Characterization and DC Conductivityof novel NiO doped PolyvinylAlcohol(PVA) Nano-composite films
advertisement

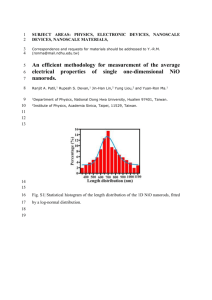
International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 Characterization and DC Conductivityof novel NiO doped PolyvinylAlcohol(PVA) Nano-composite films Chivukula Srikanth1,Chakradhar Sridhar B2,B.M.Nagabhushana3and R.D.Mathad1 1 Department of Physics, Gulbarga University, Gulbarga-585 106, Karnataka, India. 2 Department of Physics, Gulbarga University, Gulbarga-585 106, Karnataka, India 3 Department of Chemistry, M.S. Ramaiah Institute of Technology, Bangalore 560 054, India ABSTRACT DC conductivity of PVA-NiO nanocomposite films have been studied in the present work. The composites were prepared by solution-casting technique. The prepared PVA-NiO composites have been characterized by X-ray diffraction (XRD) analysis, FourierInfrared Spectroscopy (FTIR)and Scanning Electron Microscopy (SEM), which confirmed the presence of NiO in polyvinyl alcohol and the formation of the composite.DC conductivity studies show thermally activated behavior of all the composites. The conductivity was found to increase with the increase in temperature indicating the semiconducting behavior of all the compositions. The activation energy increases as the content of NiO nanoparticles increases from 1 to 4 in wt% in the PVA-NiO nanocomposites.Maximum conductivity was observed in 4 wt% of NiO in polyvinyl alcohol. KEYWORDS:-Polyvinyl alcohol, Nano Nickel oxide,X-ray diffraction, DC conductivity. 1. INTRODUCTION Polyvinyl alcohol (PVA) is semi-crystalline, water soluble, with low electrical conductivity polymer [1]. It has a carbon chain backbone with hydroxyl groups attachedto methane carbons; (Fig.1) these AOH groups can be a source of hydrogen bonding and hence assists the formation of polymer composite [2].PVA is known to be one of the most widely used polymers for the separation of water–organic mixtures.It has certain physical properties resulting from crystal– amorphous interfacial effects[3, 4].It is known to be oneof the most widely used polymers for the separation of water– organic mixtures. It is a potential material having a good charge storage capacity and dopant-dependent electrical properties.Nickel metal oxide (NiO) is a p-type semiconductor that crystallizes in a rock salt structure and has a wide energy band gap in the range from 3.6 to 4.0eV[5].It has a wide range of applications due to its good chemical stability as well as for its excellent optical and electrical properties [6].Thesehave been used as catalysts [7],electro chromic display devices [8], fuel cells [9] and gas sensors [10]. Nano-composites consisting of PVAwith NiOhavebeeninvestigatedbecause,theirproperties are quite different from PVA and NiO owing to their interfacial interactions. These composites are expected tofind commercial applications like gas sensors, electro chromic devices and supercapacitors. In the present work, attempts have been made to synthesize the nano-nickel oxide particles by low solution heat combustion methodandPVA-NiO nano-composite by solution-casting method. These polymer nanocomposites have been characterized by using various techniques such as X-ray diffraction (XRD), Fourier Infrared Spectroscopy (FTIR) and Scanning Electron Microscopy (SEM). The dc conductivity measurements are done by using two probe set-up. Fig.1. Chemical structure of PVA. Volume 3, Issue 9, September 2014 Page 200 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 2. EXPERIMENTAL 2.1. Materials Polyvinyl alcohol of analytical gradewas obtained in the powder form from Nice Chemicals Pvt.Ltd. Cochin,with the average molecular weightof 125,000 (98% hydrolyzed). Nickel Nitrate of AR grade was purchased from S.D. FineChem. Ltd.Mumbai. 