Fall, 2005 Exam I Name:____________________________
advertisement

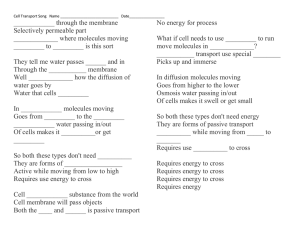
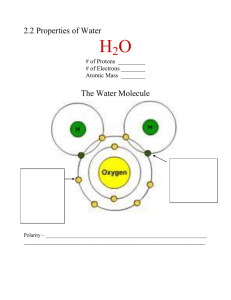
Fall, 2005 Exam I Name:____________________________ Pad Number__________ Multiple choice. Choose the BEST answer. 1. Which of these terms refers to the process of maintaining relatively constant internal conditions in a living organism? a. Order. b. Sensitivity c. Development d. Homeostasis e. Regulation 2. The cation form of an element is compared to the uncharged form of the same element. Which of the following statements is TRUE? a. The cation has less protons. b. The cation has a different half-life c. The cation has more neutrons. d. The cation has less electrons. e. The cation has more electrons. 3. Organisms that can extract energy from light are called __________; organisms that must obtain energy from molecules made by other organisms are called __________. a. Herbivores, carnivores b. Photosynthetic, herbivores c. Heterotroph, autotroph d. Autotroph, heterotroph 4. A solution with neutral pH would have a pH value of: a. 0.0 b. 1.0 c. 7.0 d. 10.0 e. 14.0 5. Isotopes have the same number of ____________, but different number of: a. Electrons, atoms b. Protons, electrons c. Protons, neutrons d. Neutrons, ions e. Ions, molecules 6. Two atoms share 2 electrons. This is an example of: a. a single bond. b. a double bond. c. a triple bond. d. a hydrogen bond. e. None of the above is a correct answer. 7. Which of the following will increase the rate of a chemical reaction? a. Higher temperature b. Higher reactant concentration c. A catalyst d. All of the above speeds up a chemical reaction. e. None of the above speeds up a chemical reaction. 8. Water is a liquid at room temperature (and not a gas) because: a. it can form ionic bonds between ionized molecules. b. it can form hydrogen bonds between individual molecules. c. it can dissociate to form hydrogen and hydroxide ions. d. it has a relatively high molecular weight compared to gases. e. it forms a strong hydration shell. 9. A crystal of sodium chloride is held together by: a. hydrogen bonds. b. ionic bonds. c. covalent bonds. d. hydration shells of the individual molecule. e. a chemical reaction between the sodium and chloride ions. 10. Which of the above properties of water was/is not important to the development of living systems? a. Cohesion—water molecules “stick together” b. High specific heat--water requires much energy to raise its temperature c. High heat of vaporization--much energy absorbed when water evaporates d. Low density of ice—it floats e. All of the above are properties of water that are important to the development and maintenance of life on this planet. 11. The basic monomeric repeat unit which is used to assemble nucleic acids is the: a. amino acid b. sugar c. lipid d. nucleotide e. phosphate 12. An example of a structural polymer made entirely of glucose repeat units is/are: a. The cell membrane. b. c. d. e. Microtubules. The nuclear membrane. A ribosome. None of the above answers is/are correct. 13. The type of reaction commonly used to disconnect (break apart) the monomeric units of macromolecules is best classified as a/an: a. exergonic reaction. b. hydrolysis reaction. c. disulfide reaction. d. hydrogen bonding. e. dehydration reaction. 14. The actual sequence of amino acids in a polypeptide/protein is called its: a. primary structure. b. secondary structure. c. tertiary structure. d. quaternary structure. e. None of the above are correct answers. 15. An example of a type of chemical bond which involves a full separation of charge (transfer of electrons) is a/an: a. hydrogen bond. b. ionic bond. c. covalent bond. d. hydrophobic bond. e. electron bond. 16. A storage fat molecule is made up of: a. two fatty acids, glycerol, phosphate and another alcohol type unit. b. three fatty acids and glycerol. c. two fatty acids and glycerol. d. two fatty acids, glycerol and a phosphate group. e. three fatty acids, glycerol, phosphate and an alcohol type unit. 17. Saturated fats_____________. a. Have no double covalent bonds b. Are solid at room temperature c. Contain the maximum number of hydrogen atoms possible d. All of the above e. None of the above 18. All biomolecules are synthesized by __________________. a. Dehydration reactions b. Hydrolysis reactions c. Peptide bonds d. Hydrogen bonds e. Chemical biodegradation 19. The beta-sheet and alpha-helix of a folded protein/polypeptide would be examples of its: a. primary structure. b. secondary structure. c. tertiary structure. d. quaternary structure. e. All of the above are correct answers. 