Extreme Non-LTE H in comets C/2000 WM1 (LINEAR) and C/2001 A2 (LINEAR)
advertisement

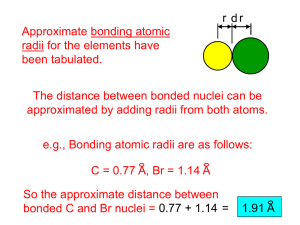
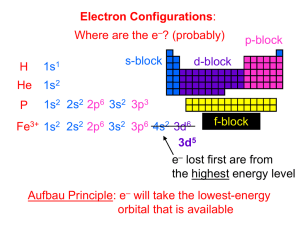
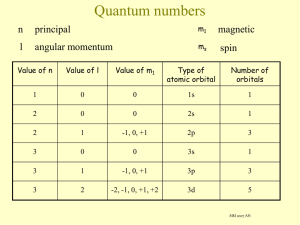
To appear in Astrophysical Journal Supplement Series Extreme Non-LTE H2 in comets C/2000 WM1 (LINEAR) and C/2001 A2 (LINEAR) Xianming Liu, Donald E. Shemansky, Janet T. Hallett Planetary and Space Science Division, Space Environment Technologies, 320 North Halstead, Pasadena, CA 91107 xliu@spacenvironment.net, dshemansky@spacenvironment.net and Harold A. Weaver Space Department, Johns Hopkins University Applied Physics Laboratory, 11100 Johns Hopkins Road, Laurel, MD 20723-6099 ABSTRACT Rotationally resolved molecular hydrogen transitions originating from excitation of highly excited ro-vibrational levels of the X 1 Σ+ g state have been systematically identified for the first time in the Far Ultraviolet Spectroscopic Explorer (F USE) observation of comets C/2000 WM1 (LINEAR) and C/2001 A2 (LINEAR). Spectral assignments for the observed lines of H2 and other atomic and molecular species are given. All observed H2 transitions belong to the Lyman 1 + 1 1 + (B 1 Σ+ u - X Σg ) and Werner (C Πu – X Σg ) band systems. Solar Lyman-α fluorescence excitation of highly ro-vibrationally excited H2 X 1 Σ+ g is found to be almost solely responsible for the observed H2 emission. Resonant excitation of H2 by Lyman-β and other solar lines is very limited. Subject headings: comets: general — comets: individual (C/2000 WM1, C/2001 A2) — molecular process — ultraviolet: solar system 1. INTRODUCTION Comets are among the most primitive objects in the solar system. The chemical and physical properties of these objects provide evidence of conditions in the early solar system. –2– On approach to the Sun, comets form a coma at a surface temperature close to sublimation (∼ 160K). As the gas expands, it initially cools rapidly and adiabatically by converting internal energy into outward flow. Molecules such as water and carbon monoxide in excited ro-vibrational levels are radiatively cooled by infrared (IR) emission. Near the inner coma, radiative loss is not efficient because of optical thickness. As expansion progresses, density decreases and cooling becomes more effective. The gas molecules cool to temperatures below 20 K and reach an outward directional velocity of ∼0.7 km/s (Combi 2002). Heating by solar radiation counteracts the cooling process. Photodissociation produces kinetically hot fragments, and excess energy is delivered collisionally (Huebner et al. 1992). During the past several years, there have been a broad range of observational investigations of comets (Bockelee-Morvan et al. 1998; Combi et al. 1998; Meier et al. 1998; Chiu et al. 2001; Cochran & Cochran 2001; Morgenthaler et al. 2001; Mumma et al. 2001; Feldman et al. 2002; Dello Russo et al. 2002, 2004, 2005; Weaver et al. 1999, 2002; Lecacheux et al. 2003; Brooke et al. 2003; Crovisier et al. 2004). Most have been carried out in submillimeter and IR wavelength regions with ground-based telescopes, although spacecraft observation in submillimeter (Chiu et al. 2001; Bensch et al. 2004), IR (Cernicharo & Crovisier 2005), vacuum ultraviolet (VUV) regions (Combi et al. 1998; Feldman et al. 2002; Weaver et al. 2002; Bemporad et al. 2005) have made significant contributions. Radiowave observations of the OH 18 cm transition have also provided valuable insight into the velocity and anisotropy of outgoing gas and collisonal quenching of the maser transition pumped by ultraviolet (UV) radiation (Colom 1999; Schloerb et al. 1999; Crovisier et al. 2002). Many stable species such as H2 O, CO, CH3 OH, CH4 CH3 CH3 , CHCH, HCN, NH3 , OCS, and H2 CO and fragments such as OH, NH2 , and CN have been investigated. The relative abundance of volatile molecules could vary significantly from comet to comet, attributed to differences in environment under which the comet was formed (Mumma et al. 2003; Ruscic et al. 2005). Rotational analysis of IR spectra reveals that the inner coma temperature of comets ranges from ∼30 to 140 K (Brooke et al. 2003; Dello Russo et al. 2004, 2005; Mumma et al. 2005). While hydrogen has the highest elemental abundance, molecular hydrogen has only recently been observed with F USE by Feldman et al. (2002), who identified three P(1) lines 1 + of the B 1 Σ+ u - X Σg (6,vi ) sequence. H2 should be present in comets in very significant quantity. While H2 is the most volatile molecule, it has been shown experimentally that H2 is readily trapped and retained by water ice (Laufer et al. 1987; Bar-Nun et al. 1987). Thus, hydrogen molecules initially trapped during comet formation and subsequently retained are released during the evaporation process. It has been demonstrated experimentally that H2 is a significant product of H2 O photodissociation (Slanger & Black 1982; Mordaunt et al. 1994; Hwang et al. 1999; Harich et al. 2000, 2001a). It is also known that highly excited hydrogen molecules are generated from amorphous ice by electron impact (Kimmel et al. 1994, 1995; –3– Herring-Captain et al. 2005). Two factors are responsible for the difficulty in detecting H2 in comet comae. The strong IR emissions from H2 O, CO, and, in particular, ro-vibrational excited OH, a major molecular photofragment of H2 O, strongly blend with many quadrupole transitions of H2 , preventing effective utilization of the IR spectrum for detection and measurement of H2 . H2 in the VUV region as we conclude here, originates from excitation of electronic states from highly excited ro-vibrational levels of the X 1 Σ+ g state, sourced in the process of solar photodissociation of H2 O. As a result, the spectral emission distribution is significantly different from those observed in laboratory or other environments, making spectral identification difficult. In the absence of radiationless deactivation, activated H2 X 1 Σ+ g has very long lifetimes, 4 10 ranging from 9.73×10 s for the (vj =9, Jj =0) level to 3.4×10 s for the(vj =0,Jj =2) level (Wolniewicz et al. 1998). These long lived hydrogen molecules are excited by solar radiation and charged particles into singlet-ungerade states, and subsequently radiatively decay to the X 1 Σ+ g state, giving rise to observable VUV emission. This paper reports the systematic assignment of spectral lines from F USE observation of comets C/2000 WM1 (LINEAR) and C/2001 A2 (LINEAR). Preliminary results, with emphasis on CO, Ar, O I and O VI, have been reported in papers by Feldman et al. (2002) and Weaver et al. (2002). Feldman et al. (2002) assigned three Lyman band transitions of H2 on the basis of the resonant excitation by Lyman-β line. However, as Feldman (2005) noted, many observed features remained unidentified. This work presents a systematic assignment of the observed transitions. As expected, almost all previously unassigned features are molecular hydrogen transitions. The analysis shows that almost all observed H2 spectral lines can be accounted by solar Lyman-α excitation of H2 X 1 Σ+ g formed in highly excited ro-vibrational levels. Section 2 briefly summarizes the observational data. Section 3 outlines the data analysis procedure and lists spectral assignments of observed features. Section 4 reviews relevant photochemistry of water molecule. Section 5 discusses the excitation mechanism. 2. Observation and Data Description Detailed descriptions of the F USE observations of comets C/2000 WM1 and C/2001 A2 have been given by Weaver et al. (2002) and Feldman et al. (2002). Only a brief summary will be given here. F USE has four co-aligned telescopes with spectrographs. The optics of two telescopes coated with silicon carbide and two coated with lithium fluoride/aluminum have spectral resolution better than 0.4 Å, and cover the 905 to 1187 Å wavelength range. –4– For both C/2000 WM1 and /2001 A2 observations, the 30 × 30 entrance aperture was used with the comet nucleus centered in the aperture. Because of the extended, nonuniform emission within the aperture, the effective spectral resolution was 0.25 Å. The observation of comet C/2001 A2 started on July 12, 2001, at 13:38 UT. An exposure time of 16,549 s was made by accumulating spectra in each of five contiguous orbits. About 60% (9,530 s) of the data were acquired through the dark terrestrial atmosphere. The heliocentric and geocentric distances of comet C/2001 A2 at the time of observation were 1.20 AU and 0.30 AU, respectively, and the heliocentric radial velocity was 22.8 km s−1 . Comet C/2000 WM1 was observed between December 7 and 10, 2001. A total exposure time of 36,557 s was made over 21 orbits. About 95% (34,577 s) of the WM1 data were obtained through the night sky. At the time of the observation, heliocentric and geocentric distances were 1.12 and 0.34 