High-speed Imaging and Analysis of the... Undercooled Alloy Melts
advertisement

High-speed Imaging and Analysis of the Solidification of
Undercooled Alloy Melts
by
John W. Lum
S.B., Massachusetts Institute of Technology (1994)
Submitted to the Department of Materials Science and Engineering
in Partial Fulfillment of the Requirements for the Degree of
MASTER OF SCIENCE
in Materials Science and Engineering
at the
Massachusetts Institute of Technology
June, 1996
© Massachusetts Institute of Technology 1996
All rights reserved
Signature
of Author.....
...................
rid Engineering
May 10, 1996
Certified
by.
(t
Accepted
Merton C. Flemings
-"-: Supervisor
by.........................
Michael F. Rubner
TDK Professor of Materials Science and Engineering
Chair, Departmental Committee on Graduate Students
OF "--ECHNOLOG
JUN 2 41996
uLBRARES
Science
High-speed Imaging and Analysis of the Solidification of Undercooled
Alloy Melts
by
John W. Lum
Submitted to the Department of Materials Science and Engineering
on May 10, 1996 in partial fulfillment of the requirements for the
degree of Master of Science in Materials Science and Engineering.
ABSTRACT
Rapid solidification of undercooled pure nickel and hyperperitectic Fe-(10-30) wt.%
Ni alloys has been imaged at sufficiently high spatial resolution (64 x 64 pixels) and
temporal resolution (40,500 frames/sec) to observe interfacial shape and solidification
velocities exceeding 45 m/s. Imaging was of 8 gram, quartz-fluxed melts at undercoolings
between 70 K and 300 K. Dendrite velocities within the melt were calculated from the
surface velocities observed employing a simple geometric model of growth. Solidification
was found to proceed invariably from a single nucleation point at every composition. In
the case of Fe-Ni, primary solidification of both the equilibrium FCC phase and the
metastable BCC phase was observed, with phase selection dependent upon the thermal
history, composition, presence of heterogeneous nucleants, and degree of undercooling
attained. In all cases of primary BCC solidification, a subsequent transition to the FCC
phase was observed, resulting in a two-stage "double recalescence" evident in the thermal
and video records. A primary solidification map was generated to predict the preferred
phase for primary solidification as a function of composition and nucleation temperature for
similar experiments with quartz-fluxed Fe-Ni specimens.
The computer-based growth model enabled determination of solidification velocities
with much greater precision than had been previously accomplished using pyrometric
techniques alone. Growth velocity in pure Ni was found to follow an approximate powerlaw relationship with respect to undercooling up to some critical value AT*, where 150 K <
AT* < 180 K. Growth velocities in BCC Fe-(10-12) wt.% Ni followed a similar powerlaw relationship, but no velocity transition was observed despite undercoolings of nearly
250 K. However, both pure Ni and Fe-Ni specimens did exhibit a secondary transition in
solidification interface morphology. At smaller undercoolings, interface curvature was
discontinuous, with tendencies toward growth along preferred crystallographic directions.
At larger undercoolings, interfaces were completely smooth, with no discontinuity in
curvature caused by irregular growth. Values for the morphological AT* were estimated at
160-170 K for pure Ni and 180-190 K for intermediate Fe-Ni compositions.
