Point of Care Testing - ISTAT Data Definition Tool
advertisement

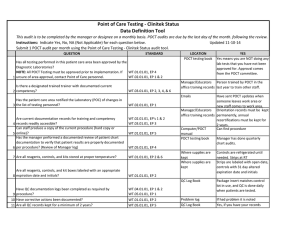
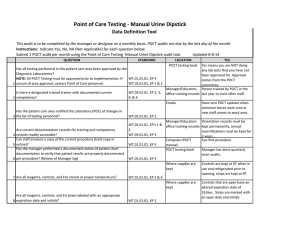
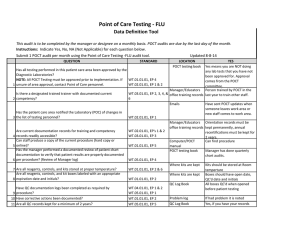
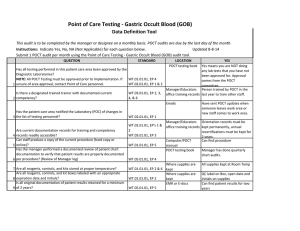
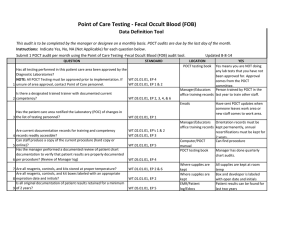
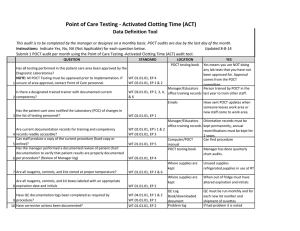
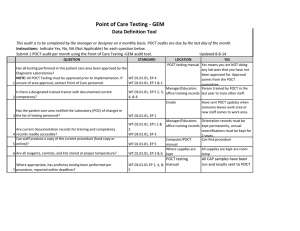
Point of Care Testing - ISTAT Data Definition Tool This audit is to be completed by the manager or designee on a monthly basis. POCT audits are due by the last day of the month. Instructions: Indicate Yes, No, NA (Not Applicable) for each question below. Updated 8-8-14 Submit 1 POCT audit per month using the Point of Care Testing -ISTAT audit tool. QUESTION 1 2 3 4 5 6 7 STANDARD LOCATION POCT testing book YES Yes means you are NOT doing Has all testing performed in this patient care area been approved by the any lab tests that you have not Diagnostic Laboratories? been approved for. Approval NOTE: All POCT Testing must be approved prior to implementation. If WT.01.01.01, EP 4 comes from the POCT unsure of area approval, contact Point of Care personnel. WT.02.01.01, EP 1 & 2 committee. Manager/Educators Person trained by POCT in the Is there a designated trained trainer with documented current WT.03.01.01, EP 2, 3, office training records last year to train other staff. competency? 4, & 6 Emails Have sent POCT updates when someone leaves work area or Has the patient care area notified the Laboratory (POC) of changes in new staff comes to work area. the list of testing personnel? WT.02.01.01, EP 1 Manager/Educators Orientation records must be WT.02.01.01, EP's 1 & office training records kept permanently, annual Are current documentation records for training and competency 2 recertifications must be kept for records readily accessible? WT.03.01.01, EP 3 2 years. Can staff produce a copy of the current procedure (hard copy or Computer/POCT Can find procedure online)? WT.01.01.01, EP 5 manual Has the manager performed a documented review of patient chart POCT testing book Manager has done quarterly documentation to verify that patient results are properly documented chart audits. per procedure? (Review of Manager log) WT.05.01.01, EP 4 Where supplies are Unused cartridges are stored in kept the refrigerator, and at RT when in use. Controls are stored refrigerated, or for 5 days at RT Are all reagents, controls, and kits stored at proper temperature? WT.01.01.01, EP 2 & 6 Are all reagents, controls, and kit boxes labeled with an appropriate 8 expiration date and initials? Have QC documentation logs been completed as required by 9 procedure? 10 Have corrective actions been documented? 11 Are all QC records kept for a minimum of 2 years? Where supplies are kept Cartridges are labeled with open date, altered date and initials. Controls labeled with altered date when removed from refrigerator and initials QC Log Book QC is performed as noted in the procedure depending on type of cartridge used If had problem it is noted Yes, if you have your records There is a signature to indicate reviewed weekly. WT.01.01.01, EP 2 WT.04.01.01, EP 1 & 2 WT.05.01.01, EP 1 WT.01.01.01, EP 2 WT.05.01.01, EP 5 Problem log QC Log Book QC Log Book WT.01.01.01, EP 2 Has the manager or designee performed a weekly review and sign off of WT.04.01.01, EP 1 & 2 WT.05.01.01, EP 1 12 quality control and other test specific required documentation? Where appropriate, has proficiency testing been performed per WT.03.01.01, EP 1, 4, & POCT testing book 5 13 procedure, reported within deadlines? All CAP samples were run and results returned to POCT