Document 13734925
advertisement

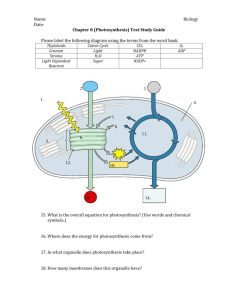
MIT Department of Biology 7.014 Introductory Biology, Spring 2005 7.014 7.014 Lecture 17: 17: Carbon and Energy Metabolism Metabolism March 14, 2005 Summary of the options for Life (the simplified view – see also Freeman Ch 25) Organism Carbon Source Energy Source Autotrophs Oxygenic Photosynthesis pro and euk CO2 sun Anoxygenic Photosynthesis prokaryotic CO2 sun Chemosynthesis prokaryotic CO2 Reduced chemical compounds Aerobic Respiration pro or euk organic C organic C Anaerobic Respiration pro or euk organic C organic C Fermentation pro or euk organic C organic C Heterotrophs euk = eukaryotic 1 PHYCOER YTHRIN YLL b CHLOROPH B- CAROTENE YLL a 400 WAVELENGTH OF LIGHT (nm) CHLOROPH ABSORPTION YANIN PHYCOC UV 700 Figure by MIT OCW. Figure removed due to copyright considerations. Please see: Freeman, Scott. Biological Science. Upper Saddle River, NJ: Prentice Hall, 2002. ISBN: 0130819239. Fig 25-6. 2 “Dark reactions” (common to all) NADPH+ NADP+ CO2 glucose ATP ADP + Pi Photosynthesis Light O2 B e- + NADP H2O A (Photo)Autotrophs NADPH H + Plant Photosynthesis ⎯→ ATP ADP + Pi ⎯ “Dark reactions” (common to all) NADPH+ NADP+ CO2 glucose ATP ADP + Pi Photosynthesis Light S, SO4 B - e + NADP H2S A (Photo)Autotrophs NADPH H + Bacterial Photosynthesis (anaerobic) ⎯→ ATP ADP + Pi ⎯ 3 “Dark reactions” (common to all) NADPH+ NADP+ CO2 glucose ATP ADP + Pi Photosynthesis Light B (Photo)Autotrophs - e + NADP A NADPH H ATP and NADPH produced in light reaction used on dark reaction + ⎯→ ATP ADP + Pi ⎯ “Dark reactions” (common to all) Photosynthesis Type A B oxygenic (plant) H2O ⇒ O2 anyoxygenic H2S ⇒ S, SO42− NADPH+ NADP+ CO2 glucose ATP ADP + Pi Net Reaction Light B (Photo)Autotrophs - e + NADP A NET: Light energy used to convert CO2 to glucose NADPH H + ⎯→ ATP ADP + Pi ⎯ Type A B oxygenic (plant) H2O ⇒ O2 anyoxygenic H2S ⇒ S, SO42− 4 (Chemo) Autotrophs “Dark reactions” NADPH+ NADP+ CO2 glucose ATP ADP + Pi (common to all) Chemosynthesis e− D E F C (releases energy) H+ gradient ADP + Pi (requires energy) e− (requires energy) ATP NADP+ D C Type C sulfur-oxidizing H2S H2S ⇒ ⇒ nitrifying NH3 NO2 ⇒ NADPH D E S, SO42− S, SO42− O2 NO3− ⇒ ⇒ H2O N2O O2 ⇒ H2O NO3 − F (Chemo) Autotrophs“Dark reactions” NADPH+ NADP+ CO2 glucose ATP ADP + Pi (common to all) Chemosynthesis e− D E F C (releases energy) H+ gradient ADP + Pi (requires energy) e− (requires energy) ATP Type C sulfur-oxidizing H2S H2S ⇒ ⇒ nitrifying NH3 NO2 ⇒ D NADP+ C NADPH D E S, SO42− S, SO42− O2 NO3− ⇒ ⇒ H2O N2O O2 ⇒ H2O NO3 − F Chemical energy used to convert CO2 to glucose 5 Figure removed due to copyright considerations. Please see: Freeman, Scott. Biological Science. Upper Saddle River, NJ: Prentice Hall, 2002. ISBN: 0130819239. Fig 25-5. glucose Respiration + NAD ATP NADH pyruvate + NAD Krebs’ cycle Heterotrophs NADH CO2 + NAD − e NADH A O2 B H2O Aerobic Respiration + H gradient ⎯→ ATP ADP + Pi ⎯ 6 glucose Respiration Fermentation + NAD ATP Lactate (3C) or Ethanol (2C) NADH pyruvate + NAD Krebs’ cycle Heterotrophs NADH Fermentation CO2 + NAD − e A NADH B + H gradient ⎯→ ATP ADP + Pi ⎯ glucose Respiration + NAD ATP NADH pyruvate + NAD Krebs’ cycle NADH Heterotrophs Anaerobic Respiration CO2 + NAD − e NADH A NO3 -- B NO, N2, N2O,NH3 + H gradient ⎯→ ATP ADP + Pi ⎯ 7 glucose Respiration + NAD ATP NADH pyruvate + NAD Krebs’ cycle Heterotrophs Anaerobic Respiration NADH CO2 + NAD − e A NADH These are examples: Many things can serve as electron acceptors B + H gradient ⎯→ ATP ADP + Pi ⎯ Type A aerobic O2 B 2− sulfate-reducing SO4 denitrifying NO3− ⇒ H2O ⇒ H2S or S ⇒ NO, N2, N2O, NH3 Figure removed due to copyright considerations. Please see: Freeman, Scott. Biological Science. Upper Saddle River, NJ: Prentice Hall, 2002. ISBN: 0130819239. Fig 25-5. 8 Glucose Oxidized Compounds SO4 – NO 3– H 2S SO4 Bacterial photosynthesis O2 NH4 + H 2S Anaerobic respiration H 2O Chemosynthesis “CH2O” Aerobic respiration Photosynthesis Reduced Compounds CO2 Oxic Conditions Anoxic Conditions Take Home Messages ¾ There is more than one way to be alive – energy and carbon and electrons ¾ Microbes have most of the metabolic diversity available ¾ Products of one organism are the substrate for another ¾ Where metabolic pathway is energetically favorable, a microbe has evolved to take advantage of it 9