Thesis Supervisor:
Merton C. Flemings
Title:
Toyota Professor of Materials Processing
TABLE OF CONTENTS:
1. Introduction
2. Literature Survey
2.1 Pure Nickel
2.1.1 Methods of Investigation
2.1.2 Experimental Results
2.2 Iron-Nickel
8
10
10
10
12
13
2.2.1 Metastable Phase Solidification
2.2.2 Experimental Results
13
15
3. Experimental Procedure
20
3.1 Specimen Preparation
20
3.2 Pyrometry
20
3.2.1 Data Acquisition
3.2.2 Data Analysis
3.3 Image Acquisition and Analysis
3.3.1 Acquisition
3.3.2 Analysis
3.4 Solidification Model
3.4.1 Assumptions
3.4.2 Operation
21
21
23
23
23
26
26
26
4. Results and Analysis
34
4.1 General Observations
34
4.1.1 Multiple Nucleation
4.1.2 Interfacial Morphology
34
34
4.2 Lens and Wetting Effects
35
4.3 Solidification Model Behavior
35
4.3.1 Iterative Convergence
4.4 Pure Ni Results
4.4.1 Solidification Velocity
4.4.2 Interfacial Morphology
4.5 Fe-Ni Alloy Results
4.5.1 Pyrometric Thermal Profiles
4.5.2 Pixel Intensity Profiles
36
38
38
38
39
39
40
4.5.3 Recalescence Behavior
4.5.4 Interfacial Morphology
4.5.5 Solidification Velocity
5. Conclusions
41
43
43
59
5.1 General
59
5.2 Pure Nickel
59
5.3 Iron-Nickel
60
6. Suggestions for Future Work
61
Appendices
62
Al. Solidification Theory
62
A2. Tabulated Thermodynamic Data
68
A2. Tabulated Experimental Data
69
A3. Computer Code
77
TABLE OF FIGURES:
Figure 2.1: Typical pyrometric method to determine solidification velocity,
16
Figure 2.2: Selected prior investigations of pure Ni. References for plotted data: Bassler,
et al [8]; Piccone, et al [4]; Walker [2]; Hofmeister, et al [6]; Schleip, et al [12];
17
Colligan and Bayles [3].
Figure 2.3: Fe-Ni equilibrium phase diagram with calculated metastable extensions
18
Figure 2.4: Prior velocity calculations for Fe-Ni alloys performed by Barth, et al [21]. A
critical undercooling of 175 K was identified for both compositions after comparison
19
with LKT predictions.
Figure 3.1: Schematic of apparatus used for pyrometric data and image acquisition.
28
Figure 3.2: Detail of experimental setup, showing specimen position and geometry during
processing.
29
Figure 3.3: Typical ratio-temperature curve obtained during slope-calibration testing for
pure Ni. Included at left are the time-ratio and time-temperature data for the specimen
which yielded the ratio-temperature plot. In this case, a best-fit linear analysis of the
data gives a ratio-temperature slope of 880 K.
30
Figure 3.4: Determination of solidification interface. At left is single image from
recalescence event with computer-defined interface shown in black and newly- formed
solid bright at top. At right is complete sequence of interfaces from same
recalescence event.
31
Figure 3.5: Cross-section of solidifying specimen, with successive model interfaces
shown at constant time intervals following nucleation.
32
Figure 3.6: Raster motion during image acquisition at 40,500 frames/sec. Raster block
begins at top left and scans from left to right, top to bottom in four passes, reaching
bottom right after slightly less than 1/40,500 sec.
33
Figure 4.1: Schematic of apparent multiple nucleation scenario, observed experimentally in
Figure 4.2.
45
Figure 4.2: Example of solidification interface propagating from back to front, almost
directly toward the camera lens. System is Ni-25%Sn, -25 ms between images.
Dendrite tips intersecting front surface appear as numerous expanding diamonds with
the same axial orientation.
46
Figure 4.3: Composite interface image showing front-surface nucleation; initial interface at
upper-right exhibits very small radius of curvature. Note also raster pattern effect at
1/4, 1/2, and 3/4 of the image height causing apparent discontinuities in interface
curvature.
47
Figure 4.4: Calibration image of oil-soaked millimeter graph paper rolled inside 11 x 13
mm quartz tubing.
48
Figure 4.5: Model vertical rules (black) overlaying observed rules (gray, traced from
Figure 4.4), verifying that the lens and wetting effects are well-modeled by assuming
a 14% increase in specimen radius.
49
Figure 4.6: Solidification velocity plotted versus undercooling for pure Ni, as determined
by this work. Also shown is LKT prediction with and without modification by a
kinetic undercooling parameter.
50
Figure 4.7: Interface morphology transition for pure Ni. Shown are typical interface
morphologies observed at undercoolings less than the critical range of 160-170 K
(left column) and greater than the critical range (right).
51
Figure 4.8: Typical thermal profiles for Fe-10 wt.% Ni specimens undergoing single
recalescence only (top) and double recalescence (bottom). Note superheat peak at top
and double-plateau behavior at bottom.
52
Figure 4.9: Pyrometer profile for Fe-10 wt.% Ni specimen which was revealed in the
video record to have undergone double recalescence. No clear indication is present in
the thermal record.
