A Comparative Study on Electrochemical Behaviour of Co O and Co -
advertisement

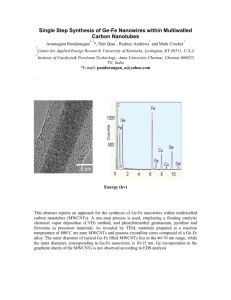
Journal of Scientific & Industrial Research Vol. 74, April 2015, pp. 202-208 A Comparative Study on Electrochemical Behaviour of Co3O4 and Co3O4MWCNTs for Supercapacitors K Chaitra1, V R Thomas1, M S Santosh1, C Srivastava2, N Nagaraju3 and H Kathyayini1* 1 Department of Nanobiosciences, Centre for Emerging Technologies, Jain Global Campus, Jain University, Jakkasandra post, Kanakapura Taluk, Ramanagara District, Bangalore Rural-562112, Karnataka, India 2 Department of Material Engineering, Indian Institute of Science, Bangalore-560012, Karnataka, India 3 Department of Chemistry, St. Joseph’s College P.G. Centre 46, Langford Road, Shanthinagar, Bangalore-560027, Karnataka India Received 18 October 2014; revised 29 January 2015; accepted 3 March 2015 Co3O4 and Co3O4/MWCNTs were prepared by hydrothermal process under autogenous pressure in Teflon lined autoclave and calcined at 2500C. Both samples were characterized by PXRD, FT-IR, SEM-EDS, TEM & FT-Raman to evaluate their surface and bulk properties. The PXRD pattern of the materials indicated the formation of cubic phase of Co 3O4. FT-IR results showed the presence of metal oxygen bond in the samples. The SEM and TEM images of the Co 3O4 / MWCNTs indicated spherical and cubic aggregates of metal oxide particles (10-30 nm) decorated both on the surface and inside the tubes of carbon nanotubes. The characteristic Ig and Id (graphitic and defects) Raman bands indicated the retention of tubular structure of MWCNTs even after the deposition of Co 3O4. The calcined Co3O4-MWCNTs composites and Co3O4 exhibited specific capacitance of 284 & 205 F/g at a sweep rate of 2mVs-1 in 6M KOH by cyclic voltammetry. The psuedocapacitance performances of calcined Co3O4-MWCNTs were found to be better than Co3O4. Chronopotentiometric studies made for the materials at a current density of 500mA/g indicated 100% columbic efficiency at 2000th cycle for Co3O4 / MWCNTs which is a better electrode material than Co3O4. Keywords: Multiwalled carbon nanotubes, cyclic voltammetry, chronopotentiometry, columbic efficiency. Introduction Supercapacitors are the chief energy storage devices which are in demand as there is energy crisis in the present world. As supercapacitors posses’ high power density and longer cyclability, these devices find application in electronics, hybrid electric vehicles etc. 1, 2 Supercapacitors are divided into two classes based on their energy storage mechanism. Electrochemical double layer capacitors (EDLCs) are the first type of supercapacitors using carbon materials such as activated carbon, graphene, carbon nanotubes, mesoporous carbon etc. The storage mechanism here is purely electrostatic and depends on the surface area3-5.The other is pseudocapacitance that includes metal oxides and conducting polymers, in which the capacitance arises due to the redox reactions and movement of π electrons6,7. Though the EDLC materials offer very high cycle life they do not offer high capacitance when compared to the pseudo capacitor materials and vice versa. Hence, a —————— Author for correspondence E-mail: nkathyayini45@gmail.com composite of both class of materials give an opportunity to utilize their advantages 8, 9. Among EDLC materials MWCNTs are interesting as they posses high accessible surface area, low electrochemical resistivity (ESI) and when compared to conventional carbon materials they allow easy diffusion of ions because of the open mesoporous network10. E. Frackowiak and K.Metenier have studied the electrochemical properties of MWCNTs and have reported a maximum