2.2. Synthesisof Nano Nickel Oxide (NiO) Low Solution combustion (LSC) technique was employedforthe preparation ofnano- nickel oxide [11]. Hydrated Nickel Nitrate [Ni (NO3)26H2O] is used for the synthesis of nickel oxide. The filtered lemon juice was used as afuel [11].Thestiochiometric ratio ofNickel nitrate and fuelwere taken into thecylindrical Petri dish of ~300 ml and well dissolved inde-ionized water. The aqueous redox mixture was dispersed uniformly by stirring the mixture using a magnetic stirrerfor5 min. Thentheresulting mixture was placedinto a pre-heated muffle furnace maintained at (400 ± 10) °C. In the beginning, the solution was thermally dehydrated and ignited with the liberation of large amount of gases (N2, O2, etc.). The entire process took place within 8 min. The resultant powder occupied the entire volume of the reaction vessel. The combustion was self-propagating and within a few minutes the reaction was completed and fluffy powder was obtained. After completion of the process, the product obtained was grinded well using mortar and pestle. The powder is voluminous, weakly agglomerated, foamy, nano in size and has a high surface area. The formation of product through LSC process can be represented by the following reaction. Ni (NO3)26H2O+Feul (aq.) Combustion product(S) + Heat + Gases LSC process can be considered as a thermally induced redox reaction of the precursor wherein a fuel acts as reductantand NO3 ions act as oxidant. 2.2.1. Synthesis of Polyvinyl alcoholfilm. The pure PVA films were prepared by solution casting technique [12].PowderedPVAofabout 2.5 gwasdissolvedin50ml of double distilled water by stirring. The solution was then warmed up to 333 K and thoroughly stirred, using a magnetic stirrer, for about 1h until the polymer became completely soluble. By removing the trapped air bubbles, the solution was poured onto a leveled clean glass plate andleft to dry at room temperature for about 48 h.The dried films were peeled offfrom the glass plate and cut into suitable pieces for measurements [13]. 2.2.2. Synthesis of PVA–NiO nano-composite films. PVA-NiO nanocomposite films were also prepared again bythe solution casting method. First, the PVA stock solution was prepared by the above method and to this solution 1.0 wt%,2.0wt%, 3.0 wt% and 4.0 wt% of NiO nano powder was mixed and stirred vigorously by using magnetic stirrer until transparent PVA–NiO multi component dispersion was obtained. Further, ultrasonication was done for complete dispersal of NiO in PVA solution. Then known volume of viscous PVA–NiO solution was poured onto a leveled clean glass plate and left to dry at room temperature for about 48 h. The dried films were peeled off from the glass plate and cut into suitable pieces for characterization. 2.3. Measurements. The X-ray diffraction (XRD) patterns of the pure PVA film, nano NiO powder and the polymer nanocomposite films were recorded at room temperature using an X-ray powder diffractometer with Cu Ka radiation (λ = 1.5416 A˚) in the 2ө (Bragg angles) range (10˚ 2ө 80˚) to elicit the information related to phase purity, crystallinity and details of their structure. The FTIR spectraofthesamples were recordedon a Perkin Elmer 1600 spectrophotometerinKBrmedium. The morphology of the compositesin the form of powder was investigated using scanning electron microscope (SEM) Model-EVO-18 Special Edison, Zeus, Germany. For DC electrical characterization of the films, electricalcontacts were painted withElectrodag 502 from Ted Pella. Temperature dependent electrical conductivity was measured from 35°c to 125°c using Keithley 6514 electrometer. 