53
Figure 4.10: Comparison between pyrometer profile and pixel intensity profile for Fe-10
wt.% Ni specimen undergoing double recalescence. Details are much more sharplydefined in the pixel intensity profile.
54
Figure 4.11: Solidification behavior in Fe-(10-30) wt.% Ni.
55
Figure 4.12: Delay time between recalescence events in Fe-10- and Fe-12 wt.% Ni
specimens.
56
Figure 4.13: Interface morphology at various compositions and undercoolings. A
transition from jagged to smooth structure appears at approximately 190 K
undercooling.
57
Figure 4.14: Solidification velocities for BCC Fe-10- and Fe-12 wt.% Ni.
58
Acknowledgements
My deepest and most heartfelt gratitude goes once again to my parents, without
whose upbringing I would not be nearly so satisfied with the person I am today. You are
still the reader I'm writing for, Mom.
I also owe a great deal to Prof. Merton Flemings for supervising my thesis work.
He has, during the last two years, provided just enough guidance to keep me moving in the
right direction, while simultaneously giving me the opportunity to make the majority of my
research-related decisions for myself. I have always felt very trusted during my time under
Prof. Flemings' supervision.
On a somewhat more personal note, I would like to thank Lisa for remaining a rock
of support for me through the difficulty we've experienced in the past few months. My
twin brother David has also been a steady companion during my graduate work, as he has
been as far back as I can remember (and probably beyond). We may not have many MIT
lunches left before one of us finally strikes out into the Real World! No less important in
the past few years were the fun times I spent with Jeanie, Noland, and my other friends
around the Institute and elsewhere.
Thanks is also due to my many colleagues here at MIT, both past and present. In
particular, Doug Matson has been a constant source of support since I first set foot in room
8-436. I also owe a great deal to Dr. Tom Piccone, both for his crucial help with the
experimental apparatus and also for his fine examples of thorough research dissertations
which will continue to assist MIT solidification researchers for many years to come. In
addition, I'd like to recognize Matthieu, Cars, Qi, Jusuf, Arvind, Chris, Rob, Hua Shen,
Honjo, and Elizabeth for offering their help and friendship to me along the way. I'd also
like to thank my youngest colleagues, undergraduates Rob and Elissa, for providing their
capable assistance during the second year of this work.
Finally, I gratefully acknowledge the sponsorhip of the National Aeronatics and
Space Administration during these two successful years of research.
1. Introduction
One of the most interesting solidification phenomena to undergo extensive study in
the last 30 years is the rapid solidification behavior of pure elements and alloy melts.
Rapidly-solidified materials have proved to offer a wide range of desirable materials
properties not exhibited by their conventionally-cast counterparts.
For specimens
exhibiting at least one thin dimension, numerous rapid solidification techniques have been
developed which rely upon ultra-high rates of heat extraction (109-1012 K/s); examples
include melt-spinning, splat-quenching, ion bombardment, laser-pulse heating, and many
others.
These methods, however, cannot be used in the production of large-scale ingots in
the foundry, because the absence of a thin dimension makes it impossible to obtain the
requisite heat-extraction rates. One alternative approach to produce rapid solidification in
these cases is to achieve a large undercooling of the liquid melt prior to solidification.
Undercooling techniques enable rapid solidification by lowering the melt
temperature well below the liquidus prior to solidification, gradually removing a significant
portion of the ingot's sensible heat. The resulting rapid solidification is caused by the
increased thermodynamic driving force inherent to the highly-undercooled liquid, and a
large portion of the solidification process can be made to occur in an essentially adiabatic
manner, a phenomenon known as "recalescence."
The undercooling phenomenon has been under considerable study for decades, and
an important parameter of characterization has been the velocity with which the newlyformed solid propagates through the undercooled liquid during the solidification process.
Prior film-based and electronic methods of measuring solidification velocities have met
with variable success.
In general, investigators have achieved either high temporal
resolution (strategically-placed photodiodes), or high spatial resolution (conventional film-
based cameras)--but not both.
As a result, the velocity measurements from these
experiments have suffered from a large degree of scatter.