capacitance of 135 F/g 11. Also, in a comparison study done by Seyed Hamed Aboutalebi and Alfred.T. Chidembo for Graphene oxide, MWCNTs and their composite, they found that MWCNTs exhibited a specific capacitance of 85 F/g only 12. Metal oxides are studied as electrode materials because of their variable oxidation states, proficient redox charge transfer and high pseudocapacitance. Though noble metal oxides such as RuO2, IrO2 exhibit good pseudocapacitance, due to their high cost and toxicity, now the focus is on the oxides of base transition metals such as Co, Ni, Cu, Fe etc, which are also known to exhibit good electrochemical properties13. Scientists have realized KATHYAYINI et al.: A COMPARATIVE STUDY ON ELECTROCHEMICAL BEHAVIOUR OF CO3O4 & MWCNTs the importance of Co3O4 as supercapacitor material as it is inexpensive and posse’s high redox activity. Shenglin Xiong and Changzhou Yuan have obtained a capacitance value of 92 F/g for mesoporous Co3O4 nanostructures14. Wang et al. have prepared mesoporous Co3O4 crater-like microspheres with a specific capacitance of 102F/g15. Hence Co3O4 and MWCNTs composite is the expected or predicted to be a better supercapacitor material. For instance, C. T. Hsieh and J.Y. Lin have reported a capacitance value of 185F/g (Co3O4-MWCNTs) which was found to be much higher than the oxidized MWCNTs (83.5 F/g) 16. Lian Gao and Yan Shan have prepared Co3O4MWCNTs composites by in situ decomposition of Co(NO3)2 and obtained specific capacitance of 200F/g and only 90 F/g for pristine MWCNTs17. Mazloumi et al. have studied the electrochemical properties of aligned carbon nanotubes-Co3O4 strips and have obtained a specific capacitance value of 123 F/g 18.Hence it is interesting to study the electrochemical behavior of composite of Co3O4 with MWCNTs. In the present work, we have synthesized Co3O4 nanoparticles and it’s composite with MWCNTs by hydrothermal method followed by calcination. Characterizations of the materials were carried out to understand their structural and physical properties. The electrochemical performance of these materials in 6M KOH was studied by cyclic voltammetry, chronopotentiometry and electrochemical impedance techniques. Experimental Materials All the reagents employed in this research work are of analytical grade. MWCNTs used in this work were purchased from Nanocyl Belgium. Cobalt nitrate hexahydrate, Sodium dodecyl sulphate (SDS), Ammonia solution, Sulphuric acid (H2SO4), Nitric acid (HNO3), Hydrogen peroxide (H2O2) were obtained from Merck Specialties Pvt. Ltd and used as received. Method Synthesis of Co3O4 and Co3O4/MWCNTs MWCNTs were functionalized using 1:3 HNO3:H2SO4 by sonicating at a frequency of 42 kHz and 250 watts power for 4 h to develop oxygen functionalities on its surface19. Further, 0.1g of functionalized MWCNTs was sonicated in distilled water for 2h. 0.15M solution of cobalt nitrate 203 hexahydrate was mixed with 10ml of SDS (0.01g) solution. The resulting solution was added to the dispersed MWCNTs suspension and further sonicated for 30 min, followed by slow addition of ammonia solution (2.5 wt %) with stirring. Subsequently 0.27ml of H2O2 was added and stirred for 5 min. The above solution was transferred to an autoclave maintained at 160oC for 5h. The reaction mixture was cooled, filtered and washed with distilled water; the resulting precipitate was dried at 100oC for overnight and was calcined at 2500 C for 3h to get the final product. For comparison of electrochemical properties, Co3O4 without MWCNTs was also prepared by following the same procedure. In this paper the two materials, Co3O4 and Co3O4/MWCNTs are denoted as Co-MO and Co-CNTs respectively. Characterization