AU, respectively. The heliocentric radial velocity was -28.3 km s−1 . Table 1 summarizes the observing configurations of both comets. Figures 1 and 2 show the F USE spectra of comets C/2001 A2 and C/2000 WM1, respectively. Comet activity was found to be very stable during the observation periods. Neither the continuum brightness nor the stronger discrete emission varied by more than a few percent over different orbits. For both comets, the exposures from different contiguous orbits were co-added and extracted fluxes were converted to average brightness in the 30 × 30 aperture. The data analyzed here were accumulated from exposures obtained when the spacecraft was in Earth shadow. The differential brightness, in units of Rayleighs per Å, is the average over the aperture. After background removal, the observed spectral features were fitted with a gaussian line profile. The center wavelength and full width at half maximum (FWHM) for both C/2001 A2 and C/2000 WM1 are listed in ascending wavelength in the first and second columns of Table 2. The third column identifies the target comet. While a few lines are unique to the comets A2 or WM1, many emission lines are common to both comets. In general, more lines were observed in comet WM1, which probably reflects better signal-to-noise ratio as a result of a longer exposure (night) time (34,577 vs 9,530 s). It should be noted that the brightness of comet A2 is actually slightly higher than that of WM1. The fourth and fifth columns list the primary and secondary spectral assignments along with their laboratory or model wavelengths (see Section 3) for the observed lines. 3. Analysis and Results Some transitions, arising from H I, O I, O VI and CO, have been assigned by Weaver et al. (2002) and Feldman et al. (2002). Feldman et al. (2002) also assigned three H2 lines –5– 1 + to the P(1) branches of the (6,1), (6,2) and (6,3) band of Lyman (B 1 Σ+ u - X Σg ) system. They attributed the appearance of the P(1) transitions to solar H Lyman-β pumping of the P(1) line of the (6,0) band. Additional assignments to C I, O I, O VI and CO beyond those given by Weaver et al. (2002) and Feldman et al. (2002) are possible using the NIST Atomic Spectra Database (Ralchenko et al. 2005) and CO atlas of Eidelsberg et al. (1991). H2 is the origin of the remaining majority of unassigned lines based on the present analysis (section 5.3). The initial identification was made using the non-LTE fine structure H2 model developed by Hallett et al. (2005). An important feature of the model is that it is capable of predicting transitions 1 + 1 1 + 1 1 + Σu from every ro-vibrational level (J<11) of the X 1 Σ+ g , B Σu , C Πu , B Σu , D Πu , B 1 and D Πu states. The interaction of H2 with solar photons, charged particles and chemical reactions at every ro-vibrational level (J<11) of these states can be included (Hallett et al. 2005). By assuming H2 is formed in some high J (e.g. 9 or 10) levels, this model is capable of assigning a few transitions in Table 2 to H2 and thus provides confidence that H2 is a spectral carrier for at least some of the transitions. However, many observed lines cannot be accounted for by the model, because of the current J < 11 restriction. An H2 architecture containing rotational levels to J = 25 is presently under construction in our program. Assuming that H2 is one of the emitting species, a second and simpler approach was taken. Given the likelihood that highly populated rotational and vibrational levels not normally observed in laboratory sources were involved, a model using accurate state energies for rotational levels as large as J = 20 was utilized to generate emission transitions for correlation with the observed lines. For this purpose the electron impact induced emission model developed by Liu et al. (1995), Abgrall et al. (1997) and Jonin et al. (2000) was utilized to find H2 transitions that are near the frequency of the likely drivers for the emissions, the H Lyman-α and Lyman-β solar lines. The model was based experimental term values of Roncin & Launay (1994) and Dabrowski (1984) and theoretical calculations of Abgrall et al. (1993a,b,c, 1994, 1997) and was conveniently used to calculate accurate H2 transition wavelength up to J=20. In the early stage of analysis, the details of H2 production and excitation mechanisms were not clearly identified. The non-discriminating nature of electron impact excitation ensured that the wavelengths of all possible emissions from J≤ 20 levels of 1 1 + 1 1 + Σu and D 1 Πu states were generated. The temperature B 1 Σ+ u , C Πu , B Σu , D Πu , B of H2 for the model was set over 6,000K so that it could also provide the wavelengths of high J lines. At the same time, the absorption oscillator strength for these transitions were calculated from the transition probabilities of Abgrall et al. (1993a,b,c, 1994). It was found that some spectral lines could be assigned to resonance excitation fluorescence of highly excited H2 by Lyman-α. These spectral lines not only agree with expected wavelength positions, but their relative intensities are also consistent with the branching-ratios calculated –6– from the transition probabilities of Abgrall et al. (1993a,b,c, 1994, 2000). Two common features were noted for these comet lines: the resonance lines are close to the Lyman-α center wavelength with reasonable oscillator strength, and the resonance transitions usually start from high (v,J) levels of the X 1 Σ+ g state. Having established that fluorescence from resonance excitation of H2 by solar Lymanα is responsible for many observed transitions, a new program searching for H2 resonance excitation by all strong solar lines was developed. Since many H2 transitions originate from the high J levels, it is critical that accurate values of their transition frequencies be 1 + 1 1 + established. Experimentally determined level energies of X 1 Σ+ g , B Σu , C Πu , B Σu and D 1 Πu states are available from work of Dabrowski (1984), Abgrall et al. (1994), Roncin & Launay (1994) and Roncin (private communication). Experimentally unavailable levels were calculated from theoretical values of Abgrall et al. (1993c, 1994, 2000) for J up to 25. As noted in Abgrall et al. (1997), Abgrall et al. (1993c, 1994, 2000) have slightly adjusted the ab initio potential so that the calculated transition frequencies for the lowest J levels agree with experimental values. As a result, calculated frequencies deviate less than 1.5 cm−1 from the high-resolution experimental frequencies of Abgrall et al. (1993a,b, 1994) and Roncin & Launay (1994). Moreover, the relative values of the calculated transition probabilities for 1 + 1 1 + 1 1 + 1 + the low-J levels of B 1 Σ+ u - X Σg , C Πu – X Σg , D Πu – X Σg and most of the B Σu – X 1 Σ+ g band system have been experimentally verified by the high-resolution electron impact induced emission investigations of Liu et al. (1995), Abgrall et al. (1997) and Jonin et al. (2000). H2 spectral assignments based on experimental results of Dabrowski (1984), Abgrall et al. (1994), Roncin & Launay (1994) and Roncin (private communication) and calculated results of Abgrall et al. (1993a,b,c, 1994, 1997, 2000) are listed in the fourth and fifth columns of Table 2. The fourth column gives the primary spectral assignment while the fifth column gives secondary assignments. Spectral carriers other than H2 are explicitly identified in the beginning of the assignment while experimentally observed or model calculated wavelengths are listed in parentheses following the assignment. If the spectral carrier is not specified, the carrier is, by default, H2 . H2 transitions are labeled in terms of Ji (vj ,vi )∆J β, where i and j refer to the lower and upper states, β is electronic designation of excited state, and ∆J=-1, 0 and +1 correspond to P, Q, and R transitions. The lower electronic state, X 1 Σ+ g , has been dropped from electronic designation. Assignment entries followed by a question mark indicate that the suggested assignments are possible but not definitive. These transitions usually arise from (vi ,Ji ) levels higher than those that can be produced with ground state H2 O and Lyman-α photolysis frequency. However, if some water molecules are in vibrationally excited states, the production of H2 in these levels becomes energetically possible. Thus, the uncertainty largely reflects the extent of the contribution from the denoted transition. On –7– the other hand, assignment entries with double question marks indicate that a reasonable assignment is presently unknown to the authors. It is possible that the unknown transitions belong to H2 with J>25. In some cases, the fifth column serves as a short explanatory note for the spectral assignment listed in the fourth column. It is important to note that the H2 assignments listed in Table 2 are primarily based on the agreement in transition wavelength with the laboratory or model value, on the calculated emission branching-ratios, absorption oscillator strength, and the H2 excitation mechanism presented in section 5. Because no production cross section of H2 (vi ,Ji ) is currently available and because the relative strength of solar photoexcitation is only partially taken into account, it is possible that the order of several primary and secondary assignments may be reversed or even revised. The lack of H2 transitions in the far ultraviolet (FUV) also leads to uncertainties in a few assignments. Furthermore, even if the photoexcitation is solely restricted to the Lyman-α line, there are usually multiple H2 transitions whose wavelengths are aligned with those of the observed comet features. The assignments in Table 2 denote one or two of the strongest transitions for a given feature. 