Longer-term and short-term variability in pollution disruptive compounds
advertisement

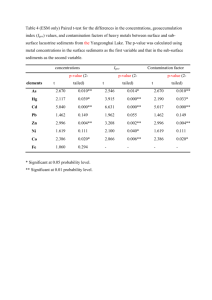
Environ Sci Pollut Res (2014) 21:5007–5022 DOI 10.1007/s11356-013-2429-8 RESEARCH ARTICLE Longer-term and short-term variability in pollution of fluvial sediments by dioxin-like and endocrine disruptive compounds P. Macikova & T. Kalabova & J. Klanova & P. Kukucka & J. P. Giesy & K. Hilscherova Received: 9 September 2013 / Accepted: 3 December 2013 / Published online: 22 December 2013 # Springer-Verlag Berlin Heidelberg 2013 Abstract Changes in pollutant loads in relatively dynamic river sediments, which contain very complex mixtures of compounds, can play a crucial role in the fate and effects of pollutants in fluvial ecosystems. The contamination of sediments by bioactive substances can be sensitively assessed by in vitro bioassays. This is the first study that characterizes detailed short- and long-term changes in concentrations of contaminants with several modes of Responsible editor: Ester Heath Electronic supplementary material The online version of this article (doi:10.1007/s11356-013-2429-8) contains supplementary material, which is available to authorized users. P. Macikova : T. Kalabova : J. Klanova : P. Kukucka : K. Hilscherova (*) Research Centre for Toxic Compounds in the Environment (RECETOX), Faculty of Science, Masaryk University, Kamenice 753/5, 625 00 Brno, Czech Republic e-mail: hilscherova@recetox.muni.cz J. P. Giesy Department of Biomedical Veterinary Sciences and Toxicology Centre, University of Saskatchewan, Saskatoon, SK, Canada J. P. Giesy Zoology Department and Centre for Integrative Toxicology, Michigan State University, East Lansing, MI 48824, USA J. P. Giesy Department of Biology and Chemistry, City University of Hong Kong, Hong Kong SAR, People’s Republic of China J. P. Giesy Zoology Department, College of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia J. P. Giesy Environmental Science Program, Nanjing University, Nanjing, China action in river sediments. One-year long monthly study described seasonal and spatial variability of contamination of sediments in a representative industrialized area by dioxin-like and endocrine disruptive chemicals. There were significant seasonal changes in both antiandrogenic and androgenic as well as dioxin-like potential of river sediments, while there were no general seasonal trends in estrogenicity. Aryl hydrocarbon receptor-dependent potency (dioxin-like potency) expressed as biological TCDDequivalents (BIOTEQ) was in the range of 0.5–17.7 ng/g, dry mass (dm). The greatest BIOTEQ levels in sediments were observed during winter, particularly at locations downstream of the industrial area. Estrogenicity expressed as estradiol equivalents (EEQ) was in the range of 0.02–3.8 ng/g, dm. Antiandrogenicity was detected in all samples, while androgenic potency in the range of 0.7–16.8 ng/g, dm dihydrotestosterone equivalents (DHT-EQ) was found in only 30 % of samples, most often during autumn, when antiandrogenicity was the least. PAHs were predominant contaminants among analyzed pollutants, responsible, on average, for 13–21 % of BIOTEQ. Longer-term changes in concentrations of BIOTEQ corresponded to seasonal fluctuations, whereas for EEQ, the inter-annual changes at some locations were greater than seasonal variability during 1 year. The inter- as well as intra-annual variability in concentrations of both BIOTEQ and EEQ at individual sites was greater in spring than in autumn which was related to hydrological conditions in the river. This study stresses the importance of river hydrology and its seasonal variations in the design of effective sampling campaigns, as well as in the interpretation of any monitoring results. Keywords Sediments . Seasonality . Monitoring . Dioxin-like potency . Estrogenicity . Antiandrogenicity 5008 Introduction Sediments are considered as an important compartment of aquatic ecosystems that provide substratum for benthic organisms and represent a deposit of nutrients that can be returned to the biocycles during natural flooding (Forstner and Salomons 2010). Association with sediments and particulate matter also plays a crucial role in the fate and effects of contaminants in aquatic systems. Sediments serve as a sink for various hazardous chemicals, especially hydrophobic organic contaminants (HOCs) due to their hydrophobic nature and low-water solubility. Important parameters for the binding of organic pollutants to sediments are the specific surface of particles as well as quantity and quality of organic carbon (Jaffe 1991). Sediments contain a wide spectrum of compounds, of both natural and anthropogenic origin, that can affect organisms through different modes of action to cause additive, supra-additive, or infra-additive effects. Among HOCs, polycyclic aromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs), organochlorine pesticides (OCPs) or polychlorinated dibenzo-p-dioxins (PCDDs), and dibenzofurans (PCDFs) have been detected in sediments worldwide (Colombo et al. 2006; Hilscherova et al. 2010; Kannan et al. 2008; Koh et al. 2004). Apart from the traditionally monitored hydrophobic pollutants, other classes of compounds such as pharmaceuticals and personal care products, polyphenolic compounds, phthalates, or various pesticides may be present in sediments (Brack et al. 2007; Jobling and Tyler 2003; Vigano et al. 2008). It has also been shown that sediments can serve as a sink of xenohormones and other endocrine disrupting compounds (Higley et al. 2012; Peck et al. 2004; Urbatzka et al. 2007). To achieve good water quality within the European Union (EU), the Water Framework Directive (Directive 2000/60/EC) has been introduced into the EU legislation and limits for concentration of several hazardous priority substances in surface waters, so-called Environmental Quality Standards (EQS), have been defined (Directive 2008/105/EC). Recently, the list of priority substances has been revisited and EQS for more compounds in surface waters as well as EQS for some compounds in biota have been proposed (European Commission 2012). Contamination of sediments plays a crucial role in the pollution of aquatic environment. The Water Framework Directive recommends the monitoring of sediments at an adequate frequency to provide sufficient data for reliable determination of long-term status and trends and to establish limits for contaminants in sediments according to the local situation in each country. Specific approaches for sediment quality assessment along with EQS for sediments are under development, which is one of the remaining challenges for better protection of aquatic ecosystems. Sediment quality guidelines (SQGs) developed on the base of ecological and ecotoxicological information for several HOCs as well as metals have been Environ Sci Pollut Res (2014) 21:5007–5022 introduced in Flanders, Belgium and incorporated into Flemish legislation in 2010 (de Deckere et al. 2011). Another approach was previously used in the Netherlands, where limits for some organic substances and pesticides were derived by use of the equilibrium partitioning method (Crommentuijn et al. 2000). An SQG for PCBs corresponding to the regulatory fish consumption limit based on biota-to-sediment accumulation factor has been derived for the Rhone River basin, France (Babut et al. 2012). No SQG have been promulgated by the Czech Republic yet. Implementation of EQS for priority substances in sediments is a crucial step in better protection of aquatic environments. However, priority pollutants remain to be identified. Recently, more attention has been driven to “emerging contaminants” in addition to HOCs since they can elicit various biological responses (Brack et al. 2007; Kaplan 2013). While quantification of individual contaminants by instrumental analysis is an important tool to investigate the fate and distribution of known pollutants in the environment, possible biological effects of complex mixture are difficult to predict solely from chemical analysis. Instrumental analysis of individual, known contaminants does not account for possible interactions among chemicals or for those compounds that are not identified or not quantified. Thus, various in vitro bioassays have been applied to characterize contamination by bioactive substances in various environmental compartments, such as surface water, sediments, soil, air, or biota (e.g., Higley et al. 2012; Martinez-Gomez et al. 2013; Novak et al. 2009; Urbatzka et al. 2007; Wolz et al. 2011). In vitro bioassays are relatively rapid, cost-effective, and useful, especially in screening and long-term monitoring of contamination. Some of these assays are applied to estimate the potency of individual compounds as well as of complex mixtures to elicit biological responses mediated through specific nuclear receptors, such as the aryl hydrocarbon receptor (AhR), estrogen receptor (ER), or androgen receptor (AR). Activation of the AhR is considered critical in mediating effects of dioxin-like compounds that have been shown to cause hepatotoxicity, teratogenicity, carcinogenesis, immunotoxicity, and other adverse effects (Janosek et al. 2006). Estrogens and androgens are endogenous steroid sex hormones that control reproduction, development, differentiation, and growth. Functions of these hormones are mainly mediated by ER and AR, and many compounds have been shown to disrupt their signaling (Janosek et al. 2006). Reproductive disorders, such as feminization or masculinization of aquatic vertebrates and invertebrates were observed in the environment (as reviewed in Sumpter 2005). Exposure to synthetic estrogens can even lead to collapse of whole fish populations (Kidd et al. 2007). River sediments represent a dynamic system and their potential risks are connected primarily with transport and deposition of contaminated solids in downstream regions