3. RESULTS AND DISCUSSION 3.1. X–Ray diffraction. Fig.2 (a) showsX-ray diffractionpatternofpure PVA. The XRD pattern of pure PVA shows a characteristic peak for an orthorhombic latticecentered at20°indicatingits semi crystalline nature [14, 15]. The peaks have resulted from the part of crystallinity in PVA polymer molecules. This crystallinity is the result of strong intermolecular and intramolecular hydrogen bonding between the PVA molecular chain [16, 17].A broad peak was noticed at2ө~20° that can be assigned to the Vander Wall distance of 5A°.This peak may be attributed to the intermolecular distances between neighbouring chains. A small hump was observed at 2ө~42°, corresponding to an approximately hexagonal orderingofthemolecular chains [18].The XRD patternsof NiO nanoparticles are shown in Fig.2 (b). NiO was synthesized by Heat Combustion method. These nanoparticles have shown good crystallinity because of the existence of sharp peaks in the XRD pattern. The crystallite size of the synthesized NiO nanoparticles was calculated using Scherer’s formula given by Volume 3, Issue 9, September 2014 Page 201 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 D 0.9 cos ISSN 2319 - 4847 (1) WhereD is the average crystallite size, is the X-ray wavelength (1.5416 A ) and is full width at half maximum in radian. The average crystallite size is found to be ~19 nm. In the XRD pattern, different lines are attributed to the (111), (200), (220), (311), (222), (400), (331) and (420) planes are in good agreement with the data of NiO powder file (JCPDS number 47-1049) which corresponds to the cubic crystalline structure with lattice constants a=b=c=4.1771 A [19].The XRD spectrum exhibits an intense (200) peak indicating preferential orientation in the<200>direction. The NiO lattice constant calculated from the XRD data is 4.1792 A , which is in good agreement with the reported data. Fig.2(c) shows the X-ray diffraction pattern of PVA,PVA–NiO composite(1%wt. -4% wt.) and nano NiO. In the XRD pattern, different lines are attributed to the (111), (200), (220), (311), (222) and (400) planes; by comparing the XRD pattern ofthe composite and NiO, it is confirmed that NiO has retained its structure even though it is dispersed in PVAduringpolymerizationreaction. (101) Intensity(arb.units.) PURE PVA 0 10 20 30 40 50 60 70 80 90 2 (degree) Fig.2 (a) XRD of Pure PVA N A N O N iO (2 0 0 ) Intensity(arb.units) (1 1 1 ) (311) 0 10 20 30 40 50 60 70 (222) (2 2 0 ) 80 2 (d e g re e ) Fig.2 (b) XRD of Nano NiO Volume 3, Issue 9, September 2014 Page 202 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 101 (1 1 1 ) (2 0 0 ) (F) (220) 101 (1 1 1 ) (E ) (200) (2 2 0 ) Intensity (arb. units) 1 0 1 (D ) (1 1 1 ) (2 0 0 ) (220) 101 (1 1 1 ) ( 2 0 0 ) (C ) (2 2 0 ) 101 (2 2 0 ) (A ) 0 10 20 30 40 (311) (2 0 0 ) (1 1 1 ) 50 60 70 (222) (B ) 80 2 (d e g re e ) Fig.2(c). XRD of (A) pure NiO (B) pure PVA (C) PVA-1%NiO (D)PVA-2%NiO (E) PVA-3%NiO (F) PVA-4%NiO 3.2. FTIR Spectra 2353 3575 PURE PVA 1724 % Transmittance 2850 1142 2934 14201326 915 3258 836 1086 3500 3000 2500 2000 1500 1000 682 500 Wave Number(cm-1) Fig.3 (a). FTIR of pure PVA In Fig.3 (a), FTIR spectrum of pure PVA sample is showed. It clearly reveals the major peaks associated with polyvinyl alcohol.Itis observed that C–H broad alkyl stretching band ( =2850-3000cm-1), typical strong hydroxyl bands for free alcohol nonbonded –OH stretching bandat( =3600-3650cm-1) and