4. Photochemistry of H2 O In this section, we summarize relevant photochemistry of H2 O to present further justification of our assignments. The ground (X̃ 1 A1 ) state of H2 O has C2V symmetry with the molecular orbital electron configuration (1a1 )2 (2a2 )2 (1b2 )2 (3a1 )2 (1b1 )2 . The excitation of an electron out of the nonbonding 1b1 orbital to a Rydberg orbital leads to Rydberg series with a bent equilibrium structure converging to the ground ionic state X̃ 2 B1 of H2 O+ . In contrast, the promotion of an electron from the inner 3a1 orbital results in a quasilinear Rydberg series converging to the second ionic à 2 A1 state (van Harrevelt & van Hemert 2000a). Crossings of the potential surfaces for linear and bent states occur frequently and are one of the important factors for the predissociation of the bound states <12 eV. The first broad absorption continuum of H2 O, from 1950 to ∼1420 Å with the maximum near 1670 Å, corresponds to the first allowed electronic transition, the X̃ 1 A1 – à 1 B1 band, which arises from the 1b1 → 3sa1 excitation (Lee & Suto 1986; Yoshino et al. 1996; Chen et al. 1999; van Harrevelt & van Hemert 2001; Parkinson & Yoshino 2003). The second broad absorption continuum, from ∼1420 to ∼1120 Å with maximum near 1280 Å arises from the excitation to the B̃ 1 A1 state which results from strongly coupled 3a1 → 3sa1 and 1b1 → 3px b1 excitation (Wiede & Schinke 1989; Chan et al. 1993; Christiansen et al. 2000; van Harrevelt & van Hemert 2003). The next higher states are C̃ 1 B1 and D̃ 1 A1 which arise from excitation of the 1b1 electron to the 3p Rydberg orbital. Unlike the broad X̃ 1 A1 – à 1 B1 –8– and X̃ 1 A1 – B̃ 1 A1 band systems, the X̃ 1 A1 – C̃ 1 B1 and X̃ 1 A1 – D̃ 1 A1 transitions, with electronic origins of 1240 and 1219 Å, respectively, are relatively sharp. The neutral excited states of water must be either dissociative or predissociative, because no discrete emission has been observed from electronically excited H2 O,. The à 1 B1 state is purely repulsive and correlates to the OH(X 2 Π)+H(2 S) limit. The X̃ 1 A1 – à 1 B1 excitation is, therefore, a direct dissociative process, and the excess energy in the dissociation from the à 1 B1 state is mainly deposited in the kinetic energy of the products (Anderson & Schinke 1987; Engel et al. 1992; Crim 1993). The measurements of Farmanara et al. (1999) have placed an upper limit of 20 fs for the lifetime of the à 1 B1 state. An oscillatory structure with an almost constant spacing of ∼810 cm−1 appears on the X̃ 1 A1 – B̃ 1 A1 continuum (Wang et al. 1977; Chen et al. 2004). While early work of Wang et al. (1977) attributed it to the activation of bending motion, Wiede & Schinke (1989) have suggested it arises from the resonant trajectories due to the coupling of stretching and bending motions in the B̃ 1 A1 state. van Harrevelt & van Hemert (2000a,b) have recently shown that the resonance persists even at high energies. The B̃ 1 A1 state is strongly predissociated through the conical intersection with the X̃ 1 A1 state. The B̃ 1 A1 state can also be predissociated by à 1 B1 through Renner-Teller coupling. Thus, while the B̃ 1 A1 state adiabatically correlates to OH(A 2 Σ+ )+H(2 S), the nonadiabatic crossing from the B̃ 1 A1 state to the potential energy surfaces of either the à 1 B1 or X̃ 1 A1 state leads to the production of ro-vibrationally excited OH(X 2 Π). In the linear approach of H to OH, the repulsive potential curve of OH(X 2 Π)+H(2 S) can cross the attractive OH(A 2 Σ+ )+H(2 S) curve. Such a crossing, however, is not possible in the lower (i.e. bent) symmetry. As a result, the conical intersection of the B̃ 1 A1 and X̃ 1 A1 states occurs at a collinear H-O-H geometry. The high torque acting in the neighborhood of the conical intersection is responsible for the extremely high rotational excitation in the OH(X 2 Π) fragment observed experimentally (Mordaunt et al. 1994; Hwang et al. 1999; Harich et al. 2000, 2001a,b). In addition to the H-O-H conical intersection, the B̃ 1 A1 state has a second conical intersection with the collinear O-H-H geometry. The calculation by Schatz (1985) suggested that the O-H-H collinear conical intersection is responsible for the O(1 D) + H2 dissociation channel of the B̃ 1 A1 state. Although O(1 D) has been observed experimentally, in the O(1 D)+H2 channel, the H2 product has not been characterized. Ab initio calculations, however, have indicated the production of highly excited H2 (van Harrevelt & van Hemert 2000a,b; van Harrevelt et al. 2001). The C̃ 1 B1 state is predissociated by two mechanisms: a heterogeneous coupling to the B̃ 1 A1 state by rotational motion along the a-axis and a homogeneous purely electronic coupling to the C̃ 1 B1 state (Ashold et al. 1984; Kuge & Kleinermanns 1989; Edery & Kanaev 2003). The first mechanism yields the OH(A 2 Σ+ ) radical while the second mechanism produces OH(X 2 Π) (Fullion et al. 2001). Steinkellner et al. (2004) have obtained 0.5±0.1 ps for the –9– lifetime of the heterogeneous predissociation out of the C̃ 1 B1 state. The D̃ 1 A1 state is also strongly predissociative by an avoided crossing with the B̃ 1 A1 state at bent HOH geometry (Hirst & Child 1992; van Harrevelt & van Hemert 2000a). No resolvable rotational structure of the D̃ 1 A1 state has been observed. Owning to the dominance of H Lyman-α, the dissociation of H2 O by solar radiation is largely characterized by the photodissociation dynamics near the Lyman-α line. In general, excitation H2 O with wavelengths shorter than 1300 Å gives rise to four possible dissociation channels. The threshold energies of these channels, based on recent thermochemical data of Ruscic et al. (2002, 2005), can be obtained as H2 O → OH(X 2Π) + H(2 S); ∆E = 41128 ± 24 cm−1 (1) H2 O → OH(A2 Σ+ ) + H(2 S); ∆E = 73530 ± 24 cm−1 (2) −1 H2 O → O( P ) + 2H( S); ∆E = 76721 ± 49 cm 3 2 H2 O → O( D) + H2 (X 1 2 Σ+ g ); (3) −1 ∆E = 56471 ± 49 cm (4) 1 Other spin-allowed dissociation channels such as H2 (X 1 Σ+ g )+O( S) are possible. As noted by Huestis & Slanger (2006), no experimental measurement has been made for the 1 −1 H2 (X 1 Σ+ g )+O( S) channel. However, with a threshold of 74395 cm , it is presumably unimportant because it requires excitation of an A (in terms of Cs point group) state that lies more than 12 eV above the X̃ 1 A1 state. Many experimental measurements (Slanger & Black 1982; Mordaunt et al. 1994; Hwang et al. 1999; Harich et al. 2000, 2001a) with 1216 Å radiation have been carried out. Most investigations have focused on the measurement of OH(A 2 Σ+ ) and OH(X 2 Π) products and on the detailed energy distribution. OH(X 2 Π) is found to be predominantly produced in the lower vibrational levels with highly excited rotational levels with inverted rotational state population distribution. The OH(A 2 Σ+ ) fragment is also formed vibrationally cold with highly inverted rotational population distribution that peaks near the highest rotational level energetically accessible at the excitation energy used. The quantum efficiency for H atom production at 1216 Å is determined to be 1.02 and absolute branching ratios for reactions (1-4) are found to be 0.64, 0.14, 0.11 and 0.11, respectively (Mordaunt et al. 1994). 