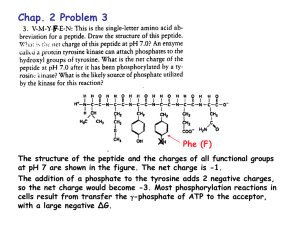
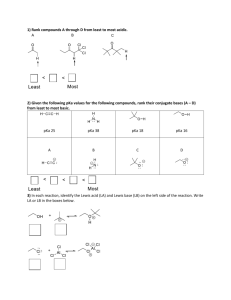
Chapter 2 Problem Set
advertisement

Chap. 2. Problem 2a. pH = - log [H+] pH = - log (1.75 x 10-5) pH = - (-4.76) pH = 4.76 Chap. 2. Problem 3a. pH = - log [H+] [H+] = 10-pH [H+] = 10-3.82 [H+] = 1.51 x 10-4 M Chap. 2. Problem 12. Part a. Acetic acid ionizes according to the following equilibrium equation HAc H+ + AcThe Henderson-Hasselbalch equation for acetic acid is pH = pKa + log [Ac-]/[HA] Substituting pKa = 4.76 and 2/1 for [Ac-]/[HA] gives pH = 4.76 + log (2/1) pH = 4.76 + 0.30 pH = 5.06 Other answers: (b) 4.28; (c) 5.46; (d) 4.76; (e) 3.76 Chap. 2. Problem 15. Ionizable proton A rearranged form of the HH equation can be used to determine the fraction of aspirin in its absorbable conjugate acid form (HA) as a function of pH in the stomach and intestines. In the stomach, 10(pH - pKa) = [A-]/[HA] 10(1.5 - 3.5) = [A-]/[HA] 10-2 = 0.01 = 1/100 = [A]/[HA] In the small intestine, 10(6 - 3.5) = [A-]/[HA] 102.5 = 316/1 = [A-]/[HA] The calculations show that a much greater fraction of aspirin is in its absorbable conjugate acid form in the stomach compared to the small intestine. Thus more aspirin is absorbed into the bloodstream from the stomach than from the small intestine. Chap. 2. Problem 18a&b. Part (a). A solution of a weak acid or base is effective in buffering pH when the pH is ± 1 unit from the pKa. In this pH range, appreciable amounts of both conjugate base and conjugate acid species are present in solution. Therefore, glycine can be used as an effective buffer in the pH range of 8.6-to-10.6. Part (b). This part of the question can be answered using the HH equation. pH = pKa + log [R-NH2]/[R-NH3+] [R-NH2]/[R-NH3+] = 10(9.0-9.6) = 10-0.6 = 0.25 = 1/4 This indicates that the amino group of glycine is about 1/5 deprotonated and 4/5 protonated at pH 9.0. Chap. 2. Problem 18c. Part (c). This part of the problem can be answered by first calculating the number of moles of R-NH2 in 1 L of the 0.1 M glycine solution at pH 9.0 and 10.0 using the HH equation. The difference in the number of moles present is stoichiometrically equivalent to the number of moles of OH- added in the form of KOH. At pH = 9.0 10(9-9.6) = [R-NH2]/[R-NH3+] = 0.25 = 1/4 = 0.02 moles/0.08 moles. At pH = 10.0 10(10-9.6) = [R-NH2]/[R-NH3+] = 2.5 = 2.5/1 = 0.0714 moles/0.0286 moles. During titration from pH 9.0 to 10.0, the number of moles of KOH added is 0.0714 moles - 0.02 moles = 0.0514 moles KOH. Thus, the volume of 5 M KOH added is (0.0514 moles)/(5 moles/L) = 0.01028 L = 10.3 ml KOH Chap. 2. Problem 18d. Part (d). This part of the question also can be answered using the HH equation. pH = pKa + log [R-NH2]/[R-NH3+] pH = pKa + log (0.01/0.99) pH = pKa + (-2) = pKa - 2 In general, a weak acid or base is almost completely protonated when the solution pH is 2 units below the pKa. Likewise, it is almost completely deprotonated when the solution pH is 2 units above the pKa. Chap. 2. Problem 22. The equation for the complete ionization of phosphoric acid is H3PO4 H+ + H2PO4- H+ + HPO42- H+ + PO43This problem is solved using the HH equation. Note that the pKa closest to the pH of the solution should be selected for substitution into the HH equation. This is because the concentrations of H3PO4 and PO43- in solution at this pH are insignificant compared to the concentrations of H2PO4- and HPO42-. pH = pKa + log [conjugate base]/[conjugate acid] pH = pKa + log [HPO42-]/[H2PO4-] [HPO42-]/[H2PO4-] = 10(7.0-6.86) [HPO42-]/[H2PO4-] = 100.14 = 1.38 = 1.4 Chap. 2. Problem 24. pH = pKa + log [A-]/[HA] pH - pKa = log [A-]/[HA] 10(pH - pKa) = [A-]/[HA] 10(5-6) = 10-1 = 0.1 = 1/10 = [A-]/[HA] Note that this acid is 1/(10+1) = 1/11 = 9.1% ionized at this pH. Chap. 2. Problem 28. pH = pKa + log [conjugate base]/[conjugate acid] pH = pKa + log [acetate]/[acetic acid] pH = 4.76 + log (0.2/0.6) pH = 4.76 + log (0.33) pH = 4.76 + (-0.48) = 4.28 = 4.3 Chap. 2. Problem 33. The overall equation for the carbon dioxide-bicarbonate buffer system is CO2 + H2O H+ + HCO3The pH of blood plasma is controlled by this system. In Part (a), during hypoventilation, as when one holds one’s breath, the concentration of CO2 in the blood increases. This shifts the equation to the right and results in an increase in the acidity of blood (lower pH). In Part (b), during hyperventilation the concentration of CO2 in the blood decreases. This shifts the carbon dioxide-bicarbonate equation to the left, decreasing [H+] and raising the pH of the blood. In Part (c), raising the pH of the blood due to hyperventilation increases the ability of the blood to resist a decrease in pH due to the input of H+ into the blood from lactate.