SI units/Metric
advertisement

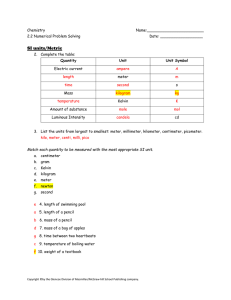
Chemistry Name:________________________ 2.2 Numerical Problem Solving Date: __________________ SI units/Metric 2. Complete the table: Quantity Unit Unit Symbol Electric current meter s Mass Kelvin Amount of substance Luminous Intensity cd 3. List the units from largest to smallest: meter, millimeter, kilometer, centimeter, picometer. ____________________________________________________________________ Match each quantity to be measured with the most appropriate SI unit. a. centimeter b. gram c. Kelvin d. kilogram e. meter f. newton g. second ___4. length of swimming pool ___ 5. length of a pencil ___ 6. mass of a pencil ___ 7. mass of a bag of apples ___ 8. time between two heartbeats ___ 9. temperature of boiling water ___10. weight of a textbook Copyright ©by the Glencoe Division of Macmillan/McGraw-hill School Publishing company. SIGNIFICANT FIGURES 11. For each group of digits indicated in the three numbers below, state whether the digits are significant. State the rule that applies. 4500.60 0.000799 220 a. ______________________________________________________________________ b. _____________________________________________________________________ c. ______________________________________________________________________ d. _____________________________________________________________________ e. ______________________________________________________________________ f. ______________________________________________________________________ 12. A stack of books contains 10 books, each of which is determined, by a ruler graduated in centimeters, to be 25.0 cm long. How do these two quantities differ in terms of significant digits? _______________________________________________________________________ ______________________________________________________________________ Percent Error 13. What is the percent error in a determination that yields a value of 8.38 g/cm 3 as the density of copper? The literature value for this quantity is 8.92 g/cm3. 14. Yusuf measures the melting point of ammonium acetate, NH 4C2H3O2, as 117 ˚C, but the literature value is 114 ˚C. What is the percent error in the measurement? Copyright ©by the Glencoe Division of Macmillan/McGraw-hill School Publishing company. Copyright ©by the Glencoe Division of Macmillan/McGraw-hill School Publishing company.