5. Discussion In this section, we discuss the excitation mechanisms of H2 based on the F USE observation of comets C/2001 A2 and C/2000 WM1. Detailed modeling of the production mechanism of H2 from H2 O will be given with predicted emission in future work. – 10 – 5.1. The primary H2 excitation mechanism The excitation of H2 emission observed in comets C/2001 A2 and C/2000 WM1 arise almost exclusively from photoexcitation by the solar H Lyman-α line. Evidence for measurable excitation by photoelectrons has not been found. With the exception for the (vj =6, Jj =0) level of the B 1 Σ+ u state, resonance excitation by solar Lyman-β is negligible. Almost all identified H2 emission features are attributable to resonant excitation by solar Lyman-α. Even for the (vj =6, Jj =0) level, the role of Lyman-β resonant excitation is limited to cold H2 , which probably is not produced by dissociation of H2 O. It is possible that Lyman-α excitation from the (vi =4, Ji =1) level of the X 1 Σ+ g state also contributes to the emission 1 + from the (vj =6, Jj =0) level of the B Σu state. The sparse distribution of the H2 lines shown in Figures 1 and 2 rules out significant emission from electron impact excited H2 . The fact that only emission from the B 1 Σ+ u and C 1 Πu states are observed and no emission from the D 1 Πu and B 1 Σ+ states are seen u between 900 and 1050 Å region, where they are fairly strong, is also consistent with the absence of excitation by charged particles. In fact, one of the leading causes for the failure in using the non-LTE fine structure H2 model in our initial analysis was the inclusion of excitation by electrons. Solar flux measurements by Solar Ultraviolet Measurement of Emitted Radiation (SUMER) Extreme Ultraviolet Spectrometer onboard the Solar and Heliospheric Observatory (SOHO) shows that between 800 and 1600 Å, the Si III line at 1206.510 Å is the second strongest feature, after Lyman-α. The Lyman-β line is actually weaker than Si III (1206.510 Å), N V (1238.821 Å), and O I (1302.168, 1304.858, 1306.029 Å) lines. In addition to being the strongest feature, the solar Lyman-α line is also very broad. Even at ±3 Å from line-center, the H Lyman-α flux is still very significant (Lemaire et al. 1998). Thus, H2 lines with significant oscillator strengths, within the 1215.672±3 Å region, and with reasonable populations can be resonantly excited and contribute to the observed comet features. 5.2. Possible minor H2 excitation mechanisms With the exception of the 1028.777 Å feature in comet A2 and the 1031.898 and 1163.786 Å lines in comet WM1, all other H2 lines in Table 2 can be at least qualitatively explained by resonant excitation by the broad Lyman-α line. The 1028.777 Å feature, having a FWHM of 0.683 Å, must consist of at least two transitions. The 3(5,3)Q C (1028.777 Å) line, which arises from pumping of 3(5,8)Q C by Lyman-α, can be considered to be one contributor. The second contributor is the 1(1,1)Q C (1028.989 Å) transition, which arises from pumping – 11 – of the 1(1,5)Q C line at 1206.639 Å by solar Si III lines whose rest and Doppler shifted wavelengths are 1206.510 and 1206.602 Å, respectively. The problem with this additional assignment is that stronger lines in Q(1) emission for the (1,0), (1,3) and (1,4) are not positively identified. So, it is questionable whether the 1(1,1)Q C line can be considered as the second contributor to the 1028.777 Å feature. Finally, the (2,0) band of the 3pπ E 1 Π - X 1 Σ+ transition of CO is at 1029.295 Å, which is within the range of 1028.777±0.683 Å. However, the oscillator strength of the (2,0) band is about 58 times weaker than the (1,0) band at 1051.714 Å, which is not identified in the F USE A2 spectra. Presumably, vibrationally excited CO could be produced from dissociation of CO2 or H2 CO (Feldman et al. 2006). As pointed out by Liu & Dalgarno (1996), there is a near coincidence of H2 3(1,1)Q C-X (1031.865Å) to the O VI 2 S1/2 - 2p 2 P3/2 (1031.912 Å) solar line. The apparent (i.e. Doppler shifted) wavelength of the O VI transition for comet WM1 is 1031.815 Å. The oscillator strength of the 3(1,1)Q C-X transition (f =2.811×10−2) is quite large, and a moderate population in the (vi =1, Ji =3) level in comet WM1 can, therefore, result in observable resonance emission induced by O VI 2 S1/2 - 2p 2 P3/2 solar resonant pumping. The first 5 members of the 3(1,vi )Q C-X transition (i.e. vi =0-4) have an emission branching-ratio of 0.215, 0.152, 0.004, 0.210, and 0.277, and transition wavelength of 989.729, 1031.865, 1075.030, 1119.079, and 1163.805 Å, respectively. For comet WM1, the feature at 1163.786 Å can be attributed to the 3(1,4)Q C-X line. The Q(3) line of the (1,0) band is difficult to identify because of the overlapping O I 2s2 2p4 3 PJ - 2s2 2p3 (2 Do )3s 3 D (J=2 & 1) transitions. The Q(3) line of the (1,2) band is too weak to be observed. The Q(3) line for the (1,3) band at 1119.079 Å, is expected to be stronger than its counterpart of the (1,1) band. However, it cannot be positively identified in comet WM1. Instead, a small dip between 1119.6 and 1120.4 Å is seen in the composite spectrum of WM1. Feldman (2005) reported that the Q(3) emission for both (1,3) and (1,4) bands has been detected in comet C/2001 Q4 (NEAT), with significantly better signal/noise than WM1, and this may be taken as evidence for the O VI pumping mechanism. Thus, it is probable that O VI resonant pumping is present in WM1 and is at least partially responsible for spectral features at 1131.898 and 1163.786 Å. The feature at 1031.898 Å may also have small contribution from the O VI 2 S1/2 - 2p 2 P3/2 emission, originated from charge exchange reaction between the O VII ion in solar wind and H I in comet. The O VI 2s 2 S - 2p 2 P transition was predicted to be the strongest lines in a calculation by Kharchenko & Dalgarno (2001). However, the emission from the other spin-orbit component of O VI, the 2s 2 S1/2 - 2p 2 P1/2 transition at 1037.613 Å, cannot be identified in comets WM1 and Q4. An unpublished calculation by one of us (DS) indicated that emission intensity from the 2 P1/2 component is ∼ 56% of the 2 P3/2 level. Thus, the production of emission at 1031.898 Å by charge capture is not strong. – 12 – As will be shown in section 5.3, the dissociation of H2 O by solar Lyman-α produces highly excited H2 . The present study raises an interesting question on the excitation source for the (vj =6, Jj =0) level of the B 1 Σ+ u state. The P(1) branch emission of the (vj =6,vi ) 1 + 1 + of the B Σu - X Σg system is normally associated with Lyman-β resonant excitation in dayglow when the(vi =0, Ji =1) level of the X 1 Σ+ g state has a very significant population. Feldman et al. (2002) first identified three P(1) branches for (6,1), (6,2) and (6,3) bands in comet A2 and attributed them to the Lyman-β resonant excitation via the P(1) line of the (6,0) band. It should be noted, however, that the 1(6, vi )P B-X emissions can also 1 + arise from the Lyman-α resonant pumping of the P(1) line of the B 1 Σ+ u - X Σg (6,4) band. If H2 were exclusively produced from photodissociation of H2 O, the (vi =4, Ji =1) level would likely have a higher population than the (vi =0, Ji =1) level. The oscillator strength of the P(1) (6,0) band (9.904×10−3 ) is about 22 times larger than that of the (6,4) band (4.223×10−4). However, the solar flux at the latter (1215.882 Å) is much stronger than that at the 1025.935 Å. The P(1) line of the (6,4) band is almost at the peak flux of the Lymanα line and is insensitive to small Doppler shifts. In contrast, solar Lyman-β is weak and relatively narrow. Thus, the effectiveness of H Lyman-β in pumping the P(1) line of the (6,0) band should have a strong dependence on Doppler shift. The rest wavelength of Lyman-β is 1025.722 Å. Because of the Doppler shifts, the apparent wavelengths for comets A2 and WM1 are 1025.800 and 1025.625 Å, respectively. Based on the smaller differences in the center wavelength for comet A2 (0.135 Å) than for WM1 (0.315 Å), the relative intensities of the P(1) (6,vi )B lines in A2 to the other H2 emission features should be much stronger than those in WM1 if the Lyman-β is dominant in resonant excitation of the (vj =6, Jj =0) level. Figures 1 and 2 do show significant changes in relative intensities. Thus, substantial emission from the (vj =6, Jj =0) level of the B 1 Σ+ u state is probably resonantly excited by Lyman-β line. On the other hand, the lack of emission from the (vj =2, Jj =1) and (vj =5, Jj =1) levels of the C 1 Π− u state for comet A2 or WM1 suggests that hydrogen molecules excited to the (vj =6, Jj =0) level are not produced from dissociation of H2 O, at least not nascently. Both 1(5,3)Q C (1025.886 Å, f =1.466×10−2) and 1(3,2)Q C (1025.911 Å, f =2.080×10−2) lines have much greater oscillator strengths and are closer to the Lyman-β transition than those of the 1(6,0)P B line. The absence of the emission from (vj =2, Jj =1) and (vj =5, Jj =1) levels of the C 1 Π− u state indicates that H2 excited to the (vj =6, Jj =0) level is probably from evaporation of the cold H2 trapped internally in the comet. 