The morphology and the surface properties of CoMO and Co-CNTs were studied by the following techniques. P-XRD was recorded by using Panalytical Xpert pro X-ray diffractometer with Cu Kα radiation (λ = 0.154 nm) in 2θ range 5o to 70o at 40 kV and a scanning rate of 2omin-1, FT-IR spectra was obtained from Nicolet Model Impact 400D FT-IR spectrometer with 4 cm–1 resolution using KBr pellet technique, SEM-EDS for the samples was carried out for elemental composition by Quanta 200 FEI instrument. Transmission electron microscopy (TEM) images were recorded using a Tecnai 10 philips microscope. Raman spectra were recorded using Bruker instrument. Fabrication of Electrode and Electrochemical Measurements The working electrode was fabricated by mixing 85 wt% of the material with 10 wt% ketjen black EC 600JD in an agate mortar. To this mixture, a binder, 5 wt% polyvinylidene difluoride (PVDF) dissolved in solvent 1-methyl-2- pyrrolidinone (NMP) was mixed well and the slurry prepared was coated on a pretreated battery grade Ni foil of area 1cm2 and 0.2mm thickness. Further the working electrode was vacuum dried at 70oC for 8h. In this study the active mass of sample Co-MO and Co-CNTs coated on Ni foil was 1.3mg and 0.4mg respectively. Electrochemical studies such as cyclic voltammetry, chonopotentiometry and electrochemical impedance spectroscopy (EIS) were performed using CHI608E electrochemical workstation. The materials coated on Ni foil was used as working electrode, Pt wire as counter electrode, Ag/AgCl as reference electrode and 204 J SCI IND RES VOL 74 APRIL 2015 deoxygenated aqueous 6M KOH as the electrolyte. The stability of the working electrode materials were checked from galvanostatic charge-discharge cycles upto 2000cycles. AC impedance studies were made by imposing a sinusoidal alternating frequency from 0.01 Hz to 105 Hz, alternating current (ac) amplitude of 5mV at open circuit potential. Results and Discussion Characterization of the materials P-XRD is an important tool for the structural characterization of composite materials. The x-ray diffraction patterns for Co-MO and Co-CNTs are given in Figure 1. The diffraction peaks at 2Ө values 18.9o, 31.4o, 36.8o, 38.7o, 44.9o, 55.9o, 59.6o, 65.4o obtained in Co-MO corresponds to the different hkl planes (111) (220) (311) (222) (400) (422) (511) (440) of cubic phase of Co3O4 respectively (JCPDS Card No. 43-1003) 20.The main P-XRD diffraction peaks at 31.2o, 36.8o, 65.2o corresponding to (220) (311) (440) planes are the clear indication of cubic spinel structure of Co3O4 as described by Shin et al.21. The XRD pattern of functionalized MWCNTs contains prominent diffraction peaks at 2Ө value 25.8 o, 43 o and 53.5 o which corresponds to (002) (100) and (004) hkl planes of graphite22,23.Co-CNTs exhibited prominent diffraction peaks for Co3O4 and diffraction peaks of MWCNTs were not noticeable due to Co3O4 particles which are deposited on MWCNTs. Further from TEM and SEM analysis of Co-CNTs reveals the formation of Co3O4 on MWCNTs with the particle size ranging from 10-30nm. In FT-IR spectrum of Co-CNTs, a broad peak at 3426cm-1 is due to the stretching vibrations of OH– groups of H2O molecules24. The two distinct bands in Co-CNTs present at 673 cm-1 and 579 cm-1 is due to the stretching vibrations of metal-oxygen bond. The peak at 673 cm-1 is attributed to the M–O vibrations in which M is Co2+ and is tetrahedral coordinated, while the peak at 579 cm-1 is due to the stretching vibration mode of M–O in which M is Co3+ and is octahedral coordinated indicating the formation of Co3O4 oxide 25. (FT-IR spectrum given as supplementary data) SEM-EDS analysis of Co-CNTs was carried out to determine