lecture 5 - kinetic theory
advertisement

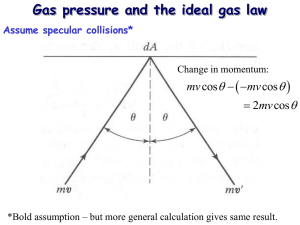
Announcements 9/9/11 Prayer Lab 1 due tomorrow Lab 2 starts tomorrow Worked problems: How much mass does the air in this room have? (MM 0.029 kg/mol) According to the ideal gas law, what is the density of air at 1 atm, for 300 K? For arbitrary T? A hot air balloon is 520 kg (including passengers). It’s spherical, with radius = 8 m. The temperature is 300K outside (80.3F), pressure is 1 atm. How hot does the pilot have to get the air inside the balloon for it to lift off? Some answers: 1.175 kg/m3; 378K (221F) Reading quiz (graded): Who was the famous Austrian physicist whose name was on two equations in today’s reading assignment? a. b. c. d. e. Niels Bohr Ludwig Boltzmann Johann Carl Friedrich Gauss Hermann von Helmholtz Erwin Schrödinger Thought question (ungraded): In air, the molecular mass of oxygen molecules is 32 g/mol; the molecular mass of nitrogen molecules is 28 g/mol. Which molecules are traveling faster on average? a. Oxygen b. Nitrogen c. Same speed Demo: heavy vs light molecules Equipartition Theorem “The total kinetic energy of a system is shared equally among all of its independent parts, on the average, once the system has reached thermal equilibrium.” “independent”: e.g. x, y, z (for translational KE) “parts”: translational, rotational, vibrational Specifically, each “degree of freedom”, of each molecule, has “thermal energy” of … ½kBT Thought quiz Compare a monatomic molecule such as Ne to a diatomic molecule such as O2. If they are at the same temperature(*), which has more kinetic energy? a. Ne b. O2 c. Same d. Not enough information to tell (*) let’s assume the temperature is “high”. Relative to what, we’ll discuss in a minute. Disclaimer Thermal energy (measured by kBT) must be comparable to the quantum energy levels, or some degrees of freedom get “frozen out” From section 21.4: diatomic hydrogen Y-axis: heat added, divided by temperature change (per mole) Units: J/molK Translational KE and vrms Worked problem: what is average speed (vrms) of oxygen molecules at 300K? Molecular View of Pressure Related problem: What is average pressure by baseballs (m = 145 g) on a wall (A = 9 m2). Speed = 85 mph (38 m/s). Elastic collisions, each lasting for 0.05 seconds. (This is the time the ball is in contact with the wall.) A baseball hits the wall every 0.5 seconds. Answer: 2.45 Pa Actual problem: a cube filled with gas a. Pressure on right wall from one molecule? Answer: 2mvx/(L2 tbetween hits) = mvx2/L3 b. Pressure on right wall from all molecules Answer: P = Nmvx2/V Molecular View of Pressure, cont. Result for v instead of vx: P = N m ⅓ v2 / V What does PV equal? Compare to: PV = N kB T What does v equal? What does T equal? What is temperature? (revisited) Demo Demo: kinetic theory machine Thought question: Which “molecules” have the most kinetic energy? a. The heavy ones b. The light ones c. Same (Repeat) Which ones have the fastest average velocity?