5.3. Inferred H2 O → H2 photochemistry Transitions listed in Table 2 provide the identification of the initial (vi ,Ji ) levels of H2 from which excitation by solar Lyman-α takes place. The P(9) and R(7) branches of the – 13 – (15,vi ) bands of Lyman system and the Q(11) branch of the (3, vi ) of the Werner band system, for instance, arise from excitation of the (vi =6,Ji =9) and (vi =6,Ji =11) levels of the −1 X 1 Σ+ g state. They are 25,013.67 and 26,480.6 cm , respectively, above the (vi =0,Ji =0) level (Dabrowski 1984). The lowest observed initial level is the Ji =5 of vi =2, located at 9,654.15 cm−1 . The highest initial level that is positively identified is (vi =4,Ji =20), which has energy term value 30,311.8 cm−1 . While these numbers indicate variation in the internal excitation in H2 formation, they clearly demonstrate that H2 is produced in highly excited levels. Inspection of the observed initial levels also suggests a tendency of H2 to be formed in very high J-levels. Excitation by Lyman-α from levels such as (vi =2, Ji =12,14, & 18), (vi =3, Ji =18,20,& 22), (vi =4, Ji =11,12,& 20), (vi =5, Ji =12,& 19) and (vi =6, Ji =9,11,& 15) have been observed. This is best illustrated by examining transitions 5(1,5)P C-X, 9(1,5)R C-X, and 20(15,3)P B-X, whose wavelengths are 1216.993, 1217.001, and 1217.031 Å, respectively. Due to the closeness of the transition wavelength, solar photon flux of Lyman-α at these positions is almost identical. The absorption oscillator strengths for the 5(1,5)P C-X, 9(1,5)R C-X, and 20(15,3)P B-X transitions are 7.105×10−3, 1.968×10−2 , and 6.747×10−3 , respectively. If the populations at (vi =5,Ji =5; 19807.03 cm−1 ), (vi =5,Ji =9; 22251.21 cm−1 ), and (vi =3,Ji =20; 27891.56 cm−1 ) were equal, the emission from the (vj =1, Jj =10) level of the C 1 Π+ u state would have been the strongest while that from the (vj =15, 1 + Jj =19) level of the B Σu would have been the weakest. However, only the emission from the (vj =15, Jj =19) level of the B 1 Σ+ u is observed in the F USE spectra, showing that H2 population at the (vi =3,Ji =20) level is significant while the (vi =5,Ji =5) and (vi =5,Ji =9) levels are negligible. It should be noted that the (vi =3,Ji =20) level can not be produced by Lyman-α dissociation of the ground state H2 O. The appearance of the 18(15,0)R B line at 1066.567 Å in comet A2 spectra suggests that cross section for producing H2 (vi =3,Ji =20) from H2 O must be very significant. The indication that excessive energy released during the dissociation of H2 O is mainly deposited in the rotational motion of H2 is similar to experimental observations of OH production from H2 O at the Lyman-α wavelength (Mordaunt et al. 1994; Hwang et al. 1999; Harich et al. 2000, 2001a), where extremely high rotational excitation of OH is observed. The similarity arises from the resemblance in H2 O structures of the B̃ 1 A1 potential energy surface, from which most of OH and H2 are formed. Theoretical calculations of van Harrevelt & van Hemert (2000a,b) have demonstrated that the potential energy surface of the B̃ 1 A1 state has two minima: one for linear H-O-H geometry, and the other for linear H-H-O geometry. Both minima occur at the intersection of the attractive H+OH(A 2 Σ+ ) and the repulsive H+OH(X 2 Π) potential energy curves. The nuclear motions of the X̃ 1 A1 and B̃ 1 A1 states are strongly coupled in the neighborhood of the intersections. The production – 14 – of OH from H2 O at Lyman-α takes place via the linear H-O-H geometry. The high torque acting in the neighborhood of the H-O-H conical intersection is responsible for the extremely high rotational excitation in the OH(X 2 Π) fragment observed experimentally (Mordaunt 1 et al. 1994; Hwang et al. 1999; Harich et al. 2000, 2001a,b). The H2 (X 1 Σ+ g ) + O( D) product channel arises from the dissociation via the H-H-O linear geometry (Schatz 1985; van Harrevelt & van Hemert 2000b, 2001). While very little laboratory data for the H2 + O(1 D) channel is available, ab initio calculations of van Harrevelt & van Hemert (2000a,b); van Harrevelt et al. (2001) have suggested H2 is formed in highly excited levels. The large torque in the neighborhood of the O-H-H geometry is responsible for the preferential H2 population at the high J levels. 1 + 1 It should be noted all observed H2 lines are assigned to the B 1 Σ+ u - X Σg and C Πu – 1 + 1 1 + Σu – X 1 Σ+ X 1 Σ+ g transitions. The absence of the B g and D Πu – X Σg transitions 1 + 1 1 + can be attributed to three factors. First, the normal B 1 Σ+ u – X Σg and D Πu – X Σg 1 + 1 1 + transitions are often weaker than their counterparts of B 1 Σ+ u - X Σg and C Πu – X Σg band systems (Jonin et al. 2000). This is because the electronic transition moment of the 1 + 1 1 + 1 + 1 + B 1 Σ+ u – X Σg and D Πu – X Σg are weaker and some levels of the B Σu and D Πu 1 states either dissociate or predissociate. Moreover, the B 1 Σ+ u and D Πu states are higher 1 in energy than the B 1 Σ+ u and C Πu states. H2 must be formed in very high ro-vibrational 1 levels to be excited to the B 1 Σ+ u and D Πu states. The solar photon energy distribution and the conservation of energy, however, prevents H2 from being produced in some of these high energy levels sourced by photodissociation of H2 O alone. As will be shown in a future paper by Liu et al. (in preparation), the principal production of mechanism of H2 is Lymanα photolysis of H2 O in its ground vibrational level. Based on the recent thermochemical data listed for reaction (4), the maximum energy available for internal excitation of H2 from Lyman-α photons is 25,788 cm−1 . Even after consideration of the width of solar Lyman-α line (±3 Å) and initial rotational population distribution of H2 O (T ≤ 140K), the available maximum excess energy is still less than 26,390 cm−1 . While other mechanisms can lead to the formation of H2 at higher energy levels, their contribution to the overall H2 production is 1 + 1 1 + small (Liu et al. in preparation). Finally, the B 1 Σ+ u – X Σg and D Πu – X Σg transitions near Lyman-α have very small oscillator strengths, primarily because of unfavorable FranckCondon overlap. 5.4. Implication of hot OH observation in comets Based on the theoretical calculations of Crovisier (1989) and van Harrevelt & van Hemert (2000a,b), and experimental work of (Mordaunt et al. 1994; Hwang et al. 1999; Harich et al. – 15 – 2000, 2001a,b) discussed in sections 4 and 5.3, a large number of highly rotationally excited OH radicals in both X 2 Π and A 2 Σ states is expected to be produced in the comets by solar Lyman-α dissociation. For the X 2 Π state OH, Harich et al. (2000, 2001a,b) have shown that up to 75% (>94% in extreme cases) of available energy is deposited into rotational motion. The inferred rotational population distributions are highly inverted, and peak around Ni =4145 for vi =0-4 levels. OH radicals at N=49 and 50 levels, which are above the dissociation limit (35426 cm−1 ), but stablized by the centrifugal potential barrier, have also been detected (Yang 2005). The probable IR emissions from these ro-vibrational excited OH(X) radical are between 1-10µm. Some of these IR transitions can be detected by groundbased observations. Indeed, Bonev et al. (2006) has recently observed IR emission of OH from N as high as 16. Finally, as noted by Feldman et al. (2002), the O(1 D) atom, the co-product of H2 (X 1 Σ+ g ), 2 4 1 2 3 2 o 1 is also observed in the transition 2s 2p D - 2s 2p ( D )3s D at 1152.175 Å in both comets A2 and WM1. However, O(1 D) is not exclusively produced from dissociation of H2 O. Dissociation of OH, CO and CO2 by solar radiation also produces O(1 D) (Morgenthaler et al. 2001). 6. Conclusion In summary, the present work has assigned rotationally resolved molecular hydrogen transitions in comets C/2000 WM1 (LINEAR) and C/2001 A2 (LINEAR) observed by F USE. These transitions originate from highly excited X 1 Σ+ g ro-vibrational levels and are almost exclusively excited by the solar H Lyman-α line. Furthermore, all observed H2 emissions belong to the Lyman and Werner band systems. The initial levels of H2 observed in the F USE spectra confirm theoretical predictions that highly excited H2 is produced by photodissociation of H2 O with VUV solar radiation. We would like to thank Dr. Abgrall and Dr. Roueff for providing us with their complete 1 + 1 1 + 1 + Σu – X 1 Σ+ calculated H2 continuum profiles of the B 1 Σ+ u - X Σg , C Πu – X Σg , B g and D 1 Πu – X 1 Σ+ transitions. The research described in this paper was performed at g Space Environment Technologies, Inc., and John Hopkins University. The work at Space Environment Technologies, Inc. was supported by the Astronomy Program of the National Science Foundation (AST-0507810). – 16 – REFERENCES Abgrall, H., Roueff, E., Launay, F., Roncin, J. Y., & Subtil, J. L., 1993a, A&AS, 101, 273 Abgrall, H., Roueff, E., Launay, F., Roncin, J. Y., & Subtil, J. L., 1993b, A&AS, 101, 323 Abgrall, H., Roueff, E., Launay, F., Roncin, J. Y., & Subtil, J. L., 1993c, J. Mol. Spectrosc., 157, 512 Abgrall, H., Roueff, E., Launay, F. & Roncin, J. Y. 1994, Can. J. Phys. 72, 856 Abgrall H., Roueff, E., Liu, X., & Shemansky, D. E., 1997, ApJ, 481, 557 Abgrall H., Roueff, E., & Drira, I., 2000, A&AS, 141, 297 Anderson, P., & Schinke, R., 1987, in Molecular Photodissociation Dynamics, edited by M. N. R. Ashfold and J. E. Baggot (Royal Society of Chemistry), Chapter 3 Ashold, M. N. R., Bayley, J. M., & Dixon, R. N., 1984 Chem. Phys. , 84, 35 Bar-Nun, A., Dror, J., Kochavi, E., & Laufer, D, Phys. Rev. B, 35, 2427 Bemporad, A., Poletto, G., Raymond, J., C., Biesecker, D. A., Marsden, B., Lamy, P., Ko, Y.