Dalton`s Atomic Theory 1) All matter is made of atoms. Atoms are
advertisement

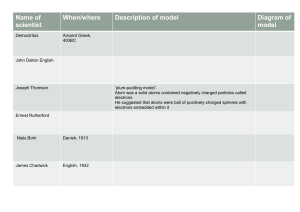
Mass Reactants = Mass products Proust expanded on LaVoisier’s experiments. (1797) •He decomposed CuCO3 into Cu + C + O and measured the masses of each separately. • The ratio of the masses were always 1:1:3 regardless of the sample source or size. John Dalton 1808 Dalton's Atomic Theory 1) All matter is made of atoms. 2.) Atoms are indivisible and indestructible. Dalton's Atomic Theory 3) All atoms of a given element are identical in size, mass and properties. Dalton's Atomic Theory 4) Compounds are formed by atoms that combine in whole numbers. Dalton's Atomic Theory 5) A chemical reaction is a rearrangement of atoms. + + 1890 Cathode Ray Experiments JJ Thomson (1897) • Plum pudding model • Cathode ray tube—electrons can be attracted and repelled by a magnet • Tiny negative particles stuck inside a sphere of positively charged stuff. Robert Millikan The Oil Drop Experiment (1909) It allowed the mass & charge of an electron to be determined. http://www.youtube.com/watch?v=XMfYHag7Liw Ernest Rutherford (1911) http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/ruther14.swf Rutherford Model of the Atom + Niels Bohr (1913) • Electrons have specific levels that they orbit around the nucleus. • Can see this in the colors that different elements emit deBroglie or Quantum Model of the Atom Electrons move as waves in orbitals. James Chadwick (1932) • While measuring the mass of atoms, discovered the neutron in the nucleus