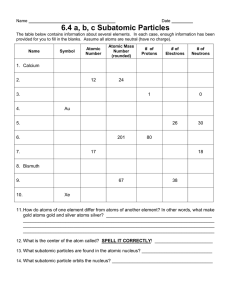
Notes: Atomic Theory (Ch3, Sec 1 & 2) An Ancient _________________ Philosopher Democritus 1. About ____________ 2. Thought that _____________ was composed of ___________ indivisible particles (atomos = ___________________) 3. He called them ______________. John Dalton’s model In the 1700’s, improved _________________ and experimentation allowed scientists to come up with some ____________ laws… Law of conservation of mass (_______________): 1)Mass(matter) is NOT created or destroyed in ordinary chemical and physical ____________. Ex. 2H2 + 20g + O2 2H2O X Ex. 2Na + 2H20 2NaOH + H2 30g 35g + X 65g + 40g 2)Law of _________________Proportions A chemical ________________ contains the ___________ proportions by mass no matter the size or source or the sample. Always the same ____________. In the year _____________ John Dalton __________________these laws using experimental _____________ of other scientists. Dalton proposed an atomic ______________ that included ________ _______________: 1. All _____________ is composed of tiny particles called ___________. 2. Atoms of the ____________ element are ______________; atoms of different elements are _______________. 3. Atoms cannot be __________________, created, or destroyed 4. Atoms of different elements combine in simple ______________ ______________ ratios to form a compound 5. In a chemical ________________, atoms are ______________, separated, or rearranged, but not __________________. Two of Dalton’s ___________ were proven ___________: # 2: atoms of the ______ element _____ be different (____________) # 3: atoms are __________________. A “picture” of Dalton’s ____________ (called the___________________ Ball) Today we define the atom as the _________________ particle of an _________________ that retains the _________________ of that element. The subatomic particles are: proton - ___________________ charged; in the _______________ neutron - ________________; in the ________________ electron - ________________ charged;_________________the nucleus JJ Thompson discoverer of the __________________in __________. (______________________ ______________) Studied the results of running _________________ currents through small amounts of _________ in a sealed glass tube - cathode _______ tube (______). He noticed a ____________ of particles he called a _________________ ray that: 1.cast a ______________ if an object was placed in their ___________ 2. had enough ___________ to move a small paddle _____________ 3. were deflected by a _________________ field the same way as a wire carrying _______________ current (electric current was known to have a ________________ charge) 4. were deflected ___________ from a ___________________ charged object (like charges _____________). From these experiments, Thomson concluded: 5. cathode rays are __________________. 6. cathode rays always have the ___________ charge to mass __________. Later these cathode rays were named __________________ (the ____________ subatomic particle discovered) A “picture” of Thomson’s model: (called the plum _______________ model) Because these guys learned about electrons, two other assumptions could be made: 1. Since atoms are electrically neutral, they must contain a _______________charge to balance the negative electrons. 2. Because electrons have such low mass, there must be _____________ __________________ to make up for the mass. Ernest Rutherford _____________, discovered the________________. Bombarded gold foil with alpha particles (positive He) He expected the alpha particles (Helium particles) to go ______________ the foil, but some of the alpha particles _________________ back. Rutherford concluded that atoms contain a ____________, dense ____________________ -charged area and the ________________ move around this _________________ like planets around the sun. Neils Bohr________ Electrons circle the nucleus in allowed paths or ___________. Electrons absorb _______________ to get to farther orbits and _________ ______ energy when returning to original orbits. Electron Cloud Model (Current Model) Erwin Schrodinger ___________ Quantum mechanics electrons can on exist in specified energy states. Electron cloud model _____________: 3-d region around the nucleus where e- are likely to be found. James Chadwick _________ Discovered neutrons - ___________ particles in the nucleus that accounted for the additional mass of an atom. (neutrons & protons make up the mass of an atom)