hydrogen bonded band at =3200-3570cm-1 [20-22]. Intramolecular and intermolecular hydrogen bondings are expected to occur among PVA chains due to high hydrophilic forces. An important absorption peak was verified at =1142cm-1. This band has been used as an assessment tool of poly (vinyl alcohol) structure because it is a semi crystalline synthetic polymer able to form some domains depending on several process parameters [20-22]. Volume 3, Issue 9, September 2014 Page 203 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 N iO 2929 1448 344 0 3902 2385 638 1025 % Transmittance 1635 590 1380 414 4000 3600 3200 2800 2400 2000 1600 1200 800 400 W a v e N u m b e r(c m -1 ) Fig.3 (b)FTIR of nano NiO. Fig.3 (b) shows the FTIR spectraof NiO nanoparticles, whichrevealed several significant absorption peaks. The broad absorption band in the region of 600-700cm-1 is assigned to Ni-O stretching vibration mode, the broadness of the absorption band indicates that the NiO powders are nanocrystals.The broad absorption band centered at 3440cm-1 is assigned to the band O-H stretching vibrations and the weak band near 1635cm-1 is assigned to H-O-H bending mode [23].A strong peak is observed to be centered at638cm-1 corresponds to the stretching vibration of Ni–O.The peaks at 1380cm-1 is due to C-H in plane bending modes and the peaks at 414cm-1 to 590cm-1correspond to metallic stretch, which can be assigned to nickel oxide groups. Fig.3(c) shows the FTIR spectrum of PVA–NiO (1%–4%) nanocomposites.It is interesting to note thatthemainabsorption bands areaffected bythe presence ofNiO in pure PVA. This change is due to loss in conjugation and molecular orderafter modificationofPVA with NiO. Since the frequency of a vibration is directly proportional to force constant of the bonds which indicates the shortening of N–C bonds suggesting localization of p-orbital electrons on N [24]. However the shift of NiO stretching band at 688cm-1 was observed in PVA–NiO hybrid nanocomposite films. The observed shiftin stretching vibration ofNi–Oisdue to binding of organic compound tothe NiO [25]. The result indicates strong interaction between PVA and NiO nanoparticles. (e ) % Transmittance (d ) (c) (b ) (a ) 3500 3000 2500 2000 1500 1000 500 W a v e N u m b e r(c m -1 ) Fig.3(c).FTIR Spectra (a) pva (b) pva-1%NiO (c) pva-2%NiO (d) pva-3%NiO (e) pva-4%NiO 3.3 Scanning electron microscopyand EDX analysis The scanning electron micrographs of PVA film, NiO nano powder andNiO doped PVA films are shown in Fig. 4(a-d). SEM of pure PVA shows uniformlyprocessed smooth PVA matrix and the semi crystalline nature of PVA supporting the observations of XRD analysis (Fig.4 (a)). The average grain size was calculated by using the linear intercept formula and it is found to be 114nm to 234nm. Well defined nanocrystalline morphology of NiO is shown in Fig.4(b)[26].The particles are irregular in shape having an average grain size from 30nm to 75nm. The grains are found to be well interconnected with each other which indicate that they have enough binding energy to combine with neighbour grains or molecules. Fig.4(c) and 4(d) show the SEM image of PVA-NiO nano-composites, whichindicate overgrown clusters. The crystallinity oftheNiOis seen to decrease with the addition of PVAin it. The average grain size is found to be 72 nm to 140 nm. Volume 3, Issue 9, September 2014 Page 204 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 b a d C Fig.4. SEM imagesof(a) Pure PVA (b) Pure NiO(c) PVA-1%NiO (d) PVA-4%NiO a b c c Fig.5. EDX of (a) NiO (b) PVA--1 %NiO (c) PVA—4%NiO Therepresentative EDX spectra of nano NiO, PVA–1% NiO and PVA–4% NiO composite films are shown in Fig.5.The spectra shows enhancementin Nickel and oxygen content when concentration ofthe dopantincreases. 