ELEMENTS, COMPOUNDS, MIXTURES VOCABULARY mixture Two or more substances that aren't chemically
advertisement
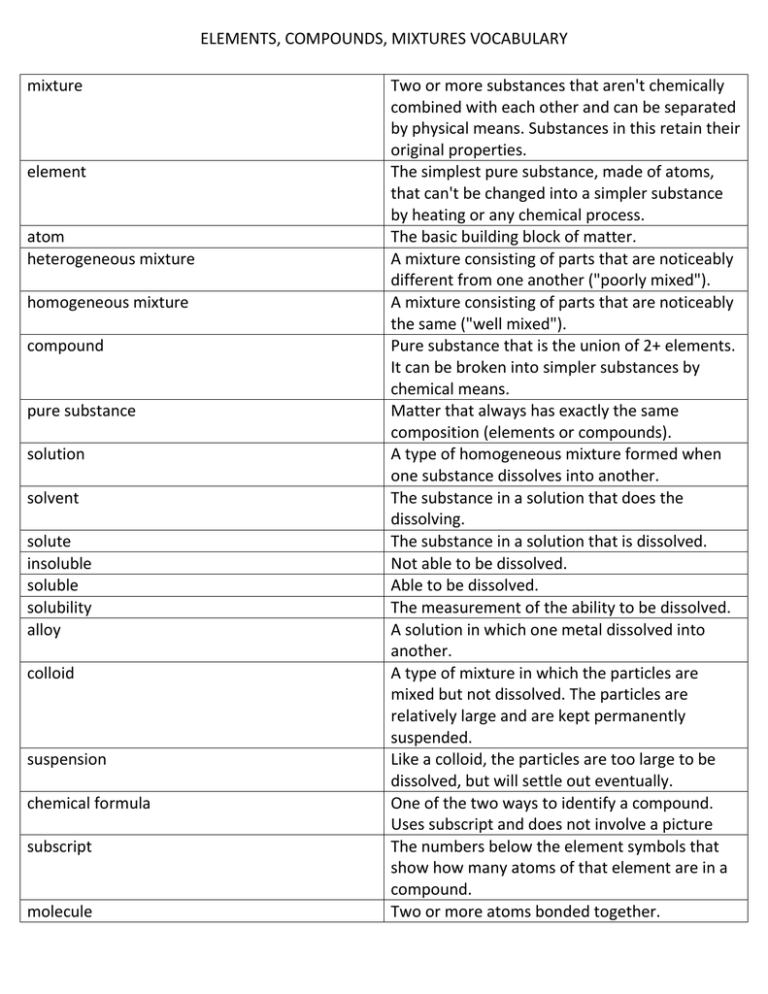
ELEMENTS, COMPOUNDS, MIXTURES VOCABULARY mixture element atom heterogeneous mixture homogeneous mixture compound pure substance solution solvent solute insoluble soluble solubility alloy colloid suspension chemical formula subscript molecule Two or more substances that aren't chemically combined with each other and can be separated by physical means. Substances in this retain their original properties. The simplest pure substance, made of atoms, that can't be changed into a simpler substance by heating or any chemical process. The basic building block of matter. A mixture consisting of parts that are noticeably different from one another ("poorly mixed"). A mixture consisting of parts that are noticeably the same ("well mixed"). Pure substance that is the union of 2+ elements. It can be broken into simpler substances by chemical means. Matter that always has exactly the same composition (elements or compounds). A type of homogeneous mixture formed when one substance dissolves into another. The substance in a solution that does the dissolving. The substance in a solution that is dissolved. Not able to be dissolved. Able to be dissolved. The measurement of the ability to be dissolved. A solution in which one metal dissolved into another. A type of mixture in which the particles are mixed but not dissolved. The particles are relatively large and are kept permanently suspended. Like a colloid, the particles are too large to be dissolved, but will settle out eventually. One of the two ways to identify a compound. Uses subscript and does not involve a picture The numbers below the element symbols that show how many atoms of that element are in a compound. Two or more atoms bonded together.