It is only recently that electronic imaging technology has developed to the point
where there exists sufficient temporal resolution (up to 40,500 Hz with the system
employed in this work) and spatial resolution (a 64 x 64 pixel image) to generate a complete
visual record of the rapid solidification of a highly-undercooled specimen. This record, if
properly interpreted, can be used to obtain the most accurate velocity measurements yet
performed in this type of experiment.
In the current work, an original computer model has been developed to analyze the
photographic records of rapid solidification events and measure the propagation velocity of
the solid/liquid interface. The method is first applied to melts of high-purity nickel--an
extensively-studied material--in order to demonstrate the validity and accuracy of the
technique. Analysis is subsequently extended to the solidification of binary Fe-Ni alloys,
with special emphasis placed upon the exploration of the metastable 8-phase and related
double-recalescence phenomenon.
2. Literature Survey
2.1 Pure Nickel
2.1.1 Methods of Investigation
Numerous investigators have studied dendrite growth velocities in undercooled
high-purity elements, particularly nickel, since the undercooling phenomenon was first
observed in the Au system by Van Riemsdyk [1] in 1880. Walker [2] made the first
thorough investigation of undercooled nickel melts using photodiodes to measure the time
required for an interface to propagate along a column of molten material. Colligan and
Bayles [3] used photodiodes in conjunction with high-speed cinematography, determining
surface solidification morphology in addition to solidification velocities
Subsequent velocity studies have primarily involved refinements in the placement of
photodiodes and interpretation of the thermal record.
Piccone, et al [4], for example,
employed a single photodiode focused on a portion of an undercooled, quartz-encased
nickel ingot in conjunction with a digital oscilloscope. Velocities were determined using
the relationship
V=
At'
(2.1)
where D is the diameter of the pyrometer view field and At is the signal "rise time" for the
recalescence, as depicted in Figure 2.1.
This technique-like any technique that does not
directly image the solidification interface--suffers from several inherent uncertainties
regarding both D and At: first, the D value is an inaccurate measure of the distance traveled
by the solidification interface unless it is certain that growth proceeded directly across the
field of view of the pyrometer. In addition, a large degree of interpretation is required to
derive a At value from a given thermal record; the sigmoidal shape of the temperature
transition makes it possible to determine At consistently, but not precisely.
Other researchers
have employed
electromagnetic
levitation
(containerless
processing) to study undercooled Ni melts. One such example is the work of Willnecker,
et al [5], who used two hemispherical photodiodes and calculated solidification velocities
using signal rise time and geometric considerations. Hofmeister, et al [6], used a single,
tightly-focused (1 mm) photodiode, supposedly to minimize the effects of potential multiple
nucleation events. In the latter investigation, the reduced pyrometer spot size may have
mitigated the effect of the unconstrained solidification path.
In the former study,
measurement accuracy may have been improved by constraining the solidification path via
manually-induced nucleation at the south pole of the specimen.
However, in both
investigations, the results of the work again depend upon the interpretation of sigmoidal
thermal profiles.
Eckler and Herlach [7] used a polar photodiode and a unique capacitance trigger in
their work, defining more accurately the duration of the solidification event. In this study,
the moment of nucleation was marked by a jump in the capacitance of a metallic stimulation
needle upon contact with the base of the specimen. As a result, pyrometry was only
required to detect the end of the solidification event.
Bassler, et al [8], were among the first to use a linear array of photodiodes--later
moving to a two-dimensional array--in an effort to improve the spatial resolution of their
studies. An iterative computer routine was employed in conjunction with the photodiode
records to produce a calculated velocity associated with minimum deviation from the
observed data. The work was also based upon the assumption that the solid/liquid interface
remains convex to the liquid at all undercoolings; deviations were assumed to result from
multiple nucleation events, and all such trials were thrown out. However, both the current
investigation and prior work [3] have shown numerous deviations from the convex-
interface restriction which clearly result from a single nucleation point. As a result, the
validity of Bassler's "multiple nucleation" assumption is in question.
2.1.2 Experimental Results
With the exception of Hofmeister and Bassler, the results of the above work are
qualitatively and quantitatively similar (Figure 2.2).
At small undercoolings, the
investigators show an approximate power-law relationship between undercooling and
solidification velocity,
V = k (AT),
(2.2)
where 13 varies between 2 and 3. This behavior is relatively well-predicted by the "LKT"
theory of Lipton, et al [9], particularly when a kinetic parameter is included in the analysis.