-K., & Uzzo, M., 2005, ApJ, 620, 523 Bensch, F., Bergin, E. A., Bockelee-Morvan, D., Melnick, G.J., & Biver, N., 2004, ApJ, 609, 1164 Bockelee-Morvan, D., Gautler, D., Lis, D.C., Young, K., Keene, J., Phillips, T., Owen, T., Crovisier, J., Bergin, E.A., Despois, D., & Wootten, A., 1998, Icarus, 133, 147 Bonev, B. P., Mumma, M. J., DiSanti, M. A., Dello Russo, N., Magee-Sauer, K., Ellis, R. S. & Stark, D. P., 2006, ApJ, 615, 1048 Brooke, T. Y., Weaver, H. A., Chin, G., Bockelee-Morvan, D., Kim, S. J., & Xu, L.-H., 2003, Icarus, 166, 167 Cernicharo, C., & Crovisier, J., 2005, Space Sci. Rev., 119, 29 Chan, W. F., Cooper, G., & Brion, C. E., 1993, Chem. Phys. , 178, 387 Chen, B.-M., Chew, E. P., Liu, C.-P., Bahou, M., Lee, Y.-P., Yung, Y. L., & Gerstell, M. F., 1999, Geophys. Res. Lett., 26, 3657 – 17 – Chen, B.-M., Chung, C.-Y., Bahou, M., Lee, Y.-P., Lee, L. C., van Harrevelt, R. & van Hemert, M. C., 2004, J. Chem. Phys., 120, 224 Chiu, K., Neufeld, D.A., Bergin, E.A., Melnick, G.J., Patten, B.M., Wang, Z. & BockeleeMorvan, D., 2001, Icarus, 154, 345 Christiansen, O., Nymand, T. M., & Mikkelsen, K. V., 2000, J. Chem. Phys., 113, 8101 Cochran, A. L., & Cochran, W. D., 2001, Icarus, 154, 381 Colom, P, Gerard, E., Crovisier, J, Bockelee-Morvan, D., Biver, N., & Rauer, H., 1999, Earth Moon & Planets, 78, 37 Combi, M. R., Brown, M. E., Feldman, P. D., Keller, H. U., Meier, R. R., & Smyth, W. H., 1998, ApJ, 494, 816 Combi, M, 2002, Earth, Moon and Plants, 98, 73 Crim, F. F., 1993, Ann. Rev. Phys. Chem., 44, 397 Crovisier, J., 1989, A&A, 213, 459 Crovisier, J, Colom, P, Gerard, E., Bockelee-Morvan, D., & Bourgois, G., 2002, å, 393, 1053 Crovisier, J., Bockelee-Morvan, D., Colom, P., Biver, N., Despois, D., & Lis, D. C., 2004, å, 418, 1141 Dabrowski, I., 1984, Can. J. Phys., 62, 1639 Edery. F. & Kanaev, A., 2003, Eur. Phys. J. D 23, 257 Ehrhardt, H., Langhans, D. L., Linder, F., & Taylor, H. S., 1968, Phys. Rev., A 2, 222 Eidelsberg, M., Benayoun, J. J., Viala, Y., & Rostas F., 1991, A&AS, 90, 231 Engel, V., Staemmler, V., Van der Wal, R. L., Crim, F. F., Sension, R. J., Hudson, B., Anderson, P., Hennig, S., Weide, K., & Schnike, 1992, J. Chem. Phys., 3201 Farmanara, P., Steinkellner, O., Wick, M. T., Wittmann, M., Korn, G., Stert, V., & W. Radloff, 1999, J. Chem. Phys., 111, 6264 Feldman, P. D., Weaver, H. A., & Burgh, E. B, 2002, ApJ, 576, L91 Feldman, P. D., 2005, Phys. Scr., T119, 7 – 18 – Feldman, P. D. McCandliss, S. R., & Weaver, H. A., 2006, BAAS, 38, 517 (abstract 20.06) Fullion, J. H., van Harrevelt, R. , Ruiz, J., Castillejo, M., Zanganeh, A. H., Lemaire, J. L., van Hemert, M. C., & Rostas, F., 2001, J. Phys. Chem., A 105, 11414 Hallett, J. T., Shemansky, D. E. & Liu, X., 2005, ApJ, 624 Harich, S. A., Hwang, D. W. H., Yang, X., Lin, J. J., Yang, X. & R. N. Dixon, 2000, J. Chem. Phys., 113, 10073 Harich, S. A., Yang, X., Hwang, D. W. H., Lin, J. J., Yang, X. & R. N. Dixon, 2001a, J. Chem. Phys., 114, 7830 Harich, S. A., Yang, X., Yang, X. & R. N. Dixon, 2001b, Phys. Rev. Lett., 87, 253201 Herring-Captain, J., Grieves, G.A., Alexandrov, A., Sieger, M.T., Chen, H., & Orlando, T.M., 2005, Phys. Rev. B, 72, 35431 Hirst, D. M., & Child, M. S., 1992, Mol. Phys. 77, 463 Hwang, D. W., Yang, X. F., Harich, S., Lin, J. J., & Yang, X., 2000, J. Chem. Phys., 110, 4123 Huebner, W. F., & Keady, J. J., 1992, Astrophys. Space Sci., 195, 1 Huestis, D. L., & Slanger, T. G., 2006, BAAS, 38, 609 (abstract 62.20) Jonin, C., Liu, X., Ajello, J. M., James, G. K., & Abgrall, H., 2000, ApJS, 129, 247 Kharchenko, V., & Dalgarno, A., 2001, ApJ, L99 Kimmel, G. A., Tonkyn, R. G., & Orlando, T. M., 1995, Nucl. Instru. Methd. Phys. Res. B1010, 179 Kimmel, G. A., Orlando, T.M., Vezina, C. & Sanche, L. 1994, J. Chem. Phys., 101, 3282 Kuge, H.-H., & Kleinermanns, K., 1989, J. Chem. Phys., 90, 46 Laufer, D., Kochavi, E., & Bar-Nun, A., 1987, Phys. Rev. B, 36, 9219 Lecacheux, A., Biver, N., Crovisier, J., Bockelee-Morvan, D., Baron, P., Booth, R.S., Encrenaz, P., Floren, H.-G., Frisk, U., Hjalmarson, A., Kwok, S., Mattila, K., Nordh, L., Olberg, M., Olofsson, A.O.H., Rickman, H., Sandqvist, A., von Scheele, F., Serra, G., Torchinsky, S., Volk, K., & Winnberg, A., 2003, å, 402, L55 – 19 – Lee, L. C., & Suto, M., 1986, Chem. Phys. , 110, 161 Lemaire, P., Emerich, C., Curdt, W., Schühle, U., & Wilhelm, K., 1998, å, 334, 1095 Liu, X., Ahmed, S. M., Multari, R. A., James, G. K., & Ajello, J. M., 1995, ApJS, 101, 375 Liu, W. & Dalgarno, A, 1996, ApJ, 462, 506 Liu, X., Shemansky, D. E., & Weaver, H. A., ApJ, (in preparation) Meier, R., Owen, T.C., Matthews, H.E., Jewitt, D.C., Bockelee-Morvan, D., Biver, N., Crovisier, J., & Gautier, D., 1998, Science, 279, 842 Mordaunt, D. H., Ashfold, M. N. R., & Dixon, R. N. 1994, J. Chem. Phys., 100, 7360 Morgenthaler, J. P., Harris, W. M., Scherb, F., Anderson, C. M., Oliversen, R. J., Doane, N. E., Combi, M. R., Marconi, M. L., & Smyth, W. H., 2001, ApJ, 563, 451 Mumma, M. J. et al.,2001, ApJ, 546, 1183 Mumma, M. J., DiSanti, M. A., Russo, N. D., Magee-Sauer, K., Gibb, E. & Novak, R., 2003, Adv. Space. Res., 31, 2563 Mumma, M. J., DiSanti, M.A., Magee-Sauer, K., Bonev, B.P., Villanueva, G.L., Kawakita, H., Russo, N.D., Gibb, E.L., Blake, G.A., Lyke, J.E., Campbell, R.D., Aycock, J., Conrad, A., & Hill, G.M., 2005 Science, 310, 270 Parkinson, W. H., & Yoshino, K., 2003, Chem. Phys. , 294, 31 Ralchenko, Yu., Jou, F.-C., Kelleher, D.E., Kramida, A.E., Musgrove, A., Reader, J., Wiese, W.L., & Olsen, K., 2005. NIST Atomic Spectra Database (version 3.0.2), http://physics.nist.gov/asd3. National Institute of Standards and Technology, Gaithersburg, MD Roncin, J. Y., & Launay, F., 1994, Atals of the Vaccum Ultraviolet Emission Spectrum of Molecular Hydrogen, J. Phys. Chem. Ref. Data, Monograph No 4. Ruscic, B. et al. 2002, J. Phys. Chem. A 106, 2727 Ruscic, B. et al. 2005, J. Phys. Chem. Ref. Data, 34, 573 Dello Russo, N. , Mumma, M. J., DiSanti, M. A., & Magee-Sauer, K., 2002, J. Geophys. Res., 107, 5059 – 20 – Dello Russo, N., Disanti, M. A., Magee-Sauer, K., Gibb, E. L., Mumma, M. J., Barber, R. J., & Tennyson, J. 2004, Icarus, 168, 186 Dello Russo, N., Bonev, B. P., Disanti, M. A., Mumma, M. J., Gibb, E. L., Magee-Sauer, K., Barber, R. J., & Tennyson, J., 2005, ApJ, 621, 537 Schatz, G. C., 1985, J. Chem. Phys., 83, 5677 Schröder, T., Schinke, Ehara, M. & Yamashita, K., 1998, J. Chem. Phys., 109, 6641 Schloerb, F. P., De Veries, C. H., Lovell, A. J., Irvine, W. H., Senay, M. & Wootten, H. A., 1999, Earth moon & Planet, 78, 45 Slanger, T. G., & Black, G. 1982, J. Chem. Phys., 77, 2432 Steinkellner, O., Noack, F., Ritze, H.-H., Radlof, W., & Hertel, I. V., 2004, J. Chem. Phys., 121, 1765 van Harrevelt, R. & van Hemert, M. C., 2000a, J. Chem. Phys., 112, 5777 van Harrevelt, R. & van Hemert, M. C., 2000b, J. Chem. Phys., 112, 5787 van Harrevelt, R. & van Hemert, M. C., 2001, J. Chem. Phys., 114, 9453 van Harrevelt, R., van Hemert, M.C., & Schatz, G.C., 2001, J. Phys. Chem. A 105, 11480 van Harrevelt, R. & van Hemert, M. C., 2003, Chem. Phys. Lett. , 370, 706 Wang, H.