3.4 DC Conductivity Measurements Fig.6 (a) shows a typical logσ vs.1000/T plot for PVA and PVA-NiO (1%--4%Wt.) nano-composite films. Theconductivityincreasedwithincreasein temperature but the rate ofincrease was different in different temperature regions. From the slopes of these plots, the activation energies were calculated using the Arrhenius relation [27], Volume 3, Issue 9, September 2014 Page 205 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 E exp kBT ISSN 2319 - 4847 (2) whereσis the conductivity at temperature T, σo is a constant, E is the activation energy and kB is the Boltzmann constant. It seems that the dominant charge transport is through the motion of electrons both at low and high temperatures becausethe magnitudes of activation energies in both regions are within the limits suggested by Jonscher [28]for electronic conduction ( 0.8eV).The activation energy increases with the increasein doping concentration ofNiOin PVA matrixboth at high temperature andlowtemperature regions (Table 1). At 308Kand 398K, thedc electrical 13 13 conductivity of purePVAwasfoundto be 5.5110 Scm 1 and 17.90 10 Scm 1 respectively. As content ofNiOincreases 13 from 1% to 4%in PVA matrix, the conductivity of PVA–NiOnano-composite increases from 5.83 10 Scm 1 to 13.13 10 13 Scm 1 at 308K, similarly at 398Kit increases from 43.35 10 13 Scm1 to 377.03 10 13 Scm1 (Table 1).The increase in conductivitywithincrease of temperatureis the characteristic behavior of semiconductors. Thetemperature dependence of the conductivity σ(T) of disorderedsemiconductingmaterialsisgenerallydescribedbytheMottVariable Range Hopping (VRH) model [29], 1 TO 4 (T ) exp T (3) where σ is the conductivity, T is the temperature and σ0 is the conductivity at characteristic temperature T0. PVA– NiO nanocomposites followthethree-dimensional VRH electrical conduction process [30].Fig. 6(b) and 6(c) shows theplotofconductivity vs. the content ofNiOnanoparticlesinthe PVA–NiO nanocomposites both at 308K and 398K. Theplot shows that the conductivity of PVA increases remarkably after insertion of NiO nanoparticles (1–4%)in the PVAmatrix.PVA, NiO and PVA–NiO nanocomposites are of P-type semiconducting materials having large number of free charge carriers available for the purpose of conduction. Hence, as the concentration of NiO increases the number of free charge carriers also increases, all together electrons from polar O2- terminated NiO nanoparticles surfaces and from the PVA chains, resulting inthe improvement in the conductivity of PVA–NiO nanocomposites [31]. - 1 0 .4 (a ) - 1 0 .6 P P P P P - 1 0 .8 log (S cm-1) - 1 1 .0 V V V V V A A A A A -1 % -2 % -3 % -4 % N N N N iO iO iO iO - 1 1 .2 - 1 1 .4 - 1 1 .6 - 1 1 .8 - 1 2 .0 - 1 2 .2 - 1 2 .4 2 .5 2 .6 2 .7 2 .8 2 .9 3 .0 3 .1 3 .2 3 .3 1 0 0 0 /T ( k - 1 ) 14 4 00 10 3 00 (c ) -1 3 50 Conductivity (10¯ )Scm 8 2 50 13 13 Conductivity ( (10¯ )Scm -1 (b ) 12 6 4 2 2 00 1 50 1 00 50 0 0 1 2 3 PV A -N iO (w t% ) 4 0 1 2 3 4 P V A - N iO (w t% ) (b) Plot of conductivity vs. PVA-NiO (wt. %) (at 308K), (c) Plot of conductivity vs. PVA-NiO (wt. %) (at 398K) Fig.6. (a) Plot of logσ vs. 1000/TPVA, PVA-NiO (1-4%) composites. Volume 3, Issue 9, September 2014 Page 206 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 . S.NO. 1. 2. 3. 4. 5. ISSN 2319 - 4847 Table 1.Activation energy and Conductivity of PVA-NiO Nano-composite films. 