Environ Sci Pollut Res (2014) 21:5007–5022 (Forstner et al. 2004; Hilscherova et al. 2003). Rivers can exhibit large differences in hydrodynamic characteristics during an annual cycle. In remobilization processes, pollutants associated with particles can be resuspended, thus, sediments can serve as a secondary source of contamination (Brinkmann et al. 2013; Hilscherova et al. 2007). Strong fluctuations in concentrations of contaminants can occur upon stronger floods that have been discussed in recent years in possible relation to the global climate change (Hunt 2002). Further, seasonal variability of contamination was observed at some places (Hilscherova et al. 2010; Zhao et al. 2011). Both temporal and spatial dynamics should be considered when assessing contamination of river ecosystems as was documented in a previous study (Hilscherova et al. 2010). Even though pollution of sediments by compounds with the abovelisted modes of action has been reported from rivers in many parts of the world, there is a lack of information regarding long- as well as short-term variations or trends in their concentrations and/or potencies. The present year-long study was focused on temporal (both seasonal and long-term) and spatial variability of contaminants in river sediments of a typical industrial area in the southeastern part of the Czech Republic (Central Europe) that represents a suitable model ecosystem for research on the accumulation and distribution of pollutants on a local and regional scale. The studied region is a part of Danube River basin, situated near the city of Zlin. It includes two rivers, the Morava and its tributary, the Drevnice (Fig. 1). This area has been affected for many years by industrial and agricultural activities as well as effluents from wastewater treatment facilities and runoff from urban landscapes. Chemical, boot-andshoe, plastics-and-rubber, food-stuff industry, agricultural crops and livestock production, as well as transport are among the most important sources of contamination (Hilscherova et al. 2007). The goal of this year-long study with monthly sampling was to characterize seasonal and spatial variability of contamination of fluvial sediments by compounds with dioxinlike and endocrine disruptive modes of action. Sediments were sampled monthly at five locations throughout a whole year. Extracts of sediments were assessed for AhR-, ER-, and ARdependent potencies. Another goal was to address longer-term trends/variability through comparison of current and previous results from the region (Hilscherova et al. 2010, 2002). Thus, a comparison of seasonal as well as inter-annual trends in contamination by bioactive compounds could be conducted. Materials and methods Sampling and locations Sediments were collected, monthly, from July 2007 to July 2008 at five locations in the south-eastern part of the Czech 5009 Republic in the Morava River and its tributary Drevnice River (Fig. 1). The Malenovice (MA) location is situated on the Drevnice River and is affected mainly by contamination from the city of Zlin and its surroundings. The Belov (BE) location is situated on the Morava River upstream from the confluence with the Drevnice River, whereas the Spytihnev (SP) location is downstream on Morava River and integrates contamination from both rivers. The Certak oxbow lake (CR) is a unique location that was separated from the active Morava River channel in the 1930s, but water communication with the river is provided via underground piping that enables the lake to act as a trap for suspended sediments from the river (Babek et al. 2008). The Certak (CE) location is situated on the Morava River near the oxbow lake to better assess differences between the active and abandoned channel. Samples were taken from each location in a period of 28 days, in 15 sampling campaigns. A total of 73 samples were collected. Two samples could not be obtained because of weather conditions. Samples were clustered according to four hydrologically defined seasons (Table S1): spring (March–May), summer (June– August), autumn (September–November), and winter (December–February). Data on river discharge and temperature were obtained from gauging stations in Zlin (representative for location MA), Kromeriz (representative for location BE), and Spytihnev (representative for locations SP, CE). The following parameters were used: Q =average discharge over the 28 days prior to each sampling campaign, T actual =temperature on the day of sampling, T average =time-weighted, average temperature over the 28 days prior to each sampling campaign. Composite samples of surface sediments were collected from the top 10-cm layer by use of pre-cleaned trowels. Large pieces of wood, leaves, and stones were removed manually and sediments were homogenized and freeze-dried. Dry sediments were sieved (2 mm). Total organic carbon content (TOC) was determined by use of high-temperature TOC/ TNb Analyzer liquiTOC II (Elementar Analysensysteme, Hanau, Germany). Chemical analysis For quantification of organic pollutants, 10 g of freeze-dried sediments were extracted with dichloromethane by use of automated warm Soxhlet extraction (1 h, min. 15 cycles; Büchi B-811, Büchi, Switzerland). Laboratory blanks and reference material were analyzed with each set of samples. Surrogate recovery standards (final amount in each sample 10 ng PCB30, 10 ng PCB185, 333 ng D8-naphthalene, 333 ng D10-phenanthrene, and 333 ng D12-perylene) and 13C labeled PCDD/Fs standards (800 pg tetra-hexa PCDD/Fs, 1,600 pg hepta-octa PCDD/Fs) were used prior to extraction. Extracts were cleaned-up on silica column (for PAHs analysis), sulfuric acid-modified silica column was used for analysis of organohalogens. Copper powder was used to remove 5010 Environ Sci Pollut Res (2014) 21:5007–5022 Fig. 1 Sampling localities on the Morava and Drevnice Rivers. MA Malenovice, BE Belov, SP Spytihnev, CE Certak Morava river, CR Certak oxbow lake; arrows indicate the river flow direction sulfur. Further fractionation step was needed to analyze dioxin-like PCBs (dl-PCBs) and PCDD/Fs. Samples were applied on columns containing charcoal/silica mixture and eluted with DCM/cyclohexane in fraction 1 (mono-ortho dlPCBs) and with toluene in fraction 2 (PCDD/Fs, non-ortho dl-PCBs). Terphenyl (200 ng/mL), PCB 121 (200 ng/mL) and 13C-labeled PCDD/Fs (16 ng/mL) were used as injection standards for quantification of PAHs, PCBs, and PCDD/Fs, respectively. Samples were analyzed using GC-MS instrument (Agilent 6890N GC–Agilent 5973N MS, Agilent, USA), separation of individual compounds was achieved on a DB-5MS (J&W Agilent, USA) for indicator PCBs (congeners 28, 52, 101, 118, 138, 153, 180), OCPs (dichlorodiphenyltrichloroethane p , p′-DDT and its metabolites p, p′-DDE, p , p′-DDD; hexachlorocyclohexane isomers α-, β-, γ-, δ-HCH; hexachlorobenzene), and 16 US EPA PAHs. Concentrations of contaminants were quantified using Pesticide Mix 13 (Dr. Ehrenstorfer GmbH, Germany) and PAH Mix 27 (Promochem, Germany) standard mixtures. HRGC/HRMS instrumental analysis for PCDD/Fs and dlPCBs (congeners 77, 81, 105, 114, 123, 126, 156, 157, 167, 169, 189) was performed on Agilent 7890A GC (Agilent, USA) coupled to AutoSpec Premier MS (Waters, Micromass, UK). The GC was fitted with a capillary column DB5-MS, 60 m×0.25 mm i.d., 0.25-μm film. MS was operated in EI+ mode at R >10 k (Kukucka et al. 2010). Total potency of samples to cause AhR-mediated effects, expressed as 2,3,7,8-tetrachlorodibenzo-p -dioxin (TCDD)equivalents (TEQ), were calculated as the sum of the product of concentrations of individual AhR-active compounds multiplied by their relative potency (REP) to activate AhRmediated responses in H4IIE-luc cells (Eq. 1). TEQ ¼ X ð1Þ cX *REPX TEQ for individual groups of pollutants were calculated (Eqs. 2 and 3). PAHs−TEQ ¼ nonPAHs−TEQ ¼ X X X cPCBs *REPPCBs þ X þ ð2Þ cPAHs *REPPAHs cPCDFs *REPPCDFs cPCDDs *REPPCDDs ð3Þ Environ Sci Pollut Res (2014) 21:5007–5022 REPs derived by Machala et al. (2001) were used for PAHs, REPs derived by Behnisch et al. (2003) were used for PCBs and PCDD/Fs (Table S2). Bioassays For in vitro testing, 20 g of freeze-dried sediments without any surrogate standards were extracted as described above (Section Chemical Analysis). Extracts were treated with copper powder to remove sulfur, enriched under a gentle stream of nitrogen, and aliquots were transferred to ethanol (EtOH) and dimethyl sulfoxide (DMSO). Final concentration of sediment equivalents (SEQ) in the extracts was 20 g/mL. Three different mammalian cell lines transfected with the luciferase gene under control of several intracellular receptors were used to determine potencies of extracts of sediments to interfere with receptor-mediated responses. The potency to elicit dioxin-like effects via activation of AhR was quantified by use of the H4IIE-luc rat hepatocarcinoma cells (Hilscherova et al. 2001). ER-mediated response was evaluated by use of MVLN human breast carcinoma cells (Demirpence et al. 1993). MDA-kb2 human breast cancer cell line was used to assess AR-dependent response (Wilson et al. 2002). H4IIE-luc cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10 % (v/v) fetal calf serum (FCS; both PAA laboratories, Austria) and exposed in the same medium supplemented with 1 % (v/v) gentamicin to prevent bacterial contamination. MVLN cells were cultured in DMEM/F12 medium (Sigma-Aldrich, Czech Republic) supplemented with 10 % (v/v) FCS and exposed in DMEM/F12 supplemented with 5 % (v/v) stripped (dextran/charcoal treated) FCS and 1 % (v /v ) gentamicin. MDA-kb2 cells were cultured in Leibowitz L-15 medium (Sigma-Aldrich, Czech Republic) supplemented with 10 % (v/v) FCS and exposed in Leibowitz L-15 medium supplemented with 5 % (v /v ) stripped FCS and 1 % (v /v ) gentamicin. H4IIE-luc and MVLN cells were incubated and exposed at 5 % CO2 and 37 °C. MDA-kb2 cells were incubated and exposed at 37 °C