-t., Felps, W. S., & McGlynn, S. P., 1977, J. Chem. Phys., 67, 2614 Weaver, H. A., Chin, G., Bockelee-Morvan, D., Crovisier, J., Brooke, T. Y., Cruikshank, D. P., Geballe, T. R., Kim, S. J., & Meier, R., 1999, Icarus, 142, 482 Weaver, H. A., Feldman, P. D., Combi, M. R., Kranopolsky, V., Lisse. C. M. & Shemansky D. E., 2002, ApJ, 576, L95 Wolniewicz, L., Simbotin, I., & Dalgarno, A., 1998, ApJS, 115, 293 Weide, K., & Schinke, R., 1989, J. Chem. Phys., 90, 7150 Yang, X., 2005, Int. Rev. Phys. Chem., 24, 37 Yoshino, K., Esmond, J. R., Parkinson, W. H., Ito, K., & Matsui, T., 1996, Chem. Phys. , 211, 387 This preprint was prepared with the AAS LATEX macros v5.2. – 21 – Table 1. Summary of F USE Cometary Observations Comet Date & Timea Exp. Timeb Exp. Timec r (AU)d ṙ (km/s)d ∆ (AU)e ˙ (km/s)e ∆ C/A2 C/WM1 Jul. 12.58-12.89 Dec. 7.37-10.01 16,485 36,557 9,530 34,577 1.20 1.12 22.8 -28.3 0.30 0.34 14.6 13∼14 a Universal time year 2001. b Total exposure times in unit of s. c Total exposure times in unit of s when F U SE was in Earth shadow. d e r and ṙ denote comet’s heliocentric distance and heliocentric radial velocity, respectively. ˙ represent comet’s geocentric distance and geocentric radial velocity, respectively. ∆ and ∆ 920.995 921.079 923.175 923.208 926.214 926.237 929.523 929.995 930.745 930.758 936.617 937.386 937.808 937.832 938.880 939.331 939.872 942.264 945.269 945.556 946.809 948.633 949.755 949.758 950.889 960.722 960.746 969.482 970.365 971.154 971.745 971.749 972.550 972.554 973.229 Obsd. Linea 0.322 0.242 0.331 0.274 0.285 0.317 0.99 0.175 0.306 0.277 0.367 0.149 0.295 0.269 0.741 0.241 0.276 2.352 1.42 0.202 0.338 0.268 0.295 0.289 0.979 0.314 0.273 0.254 0.689 0.255 0.294 0.398 0.284 0.284 0.234 FWHMa A2 WM1 WM1 A2 A2 WM1 WM1 WM1 A2 WM1 WM1 WM1 A2 WM1 WM1 WM1 WM1 WM1 WM1 WM1 WM1 WM1 WM1 A2 WM1 A2 WM1 WM1 WM1 WM1 WM1 A2 A2 WM1 WM1 Comet H I: 1s 2 S - 10p 2 P (920.963) H I: 1s 2 S - 10p 2 P (920.963) H I: 1s 2 S - 9p 2 P (923.150) H I: 1s 2 S - 9p 2 P (923.150) H I: 1s 2 S - 8p 2 P (926.226) H I: 1s 2 S - 8p 2 P (926.226) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )7d 3 D (929.517) 2(22, 1)R B (929.950) H I: 1s 2 S - 7p 2 P (930.748) H I: 1s 2 S - 7p 2 P (930.748) 4(22, 1)P B (936.688) 7(11,2)P C (937.223) ? H I: 1s 2 S - 6p 2 P (937.803) H I: 1s 2 S - 6p 2 P (937.803) 7(18, 0)R B (938.874) ? O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )7s 3 S (939.235) O I: 2s2 2p4 3 P0 - 2s2 2p3 (4 So )7s 3 S (939.841) 7( 7, 1)P C (942.272) [strongest of (7,vi )] C I: 2s2 2p2 3 P0,1 - 2s2p3 3 S1 (945.191; 945.338) C I: 2s2 2p2 3 P2 - 2s2p3 3 S1 (945.579) 12( 6, 0)Q C (946.524) ? O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )5d 3 D (948.686) H I: 1s 2 S - 5p 2 P (949.743) H I: 1s 2 S - 5p 2 P (949.743) 9(24, 1)R B (950.944) ? [req. pumping 9(24,9)R B] 7(15,0)R B (960.699) 7(15,0)R B (960.699) 11(24, 1)P B (969.558) ? [req. pumping 9(24,9)R B] CO : 4pσ 1 Σ+ (0) - X 1 Σ+ (0) (970.359) 13(21, 0)P B (971.235) ? O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )4d 3 D (971.738) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )4d 3 D (971.738) H I: 1s 2 S - 4p 2 P (972.537) H I: 1s 2 S - 4p 2 P (972.537) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )4d 3 D (972.234) Primary Assignmentb,c O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )6s 3 S (950.885) 9(15,0)P B at 977.742 very weak 9(15,0)P B at 977.742 very weak 13(27, 1)R B (969.349)? 13( 5, 0)P C (969.967),12(19,0)R B (970.360) R11@949.186 not seen 4(22, 2)P B (971.906) ? 4(22, 2)P B (971.906) ? stronger 12(6,1)Q C line @ 981.191 not seen CO : 3sσ 1 Π(2) - X 1 Σ+ (0) (941.169) ? (22,1) band strongest among all (22,vi )B-X band O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )7d 3 D (930.886) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )7d 3 D (930.886) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )6d 3 D (936.629) Require 7(11,12)P C @1214.977 pumped by Lyman-α O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )7s 3 S (937.841) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )7s 3 S (937.841) 8( 5, 0)P C (938.703) ? Secondary Assignmentb,c Table 2. Observed Transitions and Spectral Assignments – 22 – 973.287 973.958 983.921 984.051 984.651 988.747 988.750 990.198 990.220 991.003 991.025 997.322 998.345 998.352 999.094 999.115 1013.819 1013.820 1016.587 1016.592 1017.278 1017.323 1018.065 1018.089 1025.685 1025.708 1027.404 1027.414 1028.057 1028.117 1028.777d 1031.898e 1033.901 1033.952 1036.933 Obsd. Linea 0.219 0.201 0.784 0.32 0.161 0.345 0.37 0.312 0.274 0.384 0.308 0.28 0.275 0.295 0.255 0.331 0.32 0.306 0.429 0.373 0.295 0.331 0.305 0.24 0.349 0.356 0.317 0.314 0.328 0.31 0.683 0.261 0.251 0.205 0.211 FWHMa A2 WM1 WM1 A2 WM1 WM1 A2 A2 WM1 WM1 A2 WM1 WM1 A2 A2 WM1 WM1 A2 A2 WM1 A2 WM1 WM1 A2 A2 WM1 A2 WM1 A2 WM1 A2 WM1 WM1 A2 WM1 Comet O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )4d 3 D (972.234) O I: 2s2 2p4 3 P0 - 2s2 2p3 (4 So )4d 3 D (973.885) 11( 3, 0)Q C (984.050) 11( 3, 0)Q C (984.050) 11(21, 1)R B (984.655) ? O I: 2s2 2p4 3 P2 - 2s2 2p3 (2 Do )3s 3 D (988.773) O I: 2s2 2p4 3 P2 - 2s2 2p3 (2 Do )3s 3 D (988.773) O I: 2s2 2p4 3 P1 - 2s2 2p3 (2 Do )3s 3 D (990.204) O I: 2s2 2p4 3 P1 - 2s2 2p3 (2 Do )3s 3 D (990.204) 10( 2, 0)R C (991.056) 10( 2, 0)R C (991.056) 11(19, 1)R B (997.451) ? 6( 1, 0)Q C (998.332) 6( 1, 0)Q C (998.332) 7(15,1)R B (999.090) 7(15,1)R B (999.090) 12( 2, 0)P C (1013.842) 12( 2, 0)P C (1013.842) 9(15,1)P B (1016.568) 9(15,1)P B (1016.568) 13(14,0)R B (1017.302) 13(14,0)R B (1017.302) 9(10, 0)R B (1018.093) 9(10, 0)R B (1018.093) H I: 1s 2 S - 3p 2 P (1025.723) H I: 1s 2 S - 3p 2 P (1025.723) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )3d 3 D (1027.431) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )3d 3 D (1027.431) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )3d 3 D (1028.157) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )3d 3 D (1028.157) 17(19,0)P B (1028.868) 3(1,1)Q C, O VI: 2s 2 S1/2 - 2p 2 P3/2 (1031.912) 8( 0, 0)P C (1033.951) 8( 0, 0)P C (1033.951) 15(19, 1)R B (1037.066) Primary Assignmentb,c Secondary Assignmentb,c 0, 0, 7, 7, 0)R 0)R 0)R 0)R C C B B (1016.742) (1016.742) (1017.423) ? (1017.423) ? Weaker P17 @1065.848 not seen 5( 7, 0)P B (1028.248) ? 5( 7, 0)P B (1028.248) ? 3(5,3)QC (1028.777) [(5,0) band aslo present but (5,1) and (5,5) bands absent] Note: O VI 2 S1/2 - 2 P1/2 @ 1037.613 not seen O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )3d 3 D (1025.762) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )3d 3 D (1025.762) 6( 6( 3( 3( O I: 2s2 2p4 3 P0 - 2s2 2p3 (2 Do )3s 3 D (990.801) O I: 2s2 2p4 3 P0 - 2s2 2p3 (2 Do )3s 3 D (990.801) stronger 13(19, 1)P B @ 1020.496 not seen 6(10,0)R B (998.492) 6(10,0)R B (998.492) 11(21, 0)R B @ 949.186 not seen Table 2—Continued – 23 – 1037.065 1038.081 1038.123 1039.197 1039.199 1040.254 1040.276 1040.823 1040.858 1041.697 1042.737 1042.819 1043.240 1043.468 1043.762 1044.832 1046.061 1053.679 1053.700 1055.572 1055.948 1056.039 1060.863 1060.900 1064.269 1066.567 1070.362 1070.388 1071.587 1071.594 1075.576 1076.065 1077.781 1077.905 1087.960 Obsd. Linea 0.299 0.5 0.363 0.326 0.324 0.288 0.334 0.535 0.358 0.261 0.135 0.372 0.348 0.185 0.286 0.483 0.152 0.399 0.422 0.243 0.283 0.809 0.403 0.398 0.155 0.212 0.368 0.418 0.231 0.429 0.24 0.471 0.295 0.57 0.945 FWHMa A2 A2 WM1 WM1 A2 WM1 A2 WM1 A2 A2 WM1 A2 WM1 A2 WM1 WM1 WM1 WM1 A2 WM1 WM1 A2 A2 WM1 WM1 A2 A2 WM1 A2 WM1 A2 A2 WM1 A2 A2 Comet 15(19, 1)R B (1037.066) 7(15,2) R B (1038.176) 7(15,2) R B (1038.176) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )4s 3 S (1039.230) O I: 2s2 2p4 3 P2 - 2s2 2p3 (4 So )4s 3 S (1039.230) 6( 1, 1)Q C (1040.284) 6( 1, 1)Q C (1040.284) 11(10, 0)P B (1040.661) 11(10, 0)P B (1040.661) O I: 2s2 2p4 3 P0 - 2s2 2p3 (4 So )4s 3 S (1041.688) ?? ?? 13(21, 2)P B (1043.260) ? 12(0,0)R C (1043.555) 12(0,0)R C (1043.555) 12(10, 0)R B (1044.573) 17(21,1)R B (1046.108)? 12( 2, 1)P C (1053.720) 12( 2, 1)P C (1053.720) 4( 2, 2)P C (1055.339) ? 9(15,2)P B (1056.037) 9(15,2)P B (1056.037) 11( 3, 2)Q C (1060.903) 11( 3, 2)Q C (1060.903) 8( 4, 3)Q C (1064.137) ? 18(15, 0)R B (1066.622) 10( 2, 2)R C (1070.367) 10( 2, 2)R C (1070.367) 14(0,0)P C (1071.532) 14(0,0)P C (1071.532) ? CO : 3pπ 1 Π(0) - X 1 Σ+ (0) (1076.079) 7(15,3) R B (1077.783), 9(0,1)Q C (1077.838) 8( 0, 1)P C (1078.047) CO : 3pσ 1 Σ+ (0) - X 1 Σ+ (0) (1087.913) Primary Assignmentb,c Table 2—Continued 12( 7,0)R B (1077.427) ? 7(15,3) R B (1077.783) 20( 2, 0)Q C (1070.562) 20( 2, 0)Q C (1070.562) 1(6,1)P B (1071.618) ← weak 1(6,1)P B (1071.618) ← weak stronger Q(8) line of (4,0), (4,5) bands not seen 14( 0, 0)R C (1056.044) 14( 0, 0)R C (1056.044) 15(14,0)P B (1044.949) 4(22, 4)P B (1043.408) ? O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )4s 3 S (1040.942) O I: 2s2 2p4 3 P1 - 2s2 2p3 (4 So )4s 3 S (1040.942) Weaker P17 @1065.848 not seen Secondary Assignmentb,c – 24 – 1087.966 1089.275 1090.414 1094.135 1094.138 1096.593 1102.228 1103.219 1103.233 1104.976 1106.865 1109.288 1110.708 1110.728 1114.440 1114.534 1117.851 1118.103 1118.546 1122.636 1123.126 1123.215 1126.864 1126.870 1128.967 1129.088 1134.941 1134.965 1136.006 1136.078 1137.250 1138.860 1138.932 1139.360 1139.387 Obsd. Linea 1.02 0.83 0.154 0.31 0.323 0.31 0.394 0.217 0.397 0.231 0.576 0.242 0.239 0.444 0.289 0.348 0.387 1.051 0.193 2.515 0.255 0.171 0.302 0.341 0.919 0.894 0.487 0.43 0.672 0.27 0.267 0.321 0.321 0.223 0.218 FWHMa WM1 WM1 A2 A2 WM1 A2 A2 A2 WM1 A2 A2 A2 A2 WM1 A2 WM1 A2 WM1 A2 WM1 A2 WM1 A2 WM1 WM1 A2 A2 WM1 A2 WM1 WM1 A2 WM1 A2 WM1 Comet CO : 3pσ 1 Σ+ (0) - X 1 Σ+ (0) (1087.913) 12(6,0)R B (1089.544) ? 5(13, 3)P B (1090.257) ? 12( 2, 2)P C (1094.138) 12( 2, 2)P C (1094.138) 13(14,2)R B (1096.600) 8(4,4)Q C (1102.503) ? 