20. An example of a storage carbohydrate common in plants is: a. cellulose. b. glycogen. c. starch. d. chitin. e. choline. 21. The atomic number of an element refers to the: a. number of protons in the nucleus. b. number of protons plus neutrons in the nucleus. c. number of neutrons in the nucleus. d. number of electrons in the outer shell/orbital. e. number of covalent bonds it routinely makes with other atoms. 22. (CH2O)n is the common formula for: a. amino acids. b. sugars. c. fatty acids. d. hydrophylic molecules. e. hydrophobic molecules. 23. The charged particle with a mass of approximately 1 in the nuclei of atoms is called a/an: a. neutron b. proton c. nucleus d. electron e. neutrino 24. Biological membranes are composed of [choose the best answer]: a. fats. b. a peptidoglycan layer. c. a phospholipid bilayer. d. Carbohydrate fibers related to cellulose. e. All of the above are correct answers. 25. Ribose is an example of a/an: a. purine. b. pyrimidine. c. sugar. d. aldehyde. e. fat. 26. Which of the following is a nucleotide? a. ATP b. A fat c. A sugar d. A phospholipid e. Cholesterol 27. The monomer repeat units of a protein are connected to each other by what type of bonds? a. Peptide bonds b. Phosphodiester bonds c. Ester bonds d. Hydrogen bonds e. Ionic bonds 28. What is the defining characteristic of an acid? a. It donates hydrogen ions. b. It has an excess of hydroxide ions c. It accepts hydrogen ions. d. it has a pH greater than 7. e. It will donate or accept hydrogen ions depending on the pH. 29. If two atoms share a pair of electrons, they are held together by a(n) a. polar bond b. hydrogen bond c. neutral bond d. covalent bond e. ionic bond 30. Which of the following is a CORRECT statement about the scientific method? a. it distinguishes between good and bad b. it can only be done by someone with a Ph.D. or advanced training c. it organizes evidence and helps us predict what will happen in our environment d. it requires expensive laboratory equipment e. its methods are substantially different from the way people normally process information 31. Based upon your knowledge of atomic structure, an atom with 14 protons in its nucleus a. would be very stable and unreactive b. would be very positively charged c. tend to form covalent bonds d. would probably form ionic bonds e. none of the above 32. Each amino acid differs from others in the a. location of the carbon atoms b. nature of the R group c. number of carboxyl groups d. all of the above e. size of the amino group 33. The bonds between two carbon atoms or carbon and hydrogen represents a. shared electrons b. energy sinkholes c. both a and d d. slurpy factoids e. ionic bonds 34. Which of the following lists the four elements that make up 97% of all living tissue? a. hydrogen, carbon, nitrogen, oxygen b. carbon, hydrogen, nitrogen, sulfur c. oxygen, carbon, helium, nitrogen d. nitrogen, carbon, helium, sulfur e. oxygen, nitrogen, carbon, sulfur 35. A pH of 8 is _____ times more ________ than a pH of 7. a. 10, basic b. 100, basic c. 10, acid d. 1000, acid e. none of the above 36. You drink a glass of lemonade, but your body's pH does not change. This is an example of how organisms: a. maintain homeostasis. b. are immune to acid c. maintain organization d. are what they eat. e. adapt to their environment. The ________ portion of the cell membrane is responsible for the isolating functions of the membrane, while the ________ portion regulates exchange and communication with the environment. A) lipid; protein B) cholesterol; lipid C) protein; cholesterol D) carbohydrate; lipid E) nucleic acid; lipid 37. 38. In biological membranes, the phospholipids are arranged in A) a bilayer with the fatty acids pointing toward each other. B) a bilayer with the fatty acids facing outward. C) a single layer with the fatty acids facing the interior of the cell. D) a single layer with the phosphorus-containing region facing the interior E) a bilayer with the phosphorus groups in the interior of the membrane. 39. The hydrophilic regions of a membrane protein are most likely to A) only in muscle cell membranes. B) associated with the fatty acid region of the lipids. C) in the interior of the membrane. D) exposed on the surface of the membrane. E) attached to carbohydrates and facing the interior of the membrane. 40. What structure in the membrane causes A) polar heads of the phospholipids B) channel proteins C) cholesterol D) saturated fatty acid tails E) unsaturated fatty acid tails of the cell. be found plasma membranes to resist freezing? In general, which of the following is largely responsible for moving substances across the plasma membrane, communicating with other cells and identifying the cell? A) phospholipids B) carbohydrates C) proteins D) nucleic acids E) cytoskeleton 41. 