University of Babylon /College Of Engineering Electrochemical Engineering Dept.
advertisement

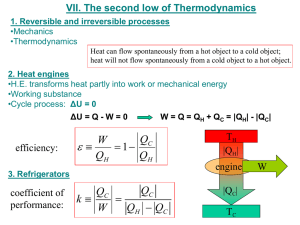
University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics Heat pump Operate between the same to temperature level but direction of heat transfer are reversible and work is required ( e.g., refrigeration cycle) ∆ U = Q net + W ( first law) 0 = QC – QH + W W = ½ QH ½ - QC ½ ……. (1) Entropy analysis DS H = QH TH , DS C = - QC and , ∆S pump = 0 TC ∆S Total = ∆S H + ∆S C + ∆S pump= 0 D S Total = 0 = Q QH - C +0 TH TC ….. (2) ö æTH - 1 ÷÷ From equations 1 and, 2 : W heatpump = Q C çç ø è TC COP = QC TC outlet = = W pump inlet TH -TC COP :coefficient performance pump Ex: Thermodynamic device cools small refrigerator and discards heat to the surroundings at 298.15K .The maximum electric power to which the device is designed is 100 W , the heat load on the refrigeration is 350 W what is the maximum temperature that can be maintained in the refrigerator? University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics If Carnot engine is connected to Carnot pump (refrigerator) so that all the work produced by engine is used by refrigerator Q1 is he heat rejected by Carnot engine Q2 is the heat rejected by Carnot refrigerator WE=WP Engine WE T =1QH TH WP T = -1 QC TC and, pump WE = QH –Q1 and , WP = Q2 -QC æT ö ö ÷÷ = Q C çç - 1 ÷÷ ø èTC ø æT -TC ö æT -T ö ÷÷ ÷÷ = Q C çç Q H çç H T T H C è ø è ø æ T Q H çç 1 TH è æ T -TC Q H = Q C çç èT H -T öæ T H ÷÷çç øè T C ö ÷÷ ø Relation between QH and QC Q =Q H +QC æTH -T Q H çç è TH æTH -T Q H çç è TH æT -TC ö ö ÷÷ ÷÷ = Q C çç T C ø ø è æT -TC ö ÷÷ = (Q - Q H )çç ø è TC æ T -TC Q H = Q çç è T H -TC öTH ÷÷ øT ö ÷÷ ø Relation between QH ( hot reservoir)and Q(heat sink) University of Babylon /College Of Engineering Electrochemical Engineering Dept. Second Stage /Thermodynamics