IONIC COMPOUNDS Aqueous Solutions Aqueous Solutions
advertisement
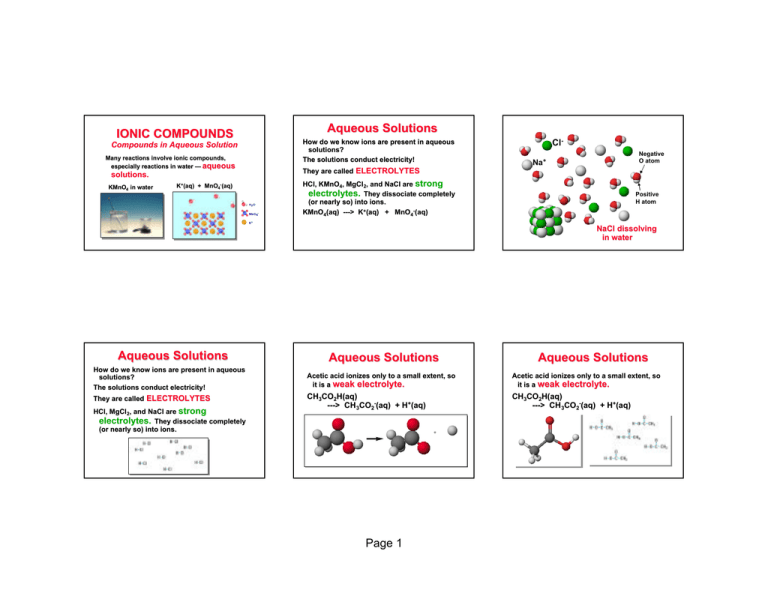
IONIC COMPOUNDS Compounds in Aqueous Solution Many reactions involve ionic compounds, especially reactions in water — aqueous solutions. KMnO4 in water K+(aq) aq) + MnO4-(aq) aq) Aqueous Solutions How do we know ions are present in aqueous solutions? The solutions conduct electricity! ClNegative O atom Na+ They are called ELECTROLYTES HCl, HCl, KMnO4, MgCl 2, and NaCl are strong electrolytes. They dissociate completely (or nearly so) into ions. KMnO4(aq) aq) ---> K+(aq) aq) + MnO4-(aq) aq) Positive H atom NaCl dissolving in water Aqueous Solutions Aqueous Solutions Aqueous Solutions How do we know ions are present in aqueous solutions? The solutions conduct electricity! Acetic acid ionizes only to a small extent, so it is a weak electrolyte. Acetic acid ionizes only to a small extent, so it is a weak electrolyte. They are called ELECTROLYTES CH3CO2H(aq H(aq)) ---> CH 3CO2-(aq) aq) + H+(aq) aq) CH3CO2H(aq H(aq)) ---> CH 3CO2-(aq) aq) + H+(aq) aq) HCl, HCl, MgCl 2, and NaCl are strong electrolytes. They dissociate completely (or nearly so) into ions. + Page 1 Acetic acid — Weak Electrolyte Ionized acetic acid H+ WATER SOLUBILITY OF IONIC COMPOUNDS Not all ionic compounds dissolve in water. Some are INSOLUBLE . See Screen 4.6 and Figure 5.4 Aqueous Solutions Nonelectrolyte— Ethanol, C 2H5OH Some compounds dissolve in water but do not conduct electricity. They are called nonelectrolytes. nonelectrolytes. Examples include: sugar ethanol ethylene glycol (in antifreeze) WATER SOLUBILITY OF IONIC COMPOUNDS Common minerals are often formed with anions that lead to insolubility: sulfide fluoride carbonate oxide As long as one ion from the list is present in a compound, the compound is water soluble. Iron pyrite, a sulfide Azurite, a copper carbonate Page 2 Orpiment, arsenic sulfide ACIDS ACIDS An An acid acid -------> -------> H H++ in in water water Some strong acids are HCl hydrochloric H2SO4 sulfuric HClO 4 perchloric HNO3 nitric HNO3 ACIDS ACIDS An An acid acid -------> -------> H H++ in in water water HCl( HCl(aq) aq) ---> H+(aq) aq) + Cl-(aq) aq) Weak Weak Acids Acids The The Nature Nature of of Water Water HCl Cl- H2O hydronium ion ACIDS ACIDS H3O+ The The Nature Nature of of Acids Acids BASES see see Screen Screen 4.8 4.8 and and Table Table 4.1 4.1 Nonmetal oxides can be acids CO2(aq) aq) + H2O(liq O(liq)) ---> H 2CO3(aq) aq) SO3(aq) aq) + H2O(liq O(liq)) ---> H 2SO4(aq) aq) and can come from burning coal and oil. Base Base ---> ---> OH OH-- in in water water NaOH( NaOH(aq) aq) ---> Na+(aq) aq) + OH-(aq) aq) NaOH is a strong base Page 3 WEAK ACIDS = weak electrolytes CH3CO2H acetic acid H2CO3 carbonic acid H3PO4 phosphoric acid Ammonia, NH3 An Important Base NH3(aq) aq) + H 2O(liq O(liq)) ---> NH 4+( aq) aq) + OH -(aq) aq) BASES BASES Metal oxides are bases CaO(s) CaO(s) + H 2O(liq O(liq)) --> Ca(OH) 2(aq) aq) Net Ionic Equations Mg(s) + 2 HCl( HCl(aq) aq) --> H2(g) + MgCl 2(aq) aq) Net Net Ionic Ionic Equations Equations Mg(s) + 2 HCl( HCl(aq) aq) --> H2(g) + MgCl 2(aq) aq) Mg(s) + 2 H +(aq) aq) + 2 Cl-(aq) aq) ---> H 2(g) + Mg 2+(aq) aq) + 2 Cl-(aq) aq) We really should write Mg(s) + 2 H +(aq) aq) + 2 Cl-(aq) aq) ---> H2(g) + Mg 2+(aq) aq) + 2 Cl-(aq) aq) CaO in water. Indicator shows solution is basic. The two Cl- ions are SPECTATOR IONS — they do not participate. Could have used NO 3-. Page 4 We leave the spectator ions out — Mg(s) + 2 H+(aq) aq) ---> H 2(g) + Mg2+(aq) aq) to give the NET IONIC EQUATION