its surface morphology and elemental composition. The SEM image of Co-CNTs shows more or less uniform spherical Co3O4 nanoparticles deposited on the micro tubular structured MWCNTs. The elemental composition of the material indicates the presence of Co, O and C in Co-CNTs. Further insight on the morphology of the Co-CNTs is obtained from the TEM analysis. It clearly shows the presence of metal oxide nanoparticles on the surface and inside the walls of MWCNTs. It is also observed that the metal oxide nanoparticles are in the form of spherical as well as cubic aggregates with a diameter ranging from 10-30 nm. (SEM, TEM images and EDS profile given as supplementary data) Raman spectra of functionalized CNTs and Co-CNTs were studied. The three characteristic bands obtained for these materials at 1285cm-1, 1600 cm-1, 2565cm-1 are attributed to D- band, G- band and G’ band respectively. The G band in MWCNTs represent the in plane vibration of C-C bonds and the D band represents hydridized vibrational mode of graphene edges indicating the disorder of carbon atoms in MWCNTs 26. The disorder density on the MWCNTs sidewall is measured from the ratio of ID/IG 27. ID/IG ratio was found to be 1.0 and 1.34 in functionalized MWCNTs and Co-CNTs respectively. The values of ID/IG indicated that there is greater extent of disruption of the graphitic planes of MWCNTs in Co-CNTs. The raman spectra of Co-CNTs and Co-MO was also compared. The raman bands for Co-O bond in Co-MO at 479, 620 and 695 cm-1 28 was observed with more intensity while the same appeared in Co-CNTs with slightly lower intensity. Thus these characterization analyses clearly indicate the presence of Co3O4 in MWCNTs. Electrochemical studies Cyclic Voltammetry Fig.1—PXRD pattern of Co-MO and Co-CNTs Cyclic voltammetry (CV) measurements of Co-MO and Co-CNTs were studied at different sweep rates in KATHYAYINI et al.: A COMPARATIVE STUDY ON ELECTROCHEMICAL BEHAVIOUR OF CO3O4 & MWCNTs 205 the fixed potential range of 0 - 0.45V (Vs Ag/AgCl electrode) in 6M KOH solution. Initially the electrodes were stabilized at a fixed potential range for 25 CV cycles at a scan rate of 5mV s-1. The CV patterns obtained for the Co-MO and Co-CNTs exhibited mainly redox peaks indicating faradic behavior. The multiple anodic (oxidation) and cathodic (reduction) peaks in the CV patterns as shown in Figure 2a is attributed to different phases of cobalt oxide having different oxidation states. These peaks are due to the Co (II) Co (III) and Co (III) Co (IV) redox transitions 29,30. The following are two plausible reactions. Co3O4+ OH + H2O 3CoOOH + e3CoOOH + e- Co3O4 + OH + H2O The shape of CV curve becomes more asymmetric with increase in scan rate indicating the kinetic irreversibility of the electrode material. The graph of anodic current against square root of scan rate of the materials showed linear behavior indicating a diffusion controlled process at the electrode surface Figure 2b & 2c 31,32. The specific capacitance (Cs in F/g) values of both samples at different scan rates (ν, Vs-1) were calculated graphically by integrating the area under CV patterns using the equation as described by Ranga Rao et al 33. Co-MO and Co-CNTs exhibited specific capacitance of 205 and 284 F/g respectively at 2mVs-1. The higher specific capacitance of Co-CNTs is presumed due to the presence of MWCNTs, which allows the easy movement of OH- ions through the electrode material. It was observed that the specific capacitance values decreases from lower scan rate to higher, as OH- ions can only access the outer surface of the electrode where as at low scan rates the ions can percolate efficiently through the surface of electrode, in