9(10, 2)R B (1103.266) 9(10, 2)R B (1103.266) 6( 0, 2)R C (1104.888) 22( 1, 0)Q C (1106.947) 5( 1, 0)P B (1109.313) 10( 2, 3)R C (1110.750) 10( 2, 3)R C (1110.750) 14(0,1)P C (1114.507) 14(0,1)P C (1114.507) 7(15,4)R B (1117.697) 1(6,2)P B (1118.508) 1(6,2)P B (1118.508) 12(7,1)R B (1122.576), 9(0,2)Q C(1122.312) 8( 0, 2)P C (1123.141) 8( 0, 2)P C (1123.141) 6( 1, 3)Q C (1126.854) 6( 1, 3)Q C (1126.854) 12(0, 2)R C (1128.824) 12(0, 2)R C (1128.824) 12( 2, 3)P C (1134.861) 12( 2, 3)P C (1134.861) 9(15, 4)P B (1136.079) 9(15, 4)P B (1136.079) 22( 0,0)P C (1137.224) 11( 3, 4)Q C (1138.882) 11( 3, 4)Q C (1138.882) 20( 0, 1)R C (1139.333) 20( 0, 1)R C (1139.333) Primary Assignmentb,c Secondary Assignmentb,c 17(19, 3)P B (1139.546) ? 17(19, 3)P B (1139.546) ? 11(10, 2)P B (1126.999) 11(10, 2)P B (1126.999) C I : 2s2 2p2 3 P - 2s2 2p2 Po 7d 3 D (1128.817 -1129.196) C I : 2s2 2p2 3 P - 2s2 2p2 Po 7d 3 D (1128.817 -1129.196) N I: 2s2 2p3 4 S - 2s2 2p4 4 P (1134.165, 1134.415, 1134.980) N I: 2s2 2p3 4 S - 2s2 2p4 4 P (1134.165, 1134.415, 1134.980) R7 @ 1117.697 weak R7 @ 1117.697 weak C I : 2s2 2p2 3 P - 2s2 2p(2 Po )8d 3 D (1122.004 -1122.985) 6( 1, 0)R B (1109.860) ? 15(19, 3)R B (1110.488)? 15(19, 3)R B (1110.488)? 2( 2, 3)R C(1089.188) ? Cl I 1090.271 18(0,0)QC (1094.273) ? 18(0,0)QC (1094.273) ? 3( 1, 0)R B (1096.725) stronger Q(8) line of (4,0), (4,5) bands not seen Table 2—Continued – 25 – 0.142 0.325 0.159 0.346 0.434 0.368 1.787 1.195 0.264 0.309 0.604 0.412 0.367 0.469 0.439 0.276 0.286 0.314 0.35 0.275 0.328 0.582 0.451 0.275 0.213 0.455 0.358 FWHMa WM1 A2 A2 WM1 WM1 A2 WM1 A2 WM1 A2 WM1 A2 WM1 A2 WM1 WM1 A2 WM1 WM1 A2 WM1 A2 WM1 A2 WM1 A2 WM1 Comet C I : 2s2 2p2 3 P - 2s2 2p2 Po 6d 3 D (1139.514 -1140.005) C I : 2s2 2p2 3 P - 2s2 2p2 Po 6d 3 D (1139.514 -1140.005) 22( 1, 1)Q C (1144.192) 14( 4, 0)P B (1147.705) ? 3( 1, 1)R B (1148.703) 3( 1, 1)R B (1148.703) CO : 3sσ 1 Σ+ (0) - X 1 Σ+ (0) (1150.534) CO : 3sσ 1 Σ+ (0) - X 1 Σ+ (0) (1150.534) O I 2s2 2p4 1 D - 2s2 2p3 (2 Do )3s 1 D (1152.152) O I 2s2 2p4 1 D - 2s2 2p3 (2 Do )3s 1 D (1152.152) 7(15, 5)R B (1157.628) 14(0,2)P C (1158.032) 14(10,2)P B (1159.289) 5( 1, 1)P B (1161.816) 5( 1, 1)P B (1161.816) 3(1,4)Q C, 2(0,1)R B (1163.645) ? 1(6,3)P B (1166.764) 1(6,3)P B (1166.764) 12( 7, 2)R B (1168.564) 6( 1, 4)Q C (1171.077) 6( 1, 4)Q C (1171.077) 12( 2, 4)P C (1175.588) 12( 2, 4)P C (1175.588) 22( 0, 1)P C (1176.570) 22( 0, 1)P C (1176.570) 20( 0, 2)R C (1178.193) 20( 0, 2)R C (1178.193) Primary Assignmentb,c 2(2,5)P C (1178.292) ? 2(2,5)P C (1178.292) ? 15(3,4)P C (1168.534) ? 11(10, 3)P B (1171.084) 11(10, 3)P B (1171.084) 18(12, 2)R B (1175.741) ? 18(12, 2)R B (1175.741) ? 14(0,2)P C (1158.032) C I : 2s2 2p2 3 P - 2s2 2p2 Po 5d 3 D (1157.769 -1158.492) R12 @1130.016 weak 6(1,1)R B (1161.953) 6(1,1)R B (1161.953) 4(4,6)R C (1163.786) ? 6( 0, 3)R C (1150.365), 13(21,5)P B (1150.811) 6( 0, 3)R C (1150.365), 13(21,5)P B (1150.811) 20(2,2)Q C (1144.251) Secondary Assignmentb,c c Transition followed by single question mark (?) means that the assignment is possible but not definitive. Lines with double question mark (??) b The spectral carrier is H unless specified otherwise. Transitions for H are labeled as J (v ,v )∆J β, where i and j refer to the lower and upper states, 2 2 i j i β is electronic designation of excited state, and ∆J=-1, 0 and +1 correspond to P, Q, and R transitions, respectively. Numbers in parentheses are the model wavelength in Å. a The observed line and FWHM refer to the center wavelength and the full width at half maximum, respectively, for the best-fit gaussian to the indicated spectral feature. Both units are Å 1139.906 1139.928 1144.222 1147.714 1148.669 1148.683 1150.408 1150.573 1152.175 1152.203 1157.937 1158.035 1159.177 1161.809 1161.884 1163.786e 1166.690 1166.871 1168.635 1171.073 1171.082 1175.603 1175.629 1176.484 1176.576 1178.188 1178.217 Obsd. Linea Table 2—Continued . – 26 – 3(1,1)Q C-X (1031.865) could be resonantly pumped by O VI 2 S1/2 - 2p 2 P3/2 (1031.912) solar line. Even though the strongest emission, 3(1,4)Q C-X (1163.805) can be identified in WM1, the absence of the corresponding features for the (1,0) and (1,3) bands at 989.729 and 1119.079 Å respectively, raised some questions. Please refer to section 5 for discussion. e The d A small contribution from the 1(1,1)Q C (1028.989) emission is possible, arising from pumping of the 1(1,5)Q C line (1206.639) by Solar Si III lines whose rest and Doppler shifted (for comet A2) wavelengths are 1206.510 and 1206.602 Å respectively. This assignment is not listed in the Table because corresponding and stronger lines for the (1,0), (1,3) and (1,4) bands are not identified indicates that no reasonable assignment of H2 or other species is known to the authors at the present time. – 27 – – 28 – Fig. 1.— Composite F USE spectrum of comet C/2001 A2 (LINEAR) labeled with the primary assignments. Only darkside data (9,535 s) are included. The 30 × 30 entrance aperture was used and exposures were summed over five contiguous orbits. A zero line is added as a noise level reference. Only primary spectral assignments are labeled. See text for notation and Table 2 for detailed assignments. Fig. 2.— Composite F USE spectrum of comet C/2000 WM1 (LINEAR) with primary spectral assignments. Dark side exposures only. The 30 × 30 entrance aperture was used and exposures were obtained over contiguous orbits. See Figure 1. – 29 – C/2001 A2 (LINEAR) 1.5 H I:1s-5p H I:1s-6p H I:1s-9p H I: 1s -10p Brightness (R/Å) 2.5 H I:1s-7p H I:1s-8p 3.5 0.5 -0.5 920 930 940 Wavelength (Å) f1a.eps 950 960 1.5 -0.5 960 970 2.5 980 Wavelength (Å) f1b.eps 7(15,1)R B O I: (988.773) H I: 1s-4p 990 6(1,0)Q C 3.5 O I: (990.204) 10(2,0)R C 11(3,0)Q C O I: (972.234) O I: (971.738) 7(15,0)R B Brightness (R/Å) – 30 – C/2001 A2 (LINEAR) 0.5 1000 1.5 0.5 -0.5 1000 1010 1020 Wavelength (Å) f1c.eps 1030 15(19,1)R B 7(15,2) R B O I: (1039.230) 8(0, 0)P C O I: (1027.431) O I: (1028.157) 17(19,0)P B 9(15,1)P B 13(14,0)R B 9(10, 0)R B 2.5 H I: 1s-3p 12(2,0)P C Brightness (R/Å) – 31 – C/2001 A2 (LINEAR) 3.5 1040 1.5 0.5 -0.5 1040 1050 1060 Wavelength (Å) f1d.eps ? 2.5 1070 8(0,1)P C CO : E(0)-X(0) (1076.079) 10( 2, 2)R C 14(0,0)P C 18(15, 0)R B 11( 3, 2)Q C 9(15,2)P B 12(2,1)P C 11(10, 0)P B O I: (1041.688) ? 12(0,0)R C 6( 1, 1)Q C Brightness (R/Å) – 32 – C/2001 A2 (LINEAR) 3.5 1080 0.5 -0.5 1080 1090 1100 Wavelength (Å) f1e.eps 1110 1(1,3)Q C (by Si III) 7(15,4)R B 1(6,2)P B 14(0,1)P C 5( 1, 0)P B 10( 2, 3)R C 22( 1, 0)Q C 6( 0, 2)R C 8(4,4)Q C ? 9(10,2)R B 12( 2, 2)P C 1.5 13(14,2)R B 5(13,3)P B? 2.5 CO : C(0)-X(0) (1087.913) Brightness (R/Å) – 33 – C/2001 A2 (LINEAR) 3.5 1120 0.5 -0.5 1120 1.5 1130 f1f.eps 1140 Wavelength (Å) 11( 3, 4)Q C C I: (1139.514 -1140.005) 1150 14(0,2)P C O I: (1152.152) CO : B(0)-X(0) (1150.534) 3( 1, 1)R B 22( 1, 1)Q C 20( 0, 1)R C 12( 2, 3)P C 9(15, 4)P B 6( 1, 3)Q C 2.5 12(0, 2)R C 8( 0, 2)P C Brightness (R/Å) – 34 – C/2001 A2 (LINEAR) 3.5 1160 Brightness (R/Å) 1.5 -0.5 1160 1170 20( 0, 2)R C 12( 2, 4)P C 22( 0, 1)P C 6( 1, 4)Q C 1(6,3)P B 5( 1, 1)P B – 35 – C/2001 A2 (LINEAR) 3.5 2.5 0.5 1180 Wavelength (Å) f1g.eps 1190 1200 -0.5 920 H I: 1s - 8p 1.5 930 H I: 1s - 7p 940 Wavelength (Å) f2a.eps 9(24, 1)R B ? O I: (948.686) H I: 1s - 5p C I: (945.191; 945.338) C I: (945.579) 12( 6, 0)Q C ? 7( 7, 1)P C H I: 1s - 6p 7(18, 0)R B O I: (939.235) O I: (939.841) 4(22, 1)P B 7(11,2)P C O I: (929.517)2(22, 1)R B 0.5 H I: 1s - 9p H I: 1s - 10p Brightness (R/Å) – 36 – C/2000 WM1 (LINEAR) 2.5 950 960 Brightness (R/Å) 1.5 0.5 -0.5 960 970 980 Wavelength (Å) f2b.eps 990 O I: (988.773) O I: (990.204) 10( 2, 0)R C 11(19, 1)R B ? 6( 1, 0)Q C 7(15,1)R B 11( 3, 0)Q C 11(21, 1)R B ? 11(24, 1)P B ? CO : K(0)-X(0) (970.359) 13(21, 0)P B ? O I: (971.738) H I: 1s - 4p O I: (972.234) O I: (973.885) 7(15,0)R B – 37 – C/2000 WM1 (LINEAR) 2.5 1000 0.5 -0.5 1000 1010 1.5 1020 Wavelength (Å) f2c.eps 1030 15(19, 1)R B 7(15,2) R B O I: (1039.230) 8( 0, 0)P C O VI: 2s - 2p (1031.912) ? O I: (1028.157) 9(15,1)P B 13(14,0)R B 9(10, 0)R B 12( 2, 0)P C Brightness (R/Å) O I: (1027.431) H I: 1s - 3p – 38 – C/2000 WM1 (LINEAR) 2.5 1040 Brightness (R/Å) 0.5 -0.5 1040 1050 12(0,0)R C f2d.eps 1060 Wavelength (Å) 1070 7(15,3) R B, 9(0,1)Q C 10( 2, 2)R C 14(0,0)P C 8( 4, 3)Q C ? 11( 3, 2)Q C 9(15,2)P B 4( 2, 2)P C ? 12( 2, 1)P C 12(10, 0)R B 17(21,1)R B ?? 13(21, 2)P B 6( 1, 1)Q C 1.5 11(10, 0)P B – 39 – C/2000 WM1 (LINEAR) 2.5 1080 Brightness (R/Å) 1.5 0.5 -0.5 1080 1090 1100 Wavelength (Å) f2e.eps 1110 1(6,2)P B 14(0,1)P C 10( 2, 3)R C 9(10, 2)R B 12( 2, 2)P C CO : C(0) - X(0) (1087.913) 12(6,0)R B ? – 40 – C/2000 WM1 (LINEAR) 2.5 1120 Brightness (R/Å) 0.5 -0.5 1120 1130 1140 Wavelength (Å) f2f.eps 11( 3, 4)Q C C I : (1139.514 -1140.005) 1150 7(15, 5)R B 14(10,2)P B O I (1152.152) CO : B(0) - X(0) (1150.534) 14( 4, 0)P B ? 3( 1, 1)R B 20( 0, 1)R C 12( 2, 3)P C 9(15, 4)P B 22( 0,0)P C 12(0, 2)R C 6( 1, 3)Q C 12(7,1)R B, 9(0,2)Q C 1.5 8( 0, 2)P C – 41 – C/2000 WM1 (LINEAR) 2.5 1160 Brightness (R/Å) 0.5 -0.5 1160 1170 20( 0, 2)R C 12( 2, 4)P C 22( 0, 1)P C 6( 1, 4)Q C 12( 7, 2)R B 1(6,3)P B 3(1,4)Q C 5( 1, 1)P B – 42 – C/2000 WM1 (LINEAR) 2.5 1.5 1180 Wavelength (Å) f2g.eps 1190 1200