without addition of CO2. In the first step, test of cytotoxicity of the sediment extracts was conducted to determine the non-cytotoxic concentrations for testing of receptor-mediated effects. Upon testing, cells were seeded into sterile 96-well microplates in exposure medium. After 24-h incubation, cells were exposed to extracts of sediment samples in several dilutions. The greatest tested concentration for cytotoxicity assessment was 100 mg SEQ/ mL. Cytotoxicity of the samples was measured using colorimetric Neutral Red (NR) uptake assay (Babich and Borenfreund 1990). Fifty microliters of NR dissolved in DMEM (0.5 mg/mL) were added into each well with cells and exposure medium after 24-h exposure. The mixture was 5011 incubated with cells for 1 h and then the medium with NR was removed. An aliquot of 150 μL of lysis solution (water, ethanol, acetic acid) was added and cells were shaken for 15 min (Orbital Shaker OS-20, BIOSAN, at 150 rpm). Absorbance was measured using a spectrophotometer (Tecan-Genios, λ =570 nm). Data from the cytotoxic sample dilutions were excluded from calculations. The interference with the receptor signaling was tested at several dilutions that did not significantly affect the viability of the cells, in three independent experiments. Cells were seeded into 96-well microplates and after 24-h incubation, exposed to extracts of sediment samples and appropriate standard calibration for agonistic potency along with blanks and solvent controls (0.5 % v/v). Reference compounds used for calibration were TCDD (Ultra Scientific, USA; concentration range 0.4–500 pM in EtOH) for H4IIE-luc, 17β-estradiol (E2; Sigma-Aldrich, Czech Republic; 1.23–100 pM in DMSO) for MVLN, and dihydrotestosterone (DHT; SigmaAldrich, Czech Republic; 10 pM–10 μM in DMSO) for MDA-kb2, respectively. For ER- and AR-antagonistic potency assessment, the exposure medium was supplemented with the reference compound (competing ligand) at approximately EC50 level, e.g., 33.3 pM E2 (MVLN) and 1 nM DHT (MDA-kb2), thus solvent concentration was 1 % (v/v). After 24-h exposure, cells were lysed, Promega Steady Glo Kit (Promega, USA) was added and the intensity of luminescence was measured by Luminoskan Ascent Microplate Luminometer (Thermo Scientific). Data analysis After subtraction of solvent control response, effects elicited by extracts of sediments were related to the luminescence caused by the reference compounds in the transactivation assay. The dose–response curves were fitted using non-linear logarithmic regression in GraphPad Prism (GraphPad Software, USA). AhR-mediated potency was expressed as TCDD-equivalents (BIOTEQ) calculated as EC50TCDD/ EC50sample. Since many of the active samples did not reach 50 % of E2max induction, to avoid any predictions beyond the measured responses, estrogenicity was expressed as estradiol equivalents (EEQ) calculated as EC25 E2 /EC25 sample. Antiestrogenicity of samples was expressed as the concentration (in sediment equivalents) that caused 25 % inhibition of luminescence in the presence of the competing ligand E2 (IC25). Androgenicity was expressed as DHT-equivalents (DHT-EQ) calculated as a point estimate based on the percentage of the luminescence induction caused by the greatest non-cytotoxic sample concentration because the dose–response curve for most samples did not exceed 20 % induction. DHT-EQ was calculated as ECX DHT/ECX sample, where X represents percentage induction of the greatest non-cytotoxic sample concentration. Antiandrogenicity was expressed as 5012 percent inhibition of luminescence in the presence of the competing ligand DHT caused by the greatest concentration that was non-cytotoxic (as described for DHT-EQ). The limit of detection (LOD) for each bioassay used in this study was derived as the ratio of the lowest amount of standard that elicits statistically significant response per the greatest tested non-cytotoxic concentration of SEQ. To calculate the LOD, the lowest concentration of reference compound significantly affecting the receptor-mediated response (lowest observed effect concentration for the receptor-mediated effect; LOEC), and the greatest non-cytotoxic sample concentration (no observable effects concentration for cytotoxicity; NOEC) were determined. Responses obtained for reference compounds and sample extracts were compared with solvent control response using ANOVA followed by Dunnet’s test to determine significant effects (p < 0.05). Nonparametric Kruskal-Wallis test was used in case of non-homogenous variances (as tested by Levene’s test). The LOD was then calculated as follows: LOD (pg/g, dry mass (dm) of sediment) = LOECstandard ligand (pg/mL)/NOECsample (g SEQ/mL). Spatial and seasonal variability of dioxin-like toxicity (TEQ, BIOTEQ), estrogenic potency (EEQ), and antiandrogenic potency (AA) was tested by nonparametric Kruskal-Wallis test and visualized using boxplots. Multivariate variation of bioassays results as well as chemical and environmental parameters was further summarized in the principal component analysis (PCA) as an effective technique simplifying the correlation structure through linear transformation of the original variables. PCA based on the correlation matrix was performed to provide component loading vectors explaining the relationships among the bioassays, pollutants, and other parameters and component score vectors as pair-wise uncorrelated variables that were used for the final exploratory survey of the data from the examined locations. Only variables with less than 10 % values below LOD were used for multivariate analysis. Values < LOD were replaced by ½ LOD. The variables with non-normal distribution were transformed by logarithmic transformation before use in PCA and parametric correlation analyses. The most important variables (estimates by eigenvalues) were selected for creating PCA (active variables), some other variables were visualized in the same ordination space as supplementary variables. Biplot was used as a common graphical tool representing not only projections on extracted principal components but also the 2-D loadings of original variables by lines. Additionally, Pearson’s correlation analysis was used to quantify relationships between variables. All statistical analyses were performed with the software STATISTICA for Windows 10 (StatSoft, Inc. USA). Environ Sci Pollut Res (2014) 21:5007–5022 Results and discussion This study documents variability of pollution in surface sediments of the rivers during the year. Sediments contained all chemically analyzed classes of pollutants (Table 1) at each location. SP was the most polluted location with the greatest concentrations of most contaminants and also with the greatest median of TOC content, whereas CR (oxbow lake) contained, overall, the least levels of contaminants. Detailed information about temporal and spatial distribution of HOCs will be described elsewhere (Prokes et al., in preparation). Comparing the contamination in assessed areas with SQGs derived from ecological and ecotoxicological data by de Deckere et al. (2011) (Table S3), all of the investigated locations are polluted. The SQGs proposed to be achieved in a long-term objective (so-called consensus 1 values) were exceeded by concentrations of PAHs, PCBs, and DDE in all samples up to 6-, 7-, and 30-fold, respectively, especially in winter and spring (PAHs), and in autumn (PCBs, DDE). In the case of DDD, the SQGs were exceeded even more than 200-fold in winter samples from location SP. According to these results, the studied locations are not in a good ecological sediment status as it was defined in de Deckere et al. (2011) during the year. The SQGs proposed to be achieved as a short-term objective (consensus 2 values) were only slightly exceeded by concentrations of some PAHs and DDE (1.3-fold), while the concentration of DDD was up to 5-fold greater in winter samples from location SP than the proposed limits. Consensus 2 values are described as values above which toxic and in situ effects are most likely to occur (de Deckere et al. 2011). From this point of view, all investigated sediments are likely to negatively affect biota. Desorption of contaminants from sediments might enhance their bioavailability, which plays a crucial role in manifestation of toxic effects on organisms. Results of previous studies indicated that sediments from the studied area represent a potential source of PAHs into the water column (Prokes et al. 2012). Comparison of chemical analysis results to SQGs documents pollution by HOCs. However, compounds other than those HOCs that were quantified were present in the mixtures in sediments; therefore, specific biological activities were assessed in order to estimate the potential effects on organisms. Three transactivation cell lines were used to investigate specific biological potential of sediment samples. Mixtures extracted from sediments were cytotoxic; therefore, extracts were first treated with copper to remove sulfur, which is a frequent cause of cytotoxicity. Cytotoxicity of treated extracts was measured by use of the NR assay for each cell line in order to avoid any interference with specific endpoints measured in this study. Only concentrations of extracts that did not cause cytotoxicity were included in the evaluation of specific potencies. MDA-kb2 cells were more sensitive to effects on viability than were H4IIE-luc and MVLN cells. For MDA- kb2 cells, the greatest NOEC corresponded to 50 mg SEQ/mL for most samples, four sediment extracts from location MA showed a greater cytotoxicity with NOEC of 15 mg SEQ/mL. For H4IIE-luc and MVLN cells, the cytotoxicity NOEC was 100 mg SEQ/mL for all samples. Total number of samples (n) with observed activity is noted in brackets in case when significant activity was not detected in all 15 samplings over the year AhR-mediated potency a Abbreviations of sites as in Fig. 1 5013 PAHs polycyclic