particularly the MWCNTs which can lead to higher specific capacitance as observed in Table 1. The increased specific capacitance of Co-CNTs at lower scan rate can be due to the presence of MWCNTs which allows easy percolation of OH- ions and provide larger surface for the redox reaction to occur. Chronopotentiometry The stability and cycle life of the electrode material is an important parameter for their application in supercapacitors which was studied by charge discharge cycles. The charge discharge cycles of these materials at different current densities 0.2, 0.5, 1, 2.5, 5 and 10 A/g at potential window of 0 – 0.36V in 6M Fig.2—(a) Cyclic volammetric curve of Co-CNTs at various sweep rate, (b) and (c) Square root of scan rate Vs anodic peak current of Co-MO and Co-CNTs respectively. 206 J SCI IND RES VOL 74 APRIL 2015 KOH solution was studied. The materials exhibited a nonlinear charge discharge curve which is primarily due to cobalt oxide reaction occurring at the electrode surface indicating psuedocapacitance behavior. The discharge curves obtained for Co-CNTs are shown in Figure 3. The specific capacitance values from chronopotentiometry are determined using the equation as reported earlier 34. The calculated specific capacitance values of Co-MO and Co-CNTs at different current densities are given in Table 2. It was observed that at higher current densities also CoCNTs exhibit higher specific capacitance. The specific capacitance decreased with increase in the applied current density which is attributed to large IR drop, polarization and insufficient utility of electro active material at very high current densities 35. The stability of electrode material was tested for 2000 charge discharge cycles and was found that Co-CNTs was stable for 2000 complete charge discharge cycles. But, Co-MO was stable for only 1000 cycles. The columbic efficiency (η) is calculated from each charge discharge cycles using the following equation. Table 1—Specific capacitance of Co-MO & Co-CNTs at various scan rates Sl. No. Scan rate (mV/s) Specific capacitance in F/g from CV measurements Co-MO Co-CNTs 1 2 205 284 2 5 197 243 3 10 195 227 4 20 189 206 5 40 182 195 6 50 178 191 7 80 171 185 8 100 163 181 Fig.3—Charge discharge curves obtained for Co-CNTs at different applied current density η = tD / tC x100 Where tD and tC represents discharge and charge time respectively. It was seen that in Co-MO columbic efficiency was 86% initially which decreased to 80% after 1000 cycles, while Co-CNTs exhibited 100% columbic efficiency even at 2000th cycle indicating the improved efficiency of Co3O4 due to the presence of MWCNTs in the Co-CNTs. This may be attributable to the backbone structure of MWCNTs for the metal oxide and helps in maintaining its structural stability. Hence Co-CNTs was found to be an excellent material in a life cycle test, when compared to Co-Mo. Electrochemical Impedance spectroscopy AC impedance studies were carried out to study the frequency dependent specific capacitance behavior. The impedance study of the two compounds was carried out at their open circuit potentials in the frequency range of 0.01Hz to 10e+5 Hz. The specific capacitance of the materials was calculated from the experimental data36.The obtained specific capacitance of Co-MO & Co-CNTs was 160F/g and 240 F/g respectively Figure 4. The Co-CNTs showed better Table 2—Specific capacitance of Co-MO & Co-CNTs at various current densities Sl. No Current density (A/g) 1. 2. 3. 4. 5. 