13 1 ACTIVATION ENERGY E(eV) Conductivity ( ) 10 Scm COMPOSITIO N High Low (At 308K) (At 398K) temperature temperature range. range. PVA 0.117 0.173 5.51 17.90 PVA-1%NiO 0.322 0.266 5.83 43.35 PVA-2%NiO 0.538 0.347 8.02 262.34 PVA-3%NiO 0.552 0.358 9.30 266.91 PVA-4%NiO 0.568 0.373 13.13 377.03 4. CONCLUSIONS Polyvinyl alcohol (PVA)-Nano Nickel oxide (NiO) composites of various weight percentages were synthesized by solution casting method. X-ray diffraction studies show the retention ofNiO in polymer matrix. Studies on Fourier Infrared Spectroscopy (FTIR) confirm the formation of PVA-NiO composites and the SEM image reveals the presence ofNiOparticles which are uniformly distributed throughoutthe composite films. Maximum conductivity was observedin the 4 wt% of NiO in PVA. The room temperature dc electrical conductivity of PVA–NiO composite films increases gradually as the NiO content increases in PVA matrix,which involves hoppingmechanism in the conduction process. 5.ACKNOWLEDGMENT Author would like to thank Dr.H.Nagabhushana, CNR Rao Centre for Advanced Materials Research,Tumkur University,Tumkur;Dr.R.Harikrishna,Department of Chem istry, M.S.Ramaiah Institute of Technology, Bangalore,S.T.Navalea,Functional Materials Research Laboratory,School o f Physical Sciences,Solapur;Prof.M.V.N.Ambika Prasad, Department ofMaterials Science, Gulbarga University, Gulbar ga andDr.Prashanth Kumar, Govt.College, Gulbarga. REFERENCES [1] A.Tawansi, M.D. Migahed, M.I.A. El-Hamid, J. Polym. Sci. Part B: Polym. Phys. 24, pp.2631–2642, 1986. [2] K.A.Abdelkader, Z. Anwar, J. Appl. Polym. Sci. 2, pp.1146, 2006. [3] I. Su, Z.Y. Ma, J.I. Scheinbeim, B.A. Newman, J. Polym. Sci. Part B: Polym. Phys. 33, pp. 85, 1995. [4] A. Tawansi, H.M. Zidan, J. Phys. D: Appl. Phys. 23, pp.1320, 1990. [5] Sato, H., Minami, T., Takata, S and Yamada, T., “Transparent conducting ptype NiO thin films prepared by magnetron sputtering”. Thin Solid Films,236, pp.7–31, 1993. [6] S.Y. Wang, W. Wang, W.Z. Wang, Y.W. Du,“Preparation and characterization of highly oriented NiO (200) films by a pulse ultrasonic spray pyrolysis method”, Mater. Sci. Eng. B 90, pp.133–137, 2002. [7] A.K. Roslik, V.N. Konev, A.M. Maltsev, Oxidation of Metals 43, pp.1, 1995. [8] K.S. Ahn, Y.C. Nah, Y.E. Sung, Applied Surface Science 199, pp.259, 2002. [9] X. Chen, N.J. Wu, L. Smith, A. Ignatiev, Applied Physical Letters 84, pp.2700,2004. [10] I. Fasaki, A. Giannoudakos, M. Stamataki, M. Kompitsas, E. György, I.N.Mihailescu, F. Roubani-Kalantzopoulou, A. Lagoyannis, S. Harissopulos, Applied Physics A 91, pp.487, 2008. [11]Mimani T, Patil K.C. “Solution Combustion Synthesis of Nanoscale Oxides and their Composites”.MaterPhys Mech.,4, pp.1, 2001. [12] H.N. Chandrakala, B. Ramaraj, Shivakumaraiah, G.M. Madhu, Siddaramaiah, J. Mater. Sci. 47, pp.8076, 2012. [13] G.N. Hemanth Kumar, J. LakshmanaRao, N.O. Gopal, K.V. Narasimhulu, R.P.S.Chakradhar,A.VaradaRajulu, Polymer 45, pp.5407–5415, 2004. [14] Sheng-Jian Huang, Hoi-Kwan Lee, Won-Ho Kang, J. Korean Ceram. Soc. 42, pp.77, 2005. [15] M. Kurumova, D. Lopez, R. Benavente, C. Mijangos, J.M. Perena, Polymer 41,pp. 9265,2000. [16] S. Ram, A. Gautam, H.J. Fecht, J. Cai, H. Bansmann, R.J. Behm, Philos. Mag. Lett.87,pp. 361,2007. [17]R. Ricciardi, F. Auriemma, C. De Rosa, F. Laupr˘etre, Macromolecules 37,pp.1921,2004 [18]GezaBodor, Structural Investigation of Polymers, Ellis Horwood Limited, England, 1991. [19]Martin, K., McCarthy, G; North Dakota StateUniv; Fargo, ND, USA. ICDD Grant-in-Aid (1991). [20]Meyers RA (ed.), Interpretation of Infrared Spectra, A Practical Approach, John Coates in Encyclopedia of Analytical Chemistry.Chichester: John Wiley & Sons Ltd, pp. 10815-10837, 2000. Volume 3, Issue 9, September 2014 Page 