At larger undercoolings, the results of prior investigations diverge more sharply, but many
conclude that there exists some critical undercooling, AT*, in the range 170-190K, above
which a normal LKT analysis is no longer valid.
In addition, several researchers [10,11,12] have observed a corresponding
microstructural transition at AT* in a similar range of values, marking the shift from a
columnar dendritic grain structure to a finer, equiaxed structure.
Notable also is the
transition in solidification interface morphology identified by Colligan and Bayles in their
high-speed cinematographic work. It was determined that surface interfaces displayed
continuous curvature at undercoolings greater than 160 K, but shifted to a discontinuous,
angular morphology as undercooling fell below 145 K.
The scatter of undercooling-velocity data at undercoolings above AT* is quite
remarkable, bounded at the lower end by the results of Hofmeister and Bassler, who report
a constant velocity of about 20 m/s at all undercoolings larger than AT*. They propose that
interfacial attachment kinetics is the limiting factor at these large undercoolings, while no
satisfactory explanation has yet been proposed to explain the relationships observed by the
other investigators.
Scatter at undercoolings less than AT* is much less pronounced than at higher
undercoolings and may be explained by variations in convective effects and methods of
pyrometry, deviations from assumed specimen geometry, and, as supported by Eckler, et
al [7], uncertainty regarding the growth direction of the dendrites.
2.2 Iron-Nickel
2.2.1 Metastable Phase Solidification
One of the first observations of the tendency of hyperperitectic Fe-Ni alloys to
solidify into more than one crystalline phase was made by Cech [13] in 1956. It was found
that micron-range particles of Fe-29.5 at.% Ni cooled from the liquid state formed not only
the expected equilibrium FCC phase upon solidification, but also frequently solidified into
an unexpected BCC phase. A simple graphical extension of the liquidus and solidus lines
of the (BCC) 8 phase on the equilibrium Fe-Ni phase diagram (Figure 2.3) supported the
hypothesis that molten droplets of Fe-Ni undercooled below these extensions could solidify
into a metastable form of 8 Fe-Ni.
2.2. 1. 1 Theoretical Basis
Kelly, et al [14] presented a strong theoretical argument for the solidification of
alternative (or nonequilibrium) crystalline phases from an undercooled melt based on the
thermodynamics and kinetics of nucleation and growth.
Numerical results for Fe-Ni
showed that the metastable phase was indeed preferred for homogeneous nucleation under
a wide range of particle sizes, cooling rates, and compositions.
In the case of
heterogeneous nucleation, wetting angle and other variables entered the analysis, but
primary metastable phase solidification remained a theoretical possibility.
2.2.1.2 Powder Studies
Subsequent powder studies have continued to reveal metastable phase formation
from the melt in Fe-Ni alloys. Kim, et al [15] reported development of the alternative BCC
phase in submicron droplets of Fe-30-, and Fe-40 at.% Ni, but not in Fe-50 at.% Ni.
Thoma, et al [16] (an excellent review paper on this topic) found BCC solidification in a
wide compositional range, proposing a map to define the structural evolution of solidified
Fe-Ni as a function of both composition and undercooling.
Although Libera, et al [17] managed to obtain thermal profiles of 44-46 Rm Fe-30
at.% Ni particles upon solidification, they were unable to salvage the individual particles
and thus did not perform any microstructural evaluation. Also, the temporal resolution of
their pyrometry was insufficient to reveal potential two-stage recalescence behavior.
2.2.1.3 Bulk Studies
Investigations of bulk Fe-Ni specimens have further characterized metastable BCC
phase solidification. The Thoma study reported no retained metastable BCC structure in
larger (1-3 mm) droplets of Fe-(10 to 30) wt.% Ni; however, it was speculated that the
cooling rate in these droplets was insufficient to suppress a solid state BCC-->FCC
transformation, and that the initial solid phase to form from the undercooled melt may well
have been the metastable 8 phase.
Stronger evidence for the proposed transition was supplied by Zhao, et al [18] and
Herlach, et al [19], who recorded fast thermal profiles of bulk Fe-Ni solidification events.