aromatic hydrocarbons, ind. PCBs indicator polychlorinated biphenyls, dl-PCBs dioxin-like PCBs, PCDDs polychlorinated dibenzo-p-dioxins, PCDFs polychlorinated dibenzofurans, OCPs organochlorine pesticides, TOC total organic carbon, TEQ TCDD-equivalent (derived based on chemical analysis), BIOTEQ TCDD-equivalent (bioassay-derived), EEQ estradiol-equivalent (bioassay derived), DHT-EQ dihydrotestosterone-equivalent (bioassay-derived), AA antiandrogenic activity (bioassay-derived) 74 (52–98) 1.9 (0.5–5.3) 3.7 (1.3–15.4) 5.3 (1.2–11.6) 2.9 (2.1–5.9) 0.3 (0.2–0.9) 21 (10–44) 5.4 (2.9–39.5) 1.8 (1.2–2.6) 0.7 (0.4–1.1) 17 (11–24) 13 (11–28) 52 (36–94) 106 (57–268) 7.1 (4.5–20.6) 317 (142–643) 6.2 (0.7–13.7) 290 (188–431) 13 (6–49) 5.2 (3.0–9.0) 2.6 (1.5–8.8) CE CR 110 (62–229) 5.4 (0.8-16.8) (n=7) 56 (17–84) 72 (32–98) 143 (45–895) 4.1 (0.7-5.9) (n=7) 6.4 (1.5–17.7) 8.5 (5.3–13.8) 12.0 (6.5–33.4) 300 (127–612) 354 (152–818) 37 (24–109) 25.3 (8.9–58.1) 4.1 (2.9–5.3) 0.9 (0.6–1.9) 17 (8–109) SP 75 (39–198) (n=12) 1.9 (n=1) 72 (51–84) 68 (17–91) 76 (40–3,753) 2.8 (1.4–8.3) (n=4) 4.9 (1.0–12.6) 3.7 (0.9–14.7) 99 (20–954) (n=13) 3.1 (1.8–3.9) (n=3) 3.3 (0.6–7.7) 0.6 (0.1–1.4) 9.2 (1.6–19) 34 (4–49) 16.7 (0.9–53.1) 3.1 (0.2–5.1) 1.0 (0.1–1.2) 15 (2–23) 38 (8–94) 90 (11–190) 7.5 (1.7–10.5) 238 (105–340) 14 (5–52) 7.5 (0.5–10.7) 15 (6–39) 4.4 (0.6–14.3) 11.6 (3.5–21.4) 399 (179–494) MA BE AA (% inhibition) DHT-EQ (ng/g)a EEQ (pg/g)a BIOTEQ (ng/g) TEQ/ BIOTEQ (%) TEQ (ng/g) TOC (%) OCPs (ng/g) PCDFs (pg/g) PCDDs (pg/g) Dl-PCBs (pg/g) Ind. PCBs (ng/g) Sampling PAHs site (μg/g) Table 1 Median and range (in brackets) of concentrations of pollutants, organic carbon, and biological potencies in extracts of sediments from studied localities from all sampling campaigns over a year (based on sediment dry mass) Environ Sci Pollut Res (2014) 21:5007–5022 AhR-mediated potency, expressed as BIOTEQ, was found in extracts of all sediments and was in the range of 0.5–17.7 ng/ g, dm of sediment (LOD=1.3 pg/g, dm). Seasonal changes in BIOTEQ were obvious at all locations except CR (Figs. 2a and 3). The greatest dioxin-like potency was detected in sediments collected during winter. Concentrations of BIOTEQ in sediments collected during winter were significantly greater than in those collected during summer, which contained the least concentration of BIOTEQ (p <0.05). The same trend was observed for content of TOC (Fig. S1), which is an important parameter in accumulation of hydrophobic pollutants (Jaffe 1991). The trend of greatest concentrations in winter was most pronounced in the Morava River below the confluence with the Drevnice River (locations SP, CE). There was a trend of increasing concentration of BIOTEQ at SP compared to upstream locations (MA, BE) in samples collected during the summer and winter (Fig. 3 and S2). However, this trend was not obvious in spring and autumn, which indicates that spatial differences can be more pronounced during some seasons. SP is an integrating location for contamination from both rivers and additional nearby sources of pollution. Location CR (oxbow lake) was the least contaminated location, with concentrations of BIOTEQ significantly lesser (p <0.05) than those in sediments from locations BE, SP, and CE (Fig. 2a, Table 1). CR also exhibited lesser variability among seasons with AhR-mediated potency only slightly greater in sediments collected during winter (Fig. 3). This finding demonstrates the function of the oxbow lake as a more stable deposit of various HOCs without greater fluctuations in pollution that are obvious in the active channel. Concentrations of BIOTEQ in sediments from all riverine locations exhibited least variability during summer (Fig. 3), which was probably related to the least fluctuations in river water discharge during this season (Fig. S3). Variability in concentrations of BIOTEQ was greater among all riverine locations during winter and spring (Fig. 3). Results of this study confirmed the role of PAHs as the predominant contributors to the overall AhR-mediated potency observed in previous studies from the region (Hilscherova et al. 2001; Vondracek et al. 2001). Total TEQ calculated from concentrations of individual AhRactive compounds (0.1–1.9 ng/g; Table 1) exhibited similar seasonal patterns as did concentrations of BIOTEQ (Fig. S4). However, concentrations of BIOTEQ were greater than concentrations of TEQ in extracts of all sediments 5014 Environ Sci Pollut Res (2014) 21:5007–5022 Fig. 2 Spatial and seasonal variability of bioassay-derived: a dioxin-like potency (BIOTEQ, pg/g, dm of sediment), b estrogenic potency (EEQ, pg/g, dm), c antiandrogenic potency (AA, % luminescence inhibition in competition with DHT caused by the highest non-cytotoxic sample concentration) in sediment samples from the 15 sampling campaigns in July 2007–July 2008 (n =73). Middle line is median, box means quartile range (25–75 %), whisker is non-outlier range and triangles are measured values with a single exception. Generally, only 13–21 % BIOTEQ (median values “TEQ/BIOTEQ (%)” among locations; Table 1) could be explained by the presence of known AhR ligands, namely PAHs. PAHs accounted on average for 99.4 % of the total TEQ, which is consistent with the results of previous studies conducted in this area (Hilscherova et al. 2001; Vondracek et al. 2001). The main contributors among PAHs were benzo[k]fluoranthene and indeno[123cd]perylene. Other source of dioxin-like toxicity might be azaarenes and oxygenated PAH derivatives (oxy-PAHs) that were previously detected in sediments from the studied area (Machala et al. 2001). Comparable levels of pollution with dioxin-like compounds were found in sediments from rivers affected by municipal and industrial activities from other areas, such as sediments from two Chinese rivers (BIOTEQ 0.3– 13.9 ng/g, dm), where greatest potencies were observed in fractions containing PAHs, OCPs, a portion of PCDD/Fs and unknown compounds (Song et al. 2006). In sediments from the Environ Sci Pollut Res (2014) 21:5007–5022 5015 Fig. 3 Seasonal variability of bioassay derived dioxin-like activity (BIOTEQ, pg/g, dm) at each sampling site during the 15 sampling campaigns in July 2007–July 2008 (n =73). Middle line is median, box means quartile range (25–75 %), whisker is non-outlier range and triangles are measured values Netherlands, most AhR-mediated potency was caused by acidlabile compounds, such as PAHs (Houtman et al. 2004). Alternatively, only 6 % of AhR-mediated potency was attributed to PAHs in sediments from Germany (Brack et al. 2008). Furthermore, PCDD/Fs were a major source of dioxin-like potency observed in the sediments in the USA where concentrations were as great as 19.9 and 17.7 ng/g, dm PCDDs and PCDFs, respectively (Hilscherova et al. 2003), and 46.5 ng/g, dm for the sum of PCDD/Fs (Kannan et al. 2008). However, in the study, the results of which are reported here, dl-PCBs and PCDD/Fs, due to their relatively small concentrations relative to PAHs, contributed little of the total concentrations of TEQ (Table 1). Despite their greater potency, the average contribution of PCDDs, PCDFs, and dl-PCBs to the total TEQ was only 0.2, 0.3, and 0.1 %, respectively. ER-mediated potency Results of the bioassay documented the presence of estrogenic compounds in almost all sediments. Estrogenic potency expressed as EEQ was detected in 93 % of samples in the range of 20–3,753 pg/g, dm, but most samples (88 %) contained 20–300 pg/g, dm (Fig. 2b). Four samples taken in summer 2007 at locations MA (n =1), BE (n =2), and SP (n = 1) were noted for great values of EEQ that reached 895– 3,753 pg/g, dm. These extreme concentrations are not included in Fig. 2b. Estrogenic potency of 7 % of the sediment samples (n =5) was less than the limit of detection (LOD= 3.25 pg/g). Median concentration of EEQ among seasons was greatest in sediments from location SP, similarly to median concentration of BIOTEQ. Samples from BE and MA were noted for a very variable estrogenicity among sampling campaigns including extreme EEQ values. When seasonal variability throughout the year was taken into account, statistically significant difference in estrogenic potency was observed only between locations SP and CR. But there were more pronounced differences in separate seasons. For example, EEQ were always greater in SP compared to BE in autumn and winter samples, while there was no such trend in the other two seasons. The greatest median estrogenic potency across locations was observed in summer (Fig. 2b), even without the extreme concentrations of EEQ observed in a few samples. These extremes could indicate exceptional inputs of (xeno-)estrogens of unknown origin that occurred during early summer 2007 at the above mentioned localities. No general significant seasonal trends in estrogenicity were observed in this study (Fig. 2b). However, sediments sampled at locations MA and CE tended to have lesser concentrations of EEQ in spring compared to autumn. A similar trend was observed previously in this region (Hilscherova et al. 2010; Table 2). Alternatively, greater estrogenicity was observed in sediments collected in spring than in those collected in autumn in a study where a smaller sample set was compared (Creusot et al. 2013). A weak antiestrogenic potency in the presence of competing E2 was detected only in two sediment samples taken from CR in November and December 2007. Concentration of extract causing 25 % inhibition of luminescence (IC25) in competition with E2 was 25.6 and 16.6 mg SEQ/mL, respectively. These two samples exhibited none and little estrogenic potency, respectively, but the presence of estrogenic pollutants might be masked by antiestrogenic compounds present in 5016 Environ Sci Pollut Res (2014) 21:5007–5022 Table 2 Concentrations of AhR-mediated potency (BIOTEQ, pg TCDD/g, dm) and estrogenic potency (EEQ, pg E2/g, dm) of sediments at Malenovice (MA), Belov (BE) and Spytihnev (SP) determined by bioassays BIOTEQ (pg TCDD/g) EEQ (pg E2/g) MA BE SP MA BE SP October 1996a 6,542 4,223 NA 239 39 NA October 1997a 6,675 4,449 NA 1,134 93 NA May 2005b 15,368 8,123 9,867 95 29 186 October 2005b 7,868 1,442 4,611 231 107 175 May 2006b 7,768 914 14,356 <1 <1 90 October 2006b 1,660 764 5,573 442 66 127 October 2007 5,506 6,488 7,779 124 85 178 May 2008 1,333 3,001 3,166 <3 51 130 Summer 2007 1,058–2,302 1,042–2,566 2,098–5,893 141–954 198–3,753 45–895 Autumn 2007 2,139–5,663 2,927–6,488 1,515–7,884 99–124 58–85 136–178 Winter 2007/08 3,708–13,797 3,413–12,624 10,276–17,722 60–167 44–117 122–154 Spring 2008 1,333–14,690 3,001–9,768 3,166–12,317 <3–76 51–212 87–130 Summer 2008 940–1,867 2,062 3,870–6,824 20–61 40 97–131 NA data not available a Hilscherova et al. (2001) (2002) b Hilscherova et al. (2010) these samples. There were four samples that elicited neither estrogenic nor antiestrogenic potency. Antiestrogenic effects might play an important role in some regions. For example, 81 % of sediments from the Pearl River, China, exhibited estrogenicity but at the same time, 61 % of all samples were antiestrogenic meaning that both estrogenic and antiestrogenic compounds were present (Zhao et al. 2011). Sediments from the Svratka and Svitava Rivers that flows into the Morava River downstream from the studied area of Zlin vicinity, elicited only antiestrogenic potencies (Jalova, personal communication) despite the fact that the region is relatively densely populated. The estrogenicity detected in sediments from the region around the city of Zlin indicates that there might be greater inputs of estrogenic compounds due to less effective wastewater treatment plants (WWTPs) and/or more intensive agriculture. ER-dependent potency was previously assessed in samples from the studied area. Concentrations of EEQ were in the range of 5–23 (Vondracek et al. 2001) and 10–1,200 pg/ g, dm in extracts of sediments (Hilscherova et al. 2002). After major floods in 1997, antiestrogenic potencies became more apparent in sediments compared to the situation before floods (Hilscherova et al. 2002). Approximately 10 years after the floods, regional median concentrations of EEQ in sediments from the studied area were in the range of 10– 340 pg/g, dm (Hilscherova et al. 2010). Estrogenic compounds (EEQ 21.3–29.9 pg/g, dm) were found in sediments from both upstream and downstream of WWTPs that are considered to be an important source of estrogenic compounds in UK; estrone (E1) and E2 were determined as major estrogenic pollutants (Peck et al. 2004). On the other hand, EEQ in the range of 3.3–10.6 pg/g, dm was detected in sediments from downstream locations from WWTPs in Korea, whereas no potency was observed in upstream locations (Oh et al. 2000). High contamination by estrogenic compounds was observed in sediment from a river in Italy, where E1, estriol (E3), and nonylphenol contributed to the observed estrogenicity; phthalates and octylphenol isomers were suggested as potential contributors (Vigano et al. 2008). In the area around the city of Zlin, rivers receive treated effluents from a number of WWTPs as well as untreated sewage effluents from smaller villages and farms. Effects of large as well as smaller towns as sources of estrogenic compounds have been documented (Jarosova et al. 2012; Vermeirssen et al. 2005). Natural and synthetic estrogens, such as E1, E2, E3, and ethinyl estradiol, were not analyzed in our study but they can enter the rivers and are likely to accumulate in sediments (Luo et al. 2011; Peck et al. 2004; Streck 2009). Therefore, they could be important contributors to the estrogenic potency of extracts of sediments. In addition, PAHs have been found to be a source of estrogenicity in sediments (Hilscherova et al. 2002, 2010; Houtman et al. 2004; Luo et al. 2011). In this study, concentrations of EEQ in sediments were not correlated with concentrations of measured PAHs. However, some of their metabolites produced in sediments by microbial degradation such as hydroxylated PAHs could play a role in the estrogenic effects (e.g., Hayakawa et al. 2007; Luan et al. 2006). Environ Sci Pollut Res (2014) 21:5007–5022 5017 AA was more profound in extracts of sediments than androgenic potency. All extracts at non-cytotoxic concentrations inhibited luminescence in competition with natural ligand DHT with median inhibition during the year at 56–74 % at all sites (Table 1). Antiandrogenicity was greater in extracts of sediments from CR than that from CE (Fig. 2c), namely in summer and winter. These results suggest that antiandrogenic compounds could accumulate better in relatively stable sediments of the oxbow lake. This could be also affected by lower TOC content at CE compared to the other sites, since AA was shown to correlate with organic carbon level (Fig. 4). The AA was least in autumn and significantly greater concentrations were observed in spring. Androgenic potency expressed as DHT-EQ greater than LOD (580 pg/g) was detected in 30 % of extracts of sediments. Concentrations of DHT-EQ were 0.7–16.8 ng/g, dm. Androgenic potency was detected in at least one sampling period at all locations, but in more than half of samples from the individual locations there was no detectable androgenicity (Table 1). Androgenic potency was detected most frequently in samples from locations SP and CE, whereas only one sample from CR exhibited androgenic potency. Androgenicity was detected most often in sediments collected during autumn (73 % of autumn samples), followed by summer (29 % of summer samples), while only three samples collected during winter and one during spring were androgenic. To our knowledge, this is the first study that documents the significant seasonal changes in both antiandrogenic and androgenic potential of organic extracts of sediments from a river. Seasonal changes in both androgenic and antiandrogenic potencies were in good agreement. The least antiandrogenic potency, which was observed during autumn, corresponded to the frequent detection of androgenicity in extracts of sediments collected during autumn. Alternatively, AA was greatest in spring when only one sample was androgenic. Previously, AA potency of few sediment samples was shown to be greater in dry season compared to wet season (Zhao et al. 2011). Furthermore, androgenic potency was observed in 34 of 50 extracts of sediments collected in Germany, but seasonal trends were not investigated (Galluba and Oehlmann 2012). Antiandrogenic potency has been frequently detected in studies of unfractionated extracts of sediments (Hilscherova et al. 2010; Zhao et al. 2011). In some studies, AA was a predominant effect in extracts of sediments, whereas androgenic potency was found in only some fractions (Urbatzka et al. 2007; Weiss et al. 2009). Effect-directed analysis was previously applied to reveal both AA and androgenic compounds in sediments. PAHs, such as fluoranthene, benz[a]anthracene, pyrene and phenanthrene, nonylphenol Fig. 4 Pearson’s correlation coefficient of bioassay-derived dioxin-like potency (BIOTEQ), estrogenic potency (EEQ), and antiandrogenic potency (AA) with other parameters. Dark bands indicate a significant correlation (p <0.05). Abbreviations as in Table 1; Σ DDT = sum of concentrations of dichlorodiphenyltrichloroethane (p, p′-DDT) and its metabolites p , p ′-DDE, p , p ′-DDD; PAHs -TEQ TCDD-equivalent calculated based on PAHs concentration, nonPAHs -TEQ TCDDequivalent calculated based on dl-PCBs and PCDD/Fs concentration, Tactual river water temperature on the day of sampling, Taverage timeweighted, average temperature over the 28 days prior to each sampling campaign, Q average discharge over the 28 days prior to each sampling campaign AR-mediated potency 5018 (Weiss et al. 2009), and the metabolite of DDT, p, p′-DDE (Urbatzka et al. 2007) were found in antiandrogenic fractions. Various compounds, including oxygenated PAHs, organophosphates, musks, and steroids, were detected in androgenic fractions (Weiss et al. 2011). A number of contaminants analyzed in this study, including some PAHs, PCBs, PCDD/Fs, and OCPs, have also been reported to be antiandrogenic (Vinggaard et al. 2008). Correlation and multivariate analysis The correlation profiles of bioassay results with environmental parameters and concentrations of measured residues are displayed as bivariate relationships (Fig. 4). Concentrations of BIOTEQ were significantly positively correlated with TOC, clay content, and flow and negatively with temperature even when the seasonal variability was taken into account. These correlations document a significant role of abiotic parameters in accumulation of dioxin-like compounds, which was demonstrated for TOC and clay also in a previous study (Hilscherova et al. 2010). The fine-grained fraction of sediment particles plays an important role in the accumulation of HOCs in sediments (Jaffe 1991). BIOTEQ was also correlated with concentrations of all studied classes of HOCs. The most significant correlation has been found with PAHs and TEQ derived from PAHs, which documents their important contribution to BIOTEQ. However, from the comparison of TEQ and BIOTEQ it was calculated that only a negligible portion of dioxin-like activity was attributed to dl-PCBs and PCDD/Fs. The correlation does not imply causal relationship but rather indicates that compounds with similar properties like measured HOCs were responsible for the observed AhRpotency of sediments. There was a significant negative correlation of concentrations of BIOTEQ with actual and average monthly temperature (Fig. 4). This corresponds with the greater concentrations of BIOTEQ observed during winter, which is probably related to slower rates of degradation of chemicals as well as greater PAHs inputs from local combustion during colder periods. Furthermore, concentrations of EEQ were significantly correlated with