0.2 0.5 1 2.5 5 Specific capacitance in F/g from CP measurements Co-MO Co-CNTs 86 129 88 114 83 104 81 93 72 86 Fig.4—Plot of Specific capacitance obtained from impedance data for Co-MO and Co-CNTs KATHYAYINI et al.: A COMPARATIVE STUDY ON ELECTROCHEMICAL BEHAVIOUR OF CO3O4 & MWCNTs specific capacitance performance by all the above experiments which could be due to improved mobility of ions at lower frequency through the tubes of the MWCNTs. Due to presence of MWCNTs in Co-CNTs which provide high surface area for redox reactions to occur at the electrode surface and they offer low resistance, indicating Co-CNTs a better material for supercapacitor applications. Conclusions In this work, a comparative electrochemical performance of Co-MO and Co-CNTs prepared by simple hydrothermal method was studied. Characterization studies and results indicated formation of cubic or spherical Co3O4 with particle size ranging from 10-30 nm on MWCNTs. The electrochemical studies showed Co-CNTs exhibiting 100% columbic efficiency even at 2000th cycle and also showed high specific capacitance in comparison to Co-MO. This improved performance of Co-CNTs is owing to the presence of MWCNTs. Hence there is a significant role of MWCNTs in Co-CNTs in enhancing the columbic efficiency. Acknowledgement The authors thank NRB- Naval Research Board for the financial support given for this research work. Project Number: NRB-290/MAT/12-13. Authors also thank Dr. Krishna Venkatesh, CEO, Centre for Emerging Technologies and Dr. Chenraj Roychand, President Jain University trust for their constant support in encouraging this research work. 8 9 10 11 12 13 14 15 16 17 18 19 Reference 1 2 3 4 5 6 7 Jayalakshmi M & Balasubramanian K, Simple capacitors to supercapacitors, Int J Electrochem Sci, 3 (2008) 1196-1217. Wang G, Zhang L & Zhang J, A review of electrode materials for electrochemical supercapacitors, Chem Soc Rev, 41 (2012) 797-828. Davies A & Yu A, Material advancements in supercapacitors: From activated carbon to carbon nanotube and graphene, Can J Chem Eng, 89 (2011) 1342-1357. Zhang L L & Zhao X S, Carbon-based materials as supercapacitor electrodes, Chem Soc Rev, 38 (2009) 2520-2531. Pandolfo A G & Hollenkamp A F, Carbon properties and their role in supercapacitors, J Power Sources, 157 (2006) 11-27. Yang Q, Lu Z, Liu J, Lei X, Chang Z, Luo L & Sun X, Metal oxide and hydroxide nanoarrays: Hydrothermal synthesis and applications as supercapacitors and nanocatalysts , Prog Nat Sci: Mater Int, 23(4) (2013) 351-366. Simon P & Gogotsi Y, Materials for electrochemical capacitors, Nat Mater, 7 (2008) 845-854. 20 21 22 23 24 25 207 Zhi M, Xiang C, Li J, Li M & Wu N, Nanostructured carbon–metal oxide composite electrodes for supercapacitors: a review, Nanoscale, 5 (2013) 72-88. Fisher R A, Watt M R & Ready W J, Functionalized Carbon Nanotube Supercapacitor Electrodes: A Review on Pseudocapacitive Materials, ECS J Solid State Sci Technol, 2(10) (2013) M3170-M3177. Pan H, Li J & Feng Y P, Carbon Nanotubes for Supercapacitor, Nanoscale Res Lett, 5 (2010) 654-668. Frackowiak E, Metenier K, Bertagna V & Beguin F, Supercapacitor electrodes from multiwalled carbon nanotubes , Appl Phys Lett, 77 (2000) 2421. Aboutalebi S H, Chidembo A T, Salari M, Konstantinov K, Wexler D, Lin H K & Dou S X, Comparison of GO, GO/MWCNTs composite and MWCNTs as potential electrode materials for supercapacitors, Energy Environ Sci, 4 (2011) 1855-1865. Lokhande C D, Dubal D P & Oh-Shim J, Metal oxide based thin film supercapacitors, Curr Appl Phys, 11 (2011) 255-270. Xiong S, Yuan C, Zhang X, Xi B & Qian Y, Controllable synthesis of mesoporous Co3O4 nanostructures with tunable morphology for application in supercapacitors, Chem Eur J, 15 (2009) 5320–5326. Wang L, Liu X, Wang X., Yang X & Lu L, Preparation and electrochemical properties of mesoporous Co3O4 crater-like microspheres as supercapacitor