These efforts revealed a double-recalescence behavior for certain hyperperitectic Fe-Ni
specimens at sufficient undercoolings: initial growth of BCC material was followed by a
plateau period near the metastable BCC solidus temperature lasting from about 1 ms to
several seconds, followed ultimately by a transformation to FCC material from either a
liquid/solid mixture or the solid state. These thermal profiles, in agreement with theoretical
work by Chuang, et al [20] which more accurately defined the metastable liquidus and
solidus lines for 8 Fe-Ni, provided an exceedingly strong indication that the metastable 8
phase was solidifying directly from the undercooled melt.
2.2.2 Experimental Results
In general, the microstructure and solidification velocities observed in undercooled
Fe-Ni melts resemble the results of the pure Ni work. The undercooling-velocity data from
Barth, et al [21] for Fe-25- and Fe-30 at.% Ni are summarized in Figure 2.4; the behavior
is reasonably well modeled by modified LKT analyses for the equilibrium FCC phase. The
investigators argued that the data in Figure 2.4 reflects the existence of a critical
undercooling (AT*) for both compositions at 175 K--similar to that of pure Ni-although
this appears to be a more valid assessment of the lower data set. Above AT*=175 K, it
was surmised that dendrite growth theory no longer offered an accurate prediction of the
AT-V correspondence, indicating some shift in solidification mechanism. It is worth noting
that this study assumed primary solidification of the equilibrium FCC phase in Fe-25- and
Fe-30 at.% Ni. The Zhao study found FCC solidification in Fe-30 wt.% Ni, but primary
BCC solidification in Fe-5- and Fe-10 wt.% Ni specimens.
Other investigations identified a microstructural AT* which marked a transition from
a dendritic structure below AT* to a much finer-grained, "spherical" structure at higher
undercoolings. Early work by Kattamis, et al [22] indicated a AT* value of 170 K for Fe25 wt.% Ni. Abbaschian, et al [23] found a similar value of 175 K for that alloy, while the
Barth study reported a microstructural AT* between 135 K and 180 K for a range of Fe-Ni
compositions.
One final experimental result of importance is the demonstration of phase selection
or "phase seeding" in Fe-Ni through the use of a heterogeneous nucleation trigger of high
catalytic potency. Herlach, et al [19], were able to induce solidification of the metastable
BCC phase in Fe-(20-30) at.% Ni by surface stimulation with a trigger wire composed of
BCC Fe 92 Mo 8 . Identical specimens displayed primary FCC solidification when allowed to
recalesce spontaneously from similar or greater undercoolings.
This result reveals the
potential for reliable phase selection in this and other alloy systems through the use of
carefully-selected heterogeneous nucleants.
Specimen
Pyrometer
Specimen
"r
r
U-
(I)
a)
4.
Time
Figure 2.1: Typical pyrometric method to determine solidification velocity,
where V = D / At.
I I I I - I I I I I I I I I I I I I --1
5
I
r I
O Bassler
x Piccone
I
I I
I I
X
X X
X
-
E x
LID
LI Schleip
o (nlil/Rq
IP.
VV111 V~JIVV
E
I
.x
X
O Hofmei, 3ter
C')
II
I
A Walker
60
40
1
'kXX-
X
XX
X
MI\
LA
LI
-
AO
I.I
0h
0
O0
O0O
(O
--mx
1
I I I I
50
I I
I
I
I II
100
I
I
I
150
200
II
I
I
I
I
250
I
300
Undercooling (K)
Figure 2.2: Selected prior investigations of pure Ni. References for plotted data:
Bassler, et al [8]; Piccone, et al [4]; Walker [2]; Hofmeister, et al [6]; Schleip, et al
[12]; Colligan and Bayles [3].
1900
"1700
1500
- 1300
1100
0.00
Fe
0.20
0.40
0.60
0.80
ATOMIC FRACTION Ni
1.00
Ni
Figure 2.3: Fe-Ni equilibrium phase diagram with calculated metastable extensions
(from Chuang, et al [20]).
Prior Fe-Ni investigations:
undercooling vs. velocity
0u
*
Fe-30 at.% Ni
40
E
0o
-0
30
*
20
*
-0
*
@0*
*
10
A
....