actual temperature (Fig. 4). Concentrations of PAHs might be partially reduced by microbial degradation that is greater during warmer months. Consequently, this could result in an increased estrogenic potency of sediments due to the formation of estrogenic metabolites, such as hydroxylated PAHs (Hayakawa et al. 2007; Luan et al. 2006; Wang et al. 2012). The opposite trend is observed during winter, because microbial degradation is lower at lower temperatures. Further, lesser dilution of (xeno)estrogens can be expected during warmer months due to the lesser discharge (Sumpter 2005; Figs. S3 and S5). However, no significant correlation between concentrations of EEQ and discharge was observed. Environ Sci Pollut Res (2014) 21:5007–5022 Antiandrogenic potency was significantly correlated with TOC (Fig. 4), which was also shown in a previous study (Hilscherova et al. 2010). Thus, relatively hydrophobic compounds are likely to contribute to the AA potency of extracts of sediments. However, no significant correlation was found between AA and concentrations of studied HOCs among locations and seasons (Fig. 4). The only correlations with AA were found with concentrations of p, p′-DDE at location MA and with both p, p′-DDE and p, p ′-DDD in sediments from CE, respectively. These DDT metabolites are considered as antiandrogenic compounds (Vinggaard et al. 2008). The data were further analyzed using multivariate PCA. Firstly, data from all localities and time points were included in the PCA. The first and second principal components (PC) accounted for 54 % of the total variance (40 and 14 %, respectively), and simplified the multivariate pattern which allowed the variables and samples to be projected onto a twodimensional space (Fig. 5). Variables with the main influence were TEQ, BIOTEQ and concentrations of measured HOCs in the direction of first PC, and EEQ and AA in the direction of second PC. Secondly, only locations from the active river channel were assessed and temperature (T actual) and discharge (Q ) of the river were included as active variables in PCA (Fig. 6).1 The first and second PC accounted for 52 % of the total variance (39 and 13 %, respectively). Variables with the main influence were concentrations of most classes of HOCs (excluding dl-PCBs) in one direction and T actual and Q in the other direction (Fig. 6a). The influence of EEQ and AA was not apparent anymore in this two-dimensional projection. AA was the dominant parameter associated with PC3, which explained 10 % of the total variance. AhR-mediated potency determined in bioassay (expressed as BIOTEQ) was clearly associated with concentrations of analyzed HOCs in the first PCA (Fig. 5a). However, if only locations from the active river channel were included, BIOTEQ was projected in the very same direction as HOCs along PC1 but somewhat separated by the direction along PC2 (Fig. 6a). This observation further supported the interpretation that the observed AhR-mediated potency of sediments cannot be fully explained by analyzed HOCs and there were other contaminants with similar properties contributing to the potency. In contrast, analyzed HOCs cannot explain concentrations of EEQ and AA that were projected in a different direction from concentrations of HOCs in both multivariate analyses (Figs. 5a and 6a). When all locations and time points were included in the analysis, the outcomes of specific bioassays used together with concentrations of the measured pollutants as active variables did not separate the sediments from different locations 1 Locality CR (oxbow lake) has no water discharge (lentic locality) and temperature was not measured, therefore, these two variables could not have been included in Fig. 5. Environ Sci Pollut Res (2014) 21:5007–5022 5019 Fig. 5 Principal component analysis (PCA) based on the data from all sampling sites. The ordination diagrams show the relationship among variables (a) and distribution of samples according to localities (b). Variables marked by full circles were used for creating PCA (active variables). Variables marked by empty circles are displayed in the same ordination space but they were not used for creating PCA (supplementary variables). Abbreviations as in Table 1 and Fig. 4 (Fig. 5b). Seasonal variability of contamination had a stronger influence on the distribution of variables and samples in PCA than the differences among locations. Results of different samplings from all locations were relatively overlapping and only individual samples from various locations were outliers. Only if T actual and Q were included as active variables in PCA, SP was obviously separated from the other locations in the direction of greater pollutant concentrations (Fig. 6a, b). In conclusion, seasonal changes play a dominant role and can be more important in the studied locations than spatial differences. This finding is consistent with the results of a previous study, which demonstrated no good separation of samples from several study regions in autumn compared to spring (Hilscherova et al. 2010). Concentrations of dioxin-like and estrogenic potencies measured in fluvial sediments from the three locations (MA, BE, and SP) (Table 2) during this study were compared to those of several previous studies (Hilscherova et al. 2010, 2002). Data from autumn (October) were available from 5 years between Fig. 6 Principal component analysis (PCA) based on data from sites in the active river channel (i.e., except CR) including also flow and temperature. The ordination diagrams show the relationship among variables (a) and distribution of samples according to localities (b). Variables marked by full circles were used for creating PCA (active variables). Variables marked by empty circles are displayed in the same ordination space but they were not used for creating PCA (supplementary variables). Abbreviations as in Table 1 and Fig. 4 Long-term trend analysis 5020 1996 and 2008, while for spring (May) from three different years (2005–2008), respectively. There was no continuous trend of changes in concentrations of BIOTEQ or EEQ that would indicate the decrease or increase of contamination in time. Rather, the long-term (inter-annual) differences corresponded well with seasonal fluctuations documented in the current study. Greater differences in potencies measured in the bioassays were observed among spring samples from different years while concentrations were more stable during autumn. Inter-annual as well as seasonal fluctuations were the least at location SP; maximally 4- and 2-fold differences were observed in case of concentrations of BIOTEQ and EEQ, respectively. This was probably related to the greater overall discharge and long-term greater contamination at this location. On the other hand, the greatest differences were found for location MA on river Drevnice (up to 11-fold for BIOTEQ), where discharge was relatively small and thus, fluctuations in discharge could have had larger effects. Both short- and longterm variability in contamination by estrogenic compounds were substantially greater than in the case of dioxin-like compounds. Inter-annual variation in concentrations of EEQ was greater than variation among seasons. As much as 95- and 51-fold difference in EEQ was observed at location MA and BE, respectively, when comparing situations between May 2005, 2006, and 2008, while 25- and 4-fold difference was observed within estrogenic potency of sediments from these two locations in spring 2008, respectively (Table 2). The greater differences on locations MA and BE are associated mainly with a strong decrease of EEQ (below limit of detection) in spring 2006, which is a result of local floods that occurred in the region (Hilscherova et al. 2010). Alternatively, differences in concentrations of EEQs in extracts of sediments from location SP were only 2-fold among spring and autumn samples across the studied years. The results of this 1-year study also show that concentrations of both BIOTEQ and EEQ were more variable in spring compared to autumn. This is probably related to the hydrology of the studied rivers. The discharge of the river was relatively less and stable in autumn 2007 (except for one major rainfall), whereas greater discharge with stronger fluctuations occurred in spring 2008 which can be linked to a greater resuspension of sediments (Fig. S3). A similar comparison of a smaller data set from sediments in a French river showed 3.6- and 5-fold inter-annual differences in dioxinlike and estrogenic potency, respectively (Creusot et al. 2013). Unlike in this study, lesser fluctuation was found in spring than in autumn. However, spring was described as dry season, whereas autumn as wet season in the French study, which differs from the hydrology situation in our study region (Fig. S3). This supports the conclusion that hydrology of the river is a very important parameter that needs to be taken into account in evaluation of river sediments contamination. Environ Sci Pollut Res (2014) 21:5007–5022 Conclusions The characterization of toxic potencies of environmental mixtures of pollutants might be an important step in the risk assessment of contaminated ecosystems allowing the assessment of potential risks connected with the exposure of organisms, next to comparing concentrations of selected contaminants with quality criteria or EQS. This study documents that the endocrine disruptive and dioxin-like potencies observed in sediments were not, respectively only to a minor extent, associated with routinely monitored hydrophobic organic pollutants. The contribution of PAHs, which were the predominant contaminants in the studied region, to the dioxin-like potency was 13–21 % across locations (median values). Despite the correlation between concentrations of dl-PCBs and PCDD/Fs with BIOTEQ, contribution of these contaminants to the dioxin-like potency was negligible as calculated based on their