electrode materials, Curr Appl Phys, 10 (2010) 1422-1426. Hsieh C T & Lin J Y, Electrochemical Capacitance from Carbon Nanotubes Decorated with Cobalt Oxide Nanoparticles in Alkali Electrolyte, ECS Trans, 13 (2008) 73-89. Shan Y & Gao L, Formation and characterization of multiwalled carbon nanotubes/Co3O4 nanocomposites for supercapacitors, Mater Chem Phys, 103 (2007) 206–210. Mazloumi M, Shadmehr S, Rangom Y, Nazar L F & Tang X S, Fabrication of three-dimensional carbon nanotube and metal oxide hybrid mesoporous architectures, ACS Nano, 7(5) (2013) 4281-4288. Fiang X, Gu F, Bai X, Lin L & Zhang Y, The influence of acid treatment on multi-walled carbon nanotubes, Pigm Resin Technol, 38 (2009) 165-173. Song X C, Wang X, Zheng Y F, Ma R & Yin H Y, Synthesis and electrocatalytic activities of Co3O4 nanocubes, J Nanopart Res, 13 (2011) 1319–1324. Shin C, Manuel J, Dul-Sun K, Ho-Suk R, Hyo-Jun A & JouHyeon A, Structural characterization and electrochemical properties of Co3O4 anode materials synthesized by a hydrothermal method, Nanoscale Res Lett, 7 (2012)73-79. Belin T & Epron F, Characterization methods of Carbon nanotubes: a review, Mater Sci Eng B, 119 (2005) 105–118. Reddy K R, Jeong H M, Lee Y & Raghu A V, Synthesis of MWCNTs-Core/Thiophene Polymer-Sheath Composite Nanocables by a Cationic Surfactant-Assisted Chemical Oxidative Polymerization and Their Structural Properties, J Polym Sci Part A: Polym Chem, 48 (2010) 1477–1484. Yudianti R, Onggo H, Sudirman, Saito Y, Iwata T & Jun-ichi A, Analysis of Functional Group Sited on Multi-Wall Carbon Nanotube Surface, The Open Mater Sci J, 5 (2011) 242-247. Chen-Bin W, Chih-Wei T, Shiue-Jiun G & Shu-Hua C, Effect of the surface area of cobaltic oxide on carbon monoxide oxidation, Catal Lett, 101 (2005) 59-63. 208 J SCI IND RES VOL 74 APRIL 2015 26 Bokobza L & Zhang J, Raman spectroscopic characterization of multiwall carbon nanotubes and of composites, eXPRESS Polym Lett, 6 (2012) 601-608. 27 Dresselhausa M S, Dresselhausb G, Saitoc R & Joriod A, Raman spectroscopy of carbon nanotubes, Phys Rep, 409 (2005) 47-99. 28 Hadjievl V G, Ilievl M N & Vergilov I V, The Raman spectra of Co3O4, J Phys C Solid State Phys, 21 (1988) L199-L201. 29 Palmas S, Ferrara F, Vacca A, Mascia M & Polcaro A M, Behavior of cobalt oxide electrodes during oxidative processes in alkaline medium, Electrochim Acta, 53 (2007) 400–406. 30 Casella I G, Electrodeposition of cobalt oxide films from carbonate solutions containing Co(II)–tartrate complexes, J Electroanal Chem, 520 (2002) 119–125. 31 Sataraddi S R & Nandibewoor S T, Voltammetric-oxidation and Determination of 5-Flurouracil and its Analysis in Pharmaceuticals and Biological Fluids at Glassy Carbon 32 33 34 35 36 Electrode Mediated by surfactant cetyltrimethyl ammonium bromide, Der Pharma Chem, 3 (4) (2011) 253-265. Öztürk F, Taşdemir I H, Erdoğan D A, Erk N & Kılıç E, A new voltammetric method for determination of lercanidipine in biological samples, Acta Chim Slov, 58 (2011) 830–839. Sumanta K M, Justin P & Ranga Rao G, Nanoscale morphology dependent pseudocapacitance of NiO: Influence of intercalating anions during synthesis, Nanoscale, 3(2) (2011) 683-92. Fan Z, Chen J, Cui K, Sun F, Xu Y & Kuang Y, Preparation and capacitive properties of cobalt–nickel oxides/carbon nanotube composites, Electrochim Acta, 52 (2007) 2959–2965. Justin P, Sumanta K M & Ranga Rao G, Tuning of Capacitance Behavior of NiO Using Anionic, Cationic, and Nonionic Surfactants by Hydrothermal Synthesis, J Phys Chem C, 114 (2010) 5203–5210. Sumanta K M, Justin P & Ranga Rao G, Pine-cone morphology and pseudocapacitive behavior of nanoporous nickel oxide, Electrochim Acta, 55 (2010) 8388–8396.