“Bohr”ing you with atoms The study of chemistry begins with .
advertisement

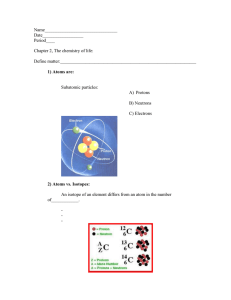
“Bohr”ing you with atoms The study of chemistry begins with the basic unit of matter, the atom. What is matter? Anything that has mass and takes up space. The subatomic particles that make up atoms are Protons (+) are positive located in the center of atom Neutrons (=) neutral located in the center of the atom Electrons (-) negative and are located in space around the nucleus. They are 1/1840 the size of a proton. Because protons and electrons have equal and opposite charges, atoms are neutral. Elements and Isotopes Element is one type of atom. Their are over 100 elements in existence, but only a few are fond in living organisms. Atom Builder 6 C 12.011 Carbon Isotopes Are atoms of the same element with a different number of neutrons. All 3 isotopes have the same chemical properties, because they all have the same number of electrons. Nonradioactive carbon-12 Nonradioactive carbon-13 6 electrons 6 protons 6 neutrons 6 electrons 6 protons 7 neutrons Radioactive carbon-14 6 electrons 6 protons 8 neutrons How radioactive isotopes work When an organism is alive it constantly absorbing (C-14). However when an organism dies the C14 begins to break down, ½ the quantity of death every 5,730 years. Web site (ingest) Radio Active Isotopes Have unstable nuclei and break down at a constant rate over time. Geologist use radioactive isotopes to determine the age of a fossil. (C-14) Other Radioactive Isotopes Can be used To treat cancer Kill bacteria As “tracers” Chemical Compounds Are formed when molecules of two or more atoms are joined together. The chemical properties of a compound are very different from the elements it is made up of. Two Types of Compounds Inorganic compounds Lack Carbon Non living For example water is made up of 2 hydrogen’s and 1 oxygen. Organic compounds Contain Carbon Must come from other living things. Sodium and Chloride Are very different elements. Chlorine (Cl) is a poisonous green gas that was used to kill soldiers in WWI. Sodium (Na) – is a silvery metal. Chemical Bonds Atoms in compounds are held together by chemical bonds. Bond formation involves the electrons that surround each nucleus known as valance electrons. Ionic Bonds An Ionic bond is formed when one or more electrons are transferred from one atom to the other. An atom that loses an electron has a (+) charge. An atom that gains an electron has a (-) charge. Positively or negatively charged atoms are known as ions. Ionic bonds are not very strong. Ionic Bonds: One big greedy thief Ionic bonding can be best dog imagined as one big greedy dog steeling the other dog’s bone. The bones represent the electrons up for grabs. When the big dog gains an electron he becomes negative, and when the little dog looses an electron he becomes positive. The two ions are attracted very strongly to each other as a result of the opposite charges. Covalent Bonds Covalent bond is formed when electrons are shared between atoms. Very strong bond. Covalent Bond: Dogs of equal strength Covalent bonds can be thought as two or more dogs with equal attraction to the bones. Since the dogs (atoms) are identical, then the dogs share the pairs of available bones evenly. Molecules When atoms are joined together by a covalent bond, they form molecules. The molecule is the smallest unit of most compounds. Van der Waals forces Some atoms have a stronger attraction fore electrons than do other atoms. There for, when atoms in a covalent bond share electrons their not always equal. A slight attraction can develop between the oppositely charged regions of nearby molecules. The results are ever changing “hot spots” of positive and negative charge that enable all atoms to stick together. Van der Waals forces in Action A geckos foot is covered by as many as a ½ a million tiny hair like projections. Each projection is divided into hundreds of tiny flat –surfaced fibers. This design allows the gecko’s foot to come in contact with an extremely large area of the wall at the molecular level allowing Van’s forces to occur. The combined strength of all the Van der Waals froces allows the gecko to walk up walls. Polar Covalent Bonds A type of covalent bond that differs in electronegativity. The electrons are pulled closer to the larger atom, making it slightly negative and the other slightly positive An example of a polar molecule is water. Hydrogen Bonding Water is a polar molecule. Water has a partial positive an negative charge. The (O) is (-) and the hydrogen’s are (+). Hydrogen bonds are weak. Shock wave demo Match the Bonds Water has strong cohesion and high surface tension Cohesion- is the attraction between molecules of the same substance. A single water molecule may be involved in as many as 4 hydrogen bonds at the same time. Cohesion explains why some insects can walk on water. Water adheres to other molecules Adhesion is an attraction between molecules of different substances. The surface water in a graduate cylinder dips slightly in the center because the adhesion between water molecules and glass is stronger than the cohesion between water molecule. Ice Floats Unlike most substances when water freezes it expands, and becomes less dense. What effects would that have on life? Water is an excellent solvent Water is a solvent – the substance in which the solute is dissolved. In a salt-water solution, table salt is the solute. Water is the solvent of life. Dissolve demo Water has a high heat capacity Water changes temperature very slowly. Thus the temperature of large bodies of water are very stable. Evaporative Cooling As water evaporates, the surface of the water that remains behind cools down. Why do we sweat more on a humid day? Mr. Walcott before a job interview What gives water its special properties Individual water molecules are polar and they stick together by means of hydrogen bonds. Hydrogen bonds are weak and only last a fraction of a second. It takes a lot of energy to overcome the combined attraction of many hydrogen bonds. This explains Waters great capacity to store heat High boiling point Surface tension Acid, Bases, and pH Water Hydrogen Ion (H+) Hydroxide Ion (OH-) Acids (H+) An acids from H+ ions in solutions have more H+ ions than pure water pH values below 7 Bases (OH-) Produce hydroxide ions (OH-) Have lower amounts of H+ ions then pure water. pH values above 7 Buffers Weak acids or bases that can react with strong acids or bases to prevent sudden changes in pH. The human body has a buffer system. The most common buffer is the bicarbonate ion. The pH scale The pH scale tells us how many H+ ions there are in a solution. The higher the pH the more basic the solution The lower the pH the more acidic a solution is. pH of 7 is neutral pH scale animation What to do when you get stung When a wasp stings it injects an alkali (base) into the victim You can neutralize the sting with vinegar (an acid) Tums Are a base Neutralize the stomachs acids.