concentrations and relative potencies in the bioassay. Analyzed HOCs could not explain the observed estrogenic and antiandrogenic activities. The bioassays used in this study provided important information indicating the presence of yet unknown pollutants with dioxin-like and endocrine disruptive potencies in sediments. This 1-year long study of fluvial sediments also revealed seasonal differences in contamination with dioxinlike AA and androgenic compounds. Further, a long-term comparison of the unique data set originating from three locations point to a greater inter-annual fluctuations in estrogenic than dioxin-like potency. Both short-term and long-term data documents greater fluctuations in biological potencies as well as in river water discharge at the individual locations during spring season. Hence, hydrology of the river and its seasonal differences should be taken into account both in design and interpretation of any monitoring studies. Locations and time points need to be chosen carefully to make sure that the variability of contamination is not overlooked. In addition, to be able to monitor longterm trends in a region, it is necessary to sample in the same period of the year and under comparable hydrological situation. If this is not possible, the interpretation of results from long-term monitoring should be corrected to these factors. Acknowledgments This research was supported by projects ENVISCREEN (Ministry of Education, Youth and Sports of Czech Republic No. 2B08036) and CETOCOEN (CZ.1.05/2.1.00/01.0001) from the European Regional Development Fund. We acknowledge Klara Komprdova, Roman Prokes, and Ondrej Sanka for their technical assistance. Prof. Giesy was supported by the Canada Research Chair program, a Visiting Distinguished Professorship in the Department of Biology and Chemistry and State Key Laboratory in Marine Pollution, City University of Hong Kong, the 2012 “Great Level Foreign Experts” (#GDW20123200120) program, funded by the State Administration of Foreign Experts Affairs, the P.R. China to Nanjing University and the Einstein Professor Program of the Chinese Academy of Sciences. Environ Sci Pollut Res (2014) 21:5007–5022 References Babek O, Hilscherova K, Nehyba S, Zeman J, Famera M, Francu J, Holoubek I, Machat J, Klanova J (2008) Contamination history of suspended river sediments accumulated in oxbow lakes over the last 25 years. J Soils Sediments 8:165–176 Babich H, Borenfreund E (1990) Cytotoxic effects of food-additives and pharmaceuticals on cells in culture as determined with the Neutral Red Assay. J Pharm Sci 79:592–594 Babut M, Lopes C, Pradelle S, Persat H, Badot PM (2012) BSAFs for freshwater fish and derivation of a sediment quality guideline for PCBs in the Rhone Basin, France. J Soils Sediments 12:241–251 Behnisch PA, Hosoe K, Sakai S (2003) Brominated dioxin-like compounds: in vitro assessment in comparison to classical dioxin-like compounds and other polyaromatic compounds. Environ Int 29: 861–877 Brack W, Klamer HJC, de Ada ML, Barcelo D (2007) Effect-directed analysis of key toxicants in European river basins—a review. Environ Sci Pollut Res 14:30–38 Brack W, Blaha L, Giesy JP, Grote M, Moeder M, Schrader S, Hecker M (2008) Polychlorinated naphthalenes and other dioxin-like compounds in Elbe River sediments. Environ Toxicol Chem 27:519–528 Brinkmann M, Hudjetz S, Kammann U, Hennig M, Kuckelkorn J, Chinoraks M, Cofalla C, Wiseman S, Giesy JP, Schaffer A, Hecker M, Wolz J, Schuttrumpf H, Hollert H (2013) How flood events affect rainbow trout: evidence of a biomarker cascade in rainbow trout after exposure to PAH contaminated sediment suspensions. Aquat Toxicol 128:13–24 Colombo JC, Cappelletti N, Lasci J, Migoya MC, Speranza E, Skorupka CN (2006) Sources, vertical fluxes, and equivalent toxicity of aromatic hydrocarbons in coastal sediments of the Rio de la Plata Estuary, Argentina. Environ Sci Technol 40:734–740 Creusot N, Tapie N, Piccini B, Balaguer P, Porcher JM, Budzinski H, AitAissa S (2013) Distribution of steroid- and dioxin-like activities between sediments, POCIS and SPMD in a French river subject to mixed pressures. Environ Sci Pollut Res 20:2784–2794 Crommentuijn T, Sijm D, de Bruijn J, van den Hoop M, van Leeuwen K, van de Plassche E (2000) Maximum permissible and negligible concentrations for metals and metalloids in the Netherlands, taking into account background concentrations. J Environ Manage 60:121– 143 de Deckere E, De Cooman W, Leloup V, Meire P, Schmitt C, von der Ohe PC (2011) Development of sediment quality guidelines for freshwater ecosystems. J Soils Sediments 11:504–517 Demirpence E, Duchesne MJ, Badia E, Gagne D, Pons M (1993) Mvln cells—a bioluminescent Mcf-7-derived cell-line to study the modulation of estrogenic activity. J Steroid Biochem Mol Biol 46:355–364 Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy, Brussels, p 72 Directive 2008/105/EC of the European Parliament and of the Council of 16 December 2008 on environmental quality standards in the field of water policy, amending and subsequently repealing Council Directives 82/176/EEC, 83/513/EEC, 84/156/EEC, 84/491/EEC, 86/280/EEC and amending Directive 2000/60/EC of the European Parliament and of the Council, Brussels, p 14 European Commission (2012) Proposal for a Directive of the European Parliament and of the Council amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy 2011/0429 (COD), Brussels, p 35 Forstner U, Salomons W (2010) Sediment research, management and policy. J Soils Sediments 10:1440–1452 Forstner U, Heise S, Schwartz R, Westrich B, Ahlf W (2004) Historical contaminated sediments and soils at the river basin scale. J Soils Sediments 4:247–260 5021 Galluba S, Oehlmann J (2012) Widespread endocrine activity in river sediments in Hesse, Germany, assessed by a combination of in vitro and in vivo bioassays. J Soils Sediments 12:252–264 Hayakawa K, Onoda Y, Tachikawa C, Hosoi S, Yoshita M, Chung SW, Kizu R, Toriba A, Kameda T, Tang N (2007) Estrogenic/ antiestrogenic activities of polycyclic aromatic hydrocarbons and their monohydroxylated derivatives by yeast two-hybrid assay. J Health Sci 53:562–570 Higley E, Grund S, Jones PD, Schulze T, Seiler TB, Lubcke-von Varel U, Brack W, Wolz J, Zielke H, Giesy JP, Hollert H, Hecker M (2012) Endocrine disrupting, mutagenic, and teratogenic effects of upper Danube River sediments using effect-directed analysis. Environ Toxicol Chem 31:1053–1062 Hilscherova K, Kannan K, Kang YS, Holoubek I, Machala M, Masunaga S, Nakanishi J, Giesy JP (2001) Characterization of dioxin-like activity of sediments from a Czech river basin. Environ Toxicol Chem 20:2768–2777 Hilscherova K, Kannan K, Holoubek I, Giesy JP (2002) Characterization of estrogenic activity of riverine sediments from the Czech Republic. Arch Environ Contam Toxicol 43:175–185 Hilscherova K, Kannan K, Nakata H, Hanari N, Yamashita N, Bradley PW, McCabe JM, Taylor AB, Giesy JP (2003) Polychlorinated dibenzo-p-dioxin and dibenzofuran concentration profiles in sediments and flood-plain soils of the Tittabawassee River, Michigan. Environ Sci Technol 37:468–474 Hilscherova K, Dusek L, Kubik V, Cupr P, Hofman J, Klanova J, Holoubek I (2007) Redistribution of organic pollutants in river sediments and alluvial soils related to major floods. J Soils Sediments 7:167–177 Hilscherova K, Dusek L, Sidlova T, Jalova V, Cupr P, Giesy JP, Nehyba S, Jarkovsky J, Klanova J, Holoubek I (2010) Seasonally and regionally determined indication potential of bioassays in contaminated river sediments. Environ Toxicol Chem 29:522–534 Houtman CJ, Cenijn PH, Hamers T, Lamoree MH, Legler J, Murk AJ, Brouwer A (2004) Toxicological profiling of sediments using in vitro bioassays, with emphasis on endocrine disruption. Environ Toxicol Chem 23:32–40 Hunt JCR (2002) Floods in a changing climate: a review. Phil Trans R Soc A Math Phys Eng Sci 360:1531–1543 Jaffe R (1991) Fate of hydrophobic organic pollutants in the aquatic environment—a review. Environ Pollut 69:237–257 Janosek J, Hilscherova K, Blaha L, Holoubek I (2006) Environmental xenobiotics and nuclear receptors—interactions, effects and in vitro assessment. Toxicol in Vitro 20:18–37 Jarosova B, Blaha L, Vrana B, Randak T, Grabic R, Giesy JP, Hilscherova K (2012) Changes in concentrations of hydrophilic organic contaminants and of endocrine-disrupting potential downstream of small communities located adjacent to headwaters. Environ Int 45:22–31 Jobling S, Tyler CR (2003) Endocrine disruption in wild freshwater fish. Pure Appl Chem 75:2219–2234 Kannan K, Yun SH, Ostaszewski A, McCabe JM, Mackenzie-Taylor D, Taylor AB (2008) Dioxin-like toxicity in the Saginaw river watershed: polychlorinated dibenzo-p-dioxins, dibenzofurans, and biphenyls in sediments and floodplain soils from the Saginaw and Shiawassee rivers and Saginaw bay, Michigan, USA. Arch Environ Contam Toxicol 54:9–19 Kaplan S (2013) Review: pharmacological pollution in water. Crit Rev Environ Sci Technol 43:1074–1116 Kidd KA, Blanchfield PJ, Mills KH, Palace VP, Evans RE, Lazorchak JM, Flick RW (2007) Collapse of a fish population after exposure to a synthetic estrogen. Proc Natl Acad Sci U S A 104:8897–8901 Koh CH, Khim JS, Kannan K, Villeneuve DL, Senthilkumar K, Giesy JP (2004) Polychlorinated dibenzo-p-dioxins (PCDDs), dibenzofurans (PCDFs), biphenyls (PCBs), and polycyclic aromatic hydrocarbons (PAHs) and 2,3,7,8-TCDD equivalents (TEQs) in sediment from the Hyeongsan River, Korea. Environ Pollut 132:489–501 5022 Kukucka P, Audy O, Prokes R, Komprdova K, Klanova J (2010) Temporal and spatial trends of selected POPs in riverine sediments: What can we learn for assessment of risks associated with frequent flood events? Organohalogen Compd 72:134–137 Luan TG, Yu KSH, Zhong Y, Zhou HW, Lan CY, Tam NFY (2006) Study of metabolites from the degradation of polycyclic aromatic hydrocarbons (PAHs) by bacterial consortium enriched from mangrove sediments. Chemosphere 