I
0
I
I
50
. I
100
I
150
Undercooling
200
250
(K)
50
*,
Fe-25 at.% Ni
40
E
20
30
20
1
S.
"
0
0
50
I
100
150
Undercooling
200
250
(K)
Figure 2.4: Prior velocity calculations for Fe-Ni alloys performed by Barth, et al [21]. A
critical undercooling of 175 K was identified for both compositions after
comparison with LKT predictions.
3. Experimental Procedure
Figure 3.1 shows a schematic of the apparatus used in the current work to obtain
thermal profiles and visual images of undercooled specimens.
3.1 Specimen Preparation
Two lots of high-purity nickel were used for this study, both obtained from
Electronic Space Products International. The first lot was 99.9+ pct. (3N) pure wire stock;
the second was rod stock of 99.999+ pct. (5N) purity.
The iron used in the Fe-Ni
specimens was 99.9 pet. pure flake obtained from Johnson Matthey Alfa Aesar.
For each pure Ni specimen, approximately eight grams of either 3N or 5N material
was cut, cleaned in ethanol, and then induction-melted inside an open-ended quartz tube on
a bed of high-purity quartz frit (see Figure 3.2). The Fe-Ni specimens were prepared in
similar manner from portions of 3N Ni wire and Fe flake pre-weighed to a nominal
accuracy of ±.001 g. Compositions under study included Fe-10-, 12-, 15-, 20-, and Fe-30
wt.% Ni. For all specimens, prior to melting, the enclosing test tube was evacuated and
flushed several times with high-purity Ar gas; subsequently, the gas was allowed to flow
continuously through the test tube.
Power for heating was supplied by a Lepel model T-10-3 10 kW radio-frequency
current generator operating at a nominal frequency of 400 kHz. Energy was input to the
specimen by an 8-turn, 1/8" copper tubing split induction coil. During a typical thermal
cycle, the cylindrical ingot was inductively heated through its liquidus temperature to a
maximum superheat of 100-150 K. The heating power was then switched off and the
specimen was allowed to cool under the flowing Ar until either spontaneous nucleation
occurred, or else nucleation was stimulated at the top surface of the ingot using a short
length of Fe or Ni wire.
3.2 Pyrometry
3.2.1 Data Acquisition
Temperatures were measured using a silicon photodiode-based Capintec Ratioscope
III two-color pyrometer operating at near-infrared wavelength bands centered at 0.81 and
0.95 gim. Pyrometric data was recorded by a Nicolet 4094B digital oscilloscope with two
model 4562 plug-ins and an XF-44 twin disk drive.
During a typical run, the four
differential analog inputs of the Nicolet were used as follows: two inputs sampled the
pyrometer channels continuously at a rate of approximately 20-50 Hz, producing a 1-3
minute thermal history of a melt-superheat-solidification cycle.
The other two inputs
produced a high-speed recalescence profile by triggering upon recalescence and recording
at the rate of 0.2-1 MHz for 4-20 ms,retaining an adjustable amount of pre-trigger data.
3.2.2 Data Analysis
The results of each thermal cycle were saved in Nicolet-specific format on 5 1/4"
floppy disk for archiving and then downloaded to an IBM PC-AT computer via a GPIB
interface using the "Henry" file-transfer package provided by Nicolet.
Usually, the
4094LTU transfer program was used, rendering the data into an older "LOTUS 1-2-3"
spreadsheet-style format. The data was ultimately transformed into simple tab-delimited
column format using a C program (see Appendix A4) in order to facilitate subsequent
analysis.
Temperatures were derived from each two-channel set of pyrometer data using the
following relation:
Tobs = Tref + m (Robs - Rref),
where
Robs is the observed ratio of the signal intensities of the two pyrometer
channels: Robs = 10.95 gtm /10.81 tm,'
(3.1)
Tref is the temperature of a known reference point on the thermal
profile, usually the liquidus temperature for a pure material,
Rref is the ratio value observed at Tref, and
m is the slope of an experimentally-obtained ratio-temperature curve,
roughly linear over a range of several hundred degrees
This linear relationship between ratio and temperature is an empirical one only; radiation
theory [24] suggests the following dependence:
R= k exp(4-,)(3.2)
with
k 2 -=(1.44x 104 m K
1
This equation predicts a theoretical variation in m, the slope from Eq. 3.1, of about 6%
between 1726 K and 1426 K (a 300 K undercooling in pure nickel).