42. What prevents your A) inadequate enzymes B) fever C) cholesterol D) recognition proteins E) receptor proteins immune system from attacking your own cells? 43. What is active transport? A) diffusion of molecules within a cell B) movement of molecules into or out of a cell against a concentration gradient C) movement of molecules into or out of a cell down a concentration gradient D) the movement of molecules into or out of a cell using special proteins and not expenditure of energy E) rapid movement of molecules in a solution requiring an When substances move through a plasma membrane and down gradients of concentration this is called A) active transport. B) passive transport. C) pinocytosis. D) exocytosis. E) entropy. 44. 45. Molecules which permeate a plasma membrane by facilitated diffusion A) require an expenditure of energy. B) require the aid of transport proteins. C) move from an area of low concentration to an area of high concentration. D) do so much more quickly than those crossing by simple diffusion. E) all of these 46. A molecule that can A) hydrophilic. B) positively charged. C) hydrophobic. D) negatively charged. E) a sugar. diffuse freely through a phospholipid bilayer is probably 47. Molecules assisted by carrier A) facilitated diffusion. B) active transport. C) osmosis. D) endocytosis. E) simple diffusion. 48. The diffusion of water A) facilitated diffusion. B) hydrolysis. C) active transport. D) exocytosis. E) osmosis. proteins may cross a differentially permeable membrane by molecules across a differentially permeable membrane is termed If red blood cells are taken from the body and placed in a hypertonic solution, what happens to the cells? A) The cells swell and burst because water moves into the cells. B) The cells shrivel up because water leaves the cells. C) The cells remain unchanged due to equal solute concentration inside and outside the cells. D) The cells remain unchanged due to equal water concentrations inside and outside the cells. E) They become white blood cells. 49. Two aqueous solutions are separated by a semipermeable membrane. Solution A is 10% starch and solution B is 5% starch. What will occur? A) Water will diffuse from solution A to solution B. B) Water will diffuse from solution B to solution A. C) Starch will diffuse from solution A to solution B. D) Starch will diffuse from solution B to solution A. E) Both B and D will occur. 50 51. Active transport requires A) transport proteins. B) ATP. C) a membrane. D) a gradient. E) all of these 52. Which process accounts A) active transport B) facilitated diffusion C) diffusion D) osmosis E) phagocytosis for the movement of solids into some animal cells? 53. What does a cell use exocytosis for? A) to move away from danger B) to release substances from the cell C) to incorporate nutrients D) to pump hydrogen molecules across the E) to create new cells 54. The electric signal membrane for a muscle to contract passes rapidly from one muscle cell to the next by way of A) tight junctions. B) desmosomes. C) gap junctions. D) internal proteins. E) external proteins. 55. Adhesion of animal A) desmosomes. B) tight junctions. C) gap junctions. D) plasmodesmata. E) cell plates. tissues is accomplished by cell-to-cell junctions called 56. The urinary bladder A) desmosomes. B) tight junctions. is protected from leaking due to cell-to-cell junctions called C) gap junctions. D) plasmodesmata. E) stretch receptors. Protein channels that provide passage for hormones and nutrients between animal cells are termed A) desmosomes. B) tight junctions. C) gap junctions. D) plasmodesmata. E) capillaries. 57. 58. What is a major component A) chitin B) cellulose C) pectin D) lignin E) protein Answer: B of primary cell walls of plants? _______________________________________________________________________ Match the following names to the proper functional group a. -OH d. -COOH b. -SH e. -NH2 c. -PO4 59. A sulfhydryl group ______ 60. An amino group ______ 61. A carboxyl group ______ 62. A hydroxyl group ______ _______________________________________________________________________ Continue in next page … Matching. Place the letter of the correct answer into the blank to the left of the term. 63. glucose ______ 64. steroid _____ 65. ATP _____ 66. phospholipids _____ 67. amino acid _____ (B) (A) (C) (D) (E) Continue to next page … Label the parts of the microscope: 68. Objective _____ 69. Fine adjustment knob _____ 70. stage _____