65:2289–2296 Luo JP, Lei BL, Ma M, Zha JM, Wang ZJ (2011) Identification of estrogen receptor agonists in sediments from Wenyu River, Beijing, China. Water Res 45:3908–3914 Machala M, Vondracek J, Blaha L, Ciganek M, Neca J (2001) Aryl hydrocarbon receptor-mediated activity of mutagenic polycyclic aromatic hydrocarbons determined using in vitro reporter gene assay. Mutat Res Genet Toxicol Environ Mutagen 497:49–62 Martinez-Gomez C, Lamoree M, Hamers T, van Velzen M, Kamstra JH, Fernandez B, Benedicto J, Leon VM, Vethaak AD (2013) Integrated chemical and biological analysis to explain estrogenic potency in bile extracts of red mullet (Mullus barbatus). Aquat Toxicol 134:1–10 Novak J, Jalova V, Giesy JP, Hilscherova K (2009) Pollutants in particulate and gaseous fractions of ambient air interfere with multiple signaling pathways in vitro. Environ Int 35:43–49 Oh SM, Choung SY, Sheen YY, Chung KH (2000) Quantitative assessment of estrogenic activity in the water environment of Korea by the E-SCREEN assay. Sci Total Environ 263:161–169 Peck M, Gibson RW, Kortenkamp A, Hill EM (2004) Sediments are major sinks of steroidal estrogens in two United Kingdom rivers. Environ Toxicol Chem 23:945–952 Prokes R, Vrana B, Klanova J (2012) Levels and distribution of dissolved hydrophobic organic contaminants in the Morava river in Zlin district, Czech Republic as derived from their accumulation in silicone rubber passive samplers. Environ Pollut 166:157–166 Song MY, Jiang QT, Xu Y, Liu HX, Lam PKS, O’Toole DK, Zhang QH, Giesy JP, Jiang GB (2006) AhR-active compounds in sediments of the Haihe and Dagu Rivers, China. Chemosphere 63:1222–1230 Streck G (2009) Chemical and biological analysis of estrogenic, progestagenic and androgenic steroids in the environment. Trac Trends Anal Chem 28:635–652 Sumpter JP (2005) Endocrine disrupters in the aquatic environment: an overview. Acta Hydrochim Hydrobiol 33:9–16 Urbatzka R, van Cauwenberge A, Maggioni S, Vigano L, Mandich A, Benfenati E, Lutz I, Kloas W (2007) Androgenic and antiandrogenic activities in water and sediment samples from the river Lambro, Italy, detected by yeast androgen screen and chemical analyses. Chemosphere 67:1080–1087 Environ Sci Pollut Res (2014) 21:5007–5022 Vermeirssen ELM, Korner O, Schonenberger R, Suter MJF, BurkhardtHolm P (2005) Characterization of environmental estrogens in river water using a three pronged approach: active and passive water sampling and the analysis of accumulated estrogens in the bile of caged fish. Environ Sci Technol 39:8191–8198 Vigano L, Benfenati E, van Cauwenberge A, Eidem JK, Erratico C, Goksoyr A, Kloas W, Maggioni S, Mandich A, Urbatzka R (2008) Estrogenicity profile and estrogenic compounds determined in river sediments by chemical analysis, ELISA and yeast assays. Chemosphere 73:1078–1089 Vinggaard AM, Niemela J, Wedebye EB, Jensen GE (2008) Screening of 397 chemicals and development of a quantitative structure-activity relationship model for androgen receptor antagonism. Chem Res Toxicol 21:813–823 Vondracek J, Machala M, Minksova K, Blaha L, Murk AJ, Kozubik A, Hofmanova J, Hilscherova K, Ulrich R, Ciganek M, Neca J, Svrckova D, Holoubek I (2001) Monitoring river sediments contaminated predominantly with polyaromatic hydrocarbons by chemical and in vitro bioassay techniques. Environ Toxicol Chem 20: 1499–1506 Wang X, Lin L, Luan T, Yang L, Tam NFY (2012) Determination of hydroxylated metabolites of polycyclic aromatic hydrocarbons in sediment samples by combining subcritical water extraction and dispersive liquid-liquid microextraction with derivatization. Anal Chim Acta 753:57–63 Weiss JM, Hamers T, Thomas KV, van der Linden S, Leonards PEG, Lamoree MH (2009) Masking effect of anti-androgens on androgenic activity in European river sediment unveiled by effect-directed analysis. Anal Bioanal Chem 394:1385–1397 Weiss JM, Simon E, Stroomberg GJ, de Boer R, de Boer J, van der Linden SC, Leonards PEG, Lamoree MH (2011) Identification strategy for unknown pollutants using high-resolution mass spectrometry: androgen-disrupting compounds identified through effectdirected analysis. Anal Bioanal Chem 400:3141–3149 Wilson VS, Bobseine K, Lambright CR, Gray LE (2002) A novel cell line, MDA-kb2, that stably expresses an androgen- and glucocorticoid-responsive reporter for the detection of hormone receptor agonists and antagonists. Toxicol Sci 66:69–81 Wolz J, Schulze T, Lubcke-von Varel U, Fleig M, Reifferscheid G, Brack W, Kuhlers D, Braunbeck T, Hollert H (2011) Investigation on soil contamination at recently inundated and non-inundated sites. J Soils Sediments 11:82–92 Zhao JL, Ying GG, Yang B, Liu S, Zhou LJ, Chen ZF, Lai HJ (2011) Screening of multiple hormonal activities in surface water and sediment from the pearl river system, South China, using effectdirected in vitro bioassays. Environ Toxicol Chem 30:2208–2215 Fig. S1 Spatial and seasonal variability of total organic carbon (TOC, %) in sediment samples from the 15 sampling campaigns in July 2007–July 2008 (n=73). Middle line is median, box means quartile range (25-75%), whisker is non-outlier range, circles are outliers, stars are extremes and triangles are measured values Fig. S2 Spatial variability of bioassay derived dioxin-like potency (BIOTEQ, pg/g, dm) in each season derived from the 15 sampling campaigns in July 2007–July 2008 (n=73). Middle line is median, box means quartile range (25-75%), whisker is non-outlier range and triangles are measured values a summer spring autumn spring winter summer autumn Flow (m3.s-1) 300 200 1 Kromeriz 15 Spytihnev 100 1.12.2008 1.11.2008 1.9.2008 1.10.2008 1.8.2008 1.7.2008 1.6.2008 1.5.2008 1.4.2008 1.3.2008 1.2.2008 1.1.2008 1.12.2007 1.11.2007 1.10.2007 1.9.2007 1.8.2007 1.7.2007 1.6.2007 1.5.2007 1.4.2007 1.3.2007 1.2.2007 1.1.2007 0 b 25 spring autumn summer winter autumn summer spring Flow (m3.s-1) 20 15 1 10 Zlin 15 5 1.12.2008 1.11.2008 1.9.2008 1.10.2008 1.8.2008 1.7.2008 1.6.2008 1.5.2008 1.4.2008 1.3.2008 1.2.2008 1.1.2008 1.12.2007 1.11.2007 1.10.2007 1.9.2007 1.8.2007 1.7.2007 1.6.2007 1.5.2007 1.4.2007 1.3.2007 1.2.2007 1.1.2007 0 Fig. S3 Daily water discharge at gauging stations a Kromeriz (representative for site BE) and Spytihnev (SP) on Morava River and b Zlin on Drevnice River (representative for site MA). The numbers 1 and 15 show the dates of first and last (15th) sampling campaigns: 1 – June 20, 2007; 15 – July 16, 2008. Fig. S4 Spatial and seasonal variability of predicted (chemically-derived) dioxin-like potency (TEQ, pg/g, dm) in sediment samples derived from the 15 sampling campaigns in July 2007– July 2008 (n=73).. Middle line is median, box means quartile range (25-75%), whisker is nonoutlier range and triangles are measured values a spring Temperature (°C) 25 summer autumn winter spring summer autumn 20 15 1 10 Kromeriz Spytihnev 15 5 1.12.2008 1.11.2008 1.9.2008 1.10.2008 1.8.2008 1.7.2008 1.6.2008 1.5.2008 1.4.2008 1.3.2008 1.2.2008 1.1.2008 1.12.2007 1.11.2007 1.10.2007 1.9.2007 1.8.2007 1.7.2007 1.6.2007 1.5.2007 1.4.2007 1.3.2007 1.2.2007 1.1.2007 0 b spring Temperature (°C) 25 summer autumn winter spring summer autumn 20 15 Zlin 10 1 15 5 1.12.2008 1.11.2008 1.9.2008 1.10.2008 1.8.2008 1.7.2008 1.5.2008 1.6.2008 1.4.2008 1.3.2008 1.2.2008 1.1.2008 1.12.2007 1.11.2007 1.10.2007 1.9.2007 1.8.2007 1.7.2007 1.6.2007 1.5.2007 1.4.2007 1.3.2007 1.2.2007 1.1.2007 0 Fig. S5 Average daily water temperature (°C) at gauging stations a Kromeriz (representative for site BE) and Spytihnev (SP) on Morava River and b Zlin on Drevnice River (representative for site MA). The numbers 1 and 15 show the dates of first and last (15th) sampling campaigns: 1 – June 20, 2007; 15 – July 16, 2008. Table S1 Sampling campaigns in each sampling season (samples were clustered according to four hydrologically defined seasons) Campaign no. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Sampling date 2007-06-20 2007-07-18 2007-08-15 2007-09-12 2007-10-10 2007-11-07 2007-12-05 2008-01-02 2008-01-30 2008-02-27 2008-03-26 2008-04-23 2008-05-21 2008-06-18 2008-07-16 Season Summer Autumn Winter Spring Summer The sampling times from previous studies are listed in Table 2. Table S2 Relative potencies (REP) of aryl hydrocarbon receptor (AhR) activation determined in the test with H4IIE-luc cells (24 h exposure) Compound PAHsa Fluoranthene Pyrene Benzo(a)anthracene Chrysene Benzo(b)fluoranthene Benzo(k)fluoranthene Benzo(a)pyrene Indeno (123cd)pyrene Dibenzo(ah)anthracene PCBsb PCB 77 PCB 81 PCB 126 PCB 169 PCB 105 PCB 114 PCB 123 PCB 156 PCB 157 PCB 167 PCB 189 a Machala et al. (2001) b Behnisch et al. (2003) REP 2.27×10-8 1.78×10-6 7.04×10-6 1.01×10-4 3.35×10-5 1.64×10-3 9.01×10-5 2.96×10-4 1.17×10-3 1.30×10-3 4.20×10-3 6.70×10-2 3.40×10-3 1.20×10-5 4.80×10-5 2.40×10-5 2.10×10-4 8.00×10-5 8.20×10-6 6.70×10-6 Compound REP PCDDsb 2,3,7,8-TCDD 1,2,3,7,8-PeCDD 1,2,3,4,7,8-HxCDD 1,2,3,6,7,8-HxCDD 1,2,3,7,8,9-HxCDD 1,2,3,4,6,7,8-HpCDD OCDD 1 0.54 0.30 0.14 6.60×10-2 4.60×10-2 5.00×10-4 PCDFsb 2,3,7,8-TCDF 1,2,3,7,8-PeCDF 2,3,4,7,8-PeCDF 1,2,3,4,7,8-HxCDF 1,2,3,6,7,8-HxCDF 1,2,3,7,8,9-HxCDF 2,3,4,6,7,8-HxCDF 1,2,3,4,6,7,8-HpCDF 1,2,3,4,7,8,9-HpCDF OCDF 0.32 0.21 0.50 0.13 3.90×10-2 0.11 0.18 2.90×10-2 4.10×10-2 6.50×10-3 Table S3 Sediment quality guidelines derived by de Deckere et al. (2011) Naphthalene Acenaphthylene Acenaphthene Fluorene Phenanthrene Anthracene Fluoranthene Pyrene Benz[a]anthracene Chrysene Benzo[b]fluoranthene Benzo[k]fluoranthene Benzo[a]pyrene Indeno[123cd]pyrene Dibenz[ah]anthracene Benzo[ghi]perylene PCB 28 PCB 52 PCB 101 PCB 118 PCB 153 PCB 138 PCB 180 p,p'-DDE p,p'-DDD HCB Consensus 1 value (µg/kg dry mass of sediment) 200 30 40 40 180 30 250 240 120 150 170 80 140 120 20 110 0.04 0.1 0.54 0.43 1.5 1 0.44 0.31 0.06 0.0004 Consensus 2 value (µg/kg dry mass of sediment) 6600 5200 3300 260 890 170 1200 940 600 830 660 320 600 480 120 450 2 4.6 6.7 6.9 9.7 7.5 5.5 6.8 3.2 0.72