207 International Journal of Application or Innovation in Engineering & Management (IJAIEM) Web Site: www.ijaiem.org Email: editor@ijaiem.org Volume 03, Issue 09, September 2014 ISSN 2319 - 4847 [21] Mansur HS, Oréfice RL, Mansur AAP. Characterization of poly (vinyl alcohol)/poly (ethylene glycol) hydrogels and PVA-derived hybrids by small-angle X-ray scattering and FTIR spectroscopy,Polymer; 45(21):pp.7193-7202, 2004. [22] Mansur HS, Oréfice RL, Pereira MM, Lobato ZIP, Vasconcelos WL, Machado LJC. FTIR and UV-vi.Study of chemically engineered biomaterial surfaces for protein immobilization.Spectroscopy-AnInternational Journal;16(34):pp.351-360, 2002. [23]SharanabasavaV.Ganachari, RavishankarBhat, RaghunandanDeshpande and VenkataramanA,Recent Research in Science and Technology, 4(4):pp.50-53, 2012. [24] M.A. Chougule, S. Sen, V.B. Patil, Facile and efficient route for preparation of Polypyrrole–ZnO nanocomposites: microstructural, optical, and charge transport properties,J. Appl. Polym. Sci. 125, pp.1418–1424, 2012. [25] A.C. Sonavane, A.I. Inamdar, D.S. Dalavi, H.P. Deshmukh, P.S. Patil,“Simpleand rapid synthesis ofNiO/PPy thin films with improved electro chromic performance”, Electrochim. Acta.55 pp.2344, 2010. [26]S.R. Nalage, M.A. Chougule, ShashvatiSen, V.B. Patil, Novel method for fabrication of NiO sensor forNO2 monitoring, J. Mater. Sci.: Mater. Electron.24, pp.368–375, 2013. [27] J.G. Simmons, in: L.I. Maissel, R. Glang (Eds.), Hand Book of Thin Film Technology, McGrawHill,NewYork,1970. [28] A.K. Jonscher, Thin Solid Films 1 pp.213, 1967. [29] N.F. Mott, E. Davis, Electronic Process in NoncrystallineMaterials, second ed., Clarendon, Oxford, 1979. [30] B.T. Raut, P.R Godse,S.G.Pawar, M.A.Chougule, D.K. Bandgar,ShashwatiSen,V.B. Patil, New process for fabrication of polyaniline–CdS nanocomposites: structural, morphological andoptoelectronic investigations. Phys.Chem. Solids 74, pp.236–244, 2013. [31] S.R.Nalage, S.T Navale, V.B. Patil, Polypyrrole - NiO hybrid nanocomposite: Structural,morphological, optical and electrical transport studies,Measurement 46, pp.3268–3275, 2013. AUTHORS Chivukulasrikanthhasreceived his M.Sc. in Solid State Physics from Osmania University,Hyderabad.Heis presently Assistant Professor of Physics, Govt.Degree College, Gulbarga. Pursuing his Ph.D. in Physics under the supervision of Prof. R.D.Mathad, Department of Physics, Gulbarga University, Gulbarga. His current research interest includes Nanomaterials, polymer nanocomposites. Chakradhar Sridhar B. received the M.Sc. degree in Physics from Gulbarga University, Gulbarga. He is currently pursuing his Ph.D. in Physics under the supervision of Prof M.V.N. Ambika Prasad, Department of Physics, Gulbarga University, Gulbarga. His current research interest includes conducting polymer nano-composites. Dr. B M Nagabhushana has over 26 years of experience in teaching at M.S. Ramaiah Institute of Technology, Bangalore, India. He as more than 15 years of teaching experience and published more than 120 research papers in international peer reviewed journals. Dr R D Mathad is born in Belgaum district of Karnataka, India and has received his M Sc andPh.D in Nuclear Physics from Karnataka University Dharwad. He is presently Professor of Physics, Gulbarga University, Gulbarga. His research comprises diverse areas such as Excitation Energy Transfer; Energy Loss & Radiation induced effects in Polymers, Plasma instabilities in Inertial Confinement Fusion, Cellular Transport Processes, Neutron diffraction studies on Glasses; Polymer Blends, Complexes and Nano- Composites. Volume 3, Issue 9, September 2014 Page 208