Cl2 ( gas ) + 4 NH 3 ( gas ) = Si3 N 4 ( solid ) + 6 H 2 + 6 HCl Applied Physics 298r 29 E. Chen (4-12-2004) CVD Deposition Condition Mass-Transport Limited Deposition - At high temperature such that the reaction rate - Gas delivering controls film deposition rate - Film growth rate insensitive to temperature - Film uniformity depends on whether reactant can be uniformly delivered across a wafer and wafer-to-wafer Reaction-Rate Limited Deposition Deposition Rate (log) exceeds the gas delivering rate Mass-Transport Limited Regime Reaction-Rate Limited Regime - At low temperature or high vacuum such that 1/T (K) the reaction rate is below gas arriving rate - Temperature controls film deposition rate - Film uniformity depends on temperature uniformity across a wafer and wafer-to-wafer Applied Physics 298r 30 E. Chen (4-12-2004) Low-Pressure CVD (LPCVD) Heater Heater Heater Reactant Gas Horizontal Quartz Tube Exhausted Gas Wafer Z-1 Z-2 Z-3 • Thermal energy for reaction activation • System works at vacuum (~ 0.1 – 1.0 torr), resulting in high diffusivity of reactants ¨ reaction-rate limited • Wafer can stacked closely without lose uniformity as long as they have the same temperature • Temperature is controlled around 600 - 900ºC by “flat” temperature zone through using multiple heaters • Low gas pressure reduce gas-phase reaction which causes particle cluster that contaminants the wafer and system Applied Physics 298r 31 E. Chen (4-12-2004) Plasma-Enhanced CVD (PECVD) RF • Use rf-induced plasma (as in sputtering ~ Gases case) to transfer energy into the reactant gases, forming radicals (decomposition) • Low temperature process (< 300 ºC) • For depositing film on metals and other materials that cannot sustain high eA temperature Shaw Heads B e- eA+ B+ e- • Surface reaction limited deposition; substrate temperature control (typically Substrate cooling) is important to ensure uniformity Applied Physics 298r 32 E. Chen (4-12-2004) Common CVD Reactants Material LPCVD PECVD α-Si SiH4 SiH4 SiH2Cl2 SiO2 Si(OC2H5)4 (TEOS) SiH2Cl2 + N2O SiH4 + N2O SiH4 + O2 Si3N4 SiH4 + NH3 SH2Cl2 + NH3 SiH4 + NH3 SiH4 + N2 Applied Physics 298r 33 E. Chen (4-12-2004) Comparison of Typical Thin Film Deposition Technology Process Material Uniformity Impurity Grain Size Film Density Deposition Rate Substrate Temperature Directional Cost Thermal Evaporation Metal or low meltingpoint materials Poor High 10 ~ 100 nm Poor 1 ~ 20 A/s 50 ~ 100 ºC Yes Very low Poor Low 10 ~ 100 nm Poor 10 ~ 100 A/s 50 ~ 100 ºC Yes High ~ 200 ºC Some degree High E-beam Evaporation Both metal and dielectrics Sputtering Both metal and dielectrics Very good Low ~ 10 nm Good Metal: ~ 100 A/s Dielectric: ~ 1-10 A/s PECVD Mainly Dielectrics Good Very low 10 ~ 100 nm Good 10 - 100 A/s 200 ~ 300 ºC Some degree Very High LPCVD Mainly Dielectrics Very Good Very low 1 ~ 10 nm Excellent 10 - 100 A/s 600 ~ 1200 ºC Isotropic Very High Applied Physics 298r 34 E. Chen (4-12-2004)