However, the error in temperature measurement is reduced somewhat by defining m
as the average slope observed in a wide range of undercoolings during a set of calibration
experiments. These experiments require the immersion of a thin, zirconia-sheathed type-B
thermocouple in the molten ingot, allowing simultaneous measurement of both pyrometer
signal ratio and true specimen temperature. A best-fit linear analysis of the undercooling
segment of the resulting ratio-temperature plot (Figure 3.3) yields the average m value for a
given run. In the present work, the overall average slope value for nickel was found to be
880 +20 K-l1 ; this compares well with a value of 850 K-1 previously obtained [26] using
the same setup. No reliable calibrations were performed for the Fe-Ni material; instead, an
approximate value of 900 K- 1 was assumed.
As a result, the absolute accuracy of the
temperature measurements is estimated to be ±10 K for nickel and, conservatively, ±25 K
for Fe-Ni specimens at the highest undercoolings.
The conversion in Eq. 3.1 was applied interactively to the pyrometer data using a
virtual instrument written in LabVIEW i running on a 100 MHz Pentium-based IBMcompatible computer. The resulting time-temperature profiles could then be evaluated to
determine the extent of undercooling for each thermal cycle.
3.3 Image Acquisition and Analysis
3.3.1 Acquisition
3.3.1.1 Hardware
The camera depicted in Figure 3.1 is a Kodak EktaPro HS Motion Analyzer, Model
4540--a high-speed digital camera. The device supports a maximum acquisition rate of
40,500 frames/sec, at which speed the image consists of a 64 x 64 pixel array with a
precision of 8 bits/pixel.
A 200 mm lens was employed in this work to capture an
approximately 10 mm x 10 mm image from a working distance of about 40 cm.
3.3.1.2 Technique
After each thermal cycle, selected video frames comprising the entire solidification
event were recorded onto S-VHS videotape for archiving. In order to perform subsequent
computer-assisted image analysis, the images from the videotape were re-digitized and
stored on a Macintosh IIci personal computer using a frame-grabber expansion card and an
image-processing package called IPLab".
The frame-grabber sampled the videotaped
images at twice the pixel resolution of the EktaPro 4540, producing 128 x 128 pixel images
with an artificially enhanced resolution. These images were suitable for further analysis
and digital image-processing using IPLab.
i LabVIEW is a registered trademark of National Instruments, Inc.
ii IPLab is a registered trademark of Signal Analytics, Inc.
3.3.2 Analysis
3.3.2. 1 Solidification Interface
The camera observes growth of the solidification front by sensing a change in light
intensity (and therefore temperature) near the front comprising the array of dendrite tips.
This temperature change takes place over a distance of approximately a/V from the growth
front interface, where a is thermal diffusivity for nickel and V is dendrite tip velocity.
Taking a = 1.6 x 10-5 m2/s (refer to Appendix A2 for a summary of physical constants
used) and a conservative solidification velocity of 5 m/s, the ratio of 3.2 gm indicates that
the thermally measured growth front closely approximates the actual physical interface.
3.3.2.2 Processing
The digitized images of the solidification interface were manipulated as follows:
first, the location of the solid/liquid interface was consistently defined in each recorded
frame by isolating only those pixels (xi, yi) included in a numerically-defined transition
zone between the neighboring bright (solid) and dark (undercooled liquid) pixels (Figure
3.4a). The zone typically spanned about 5% of the intensity transition and was centered
near 25% maximum brightness.
As this interfacial band was identified in each individual frame, the interface pixels
were incrementally incorporated into a 32-bit composite master image (Figure 3.4b).
Since, in the general case, it was possible for a specific composite pixel (xc, Yc) to have
appeared in the interfacial bands of multiple frames (for example, a slow-moving interface),
special care had to be taken to ensure that the frame-by-frame pixel record could be
accurately reproduced from the composite image.
Thus, each interface pixel from an
individual frame was made to contribute an increase in intensity of 2 N in the corresponding
composite pixel, where N={0,1,2,...} was the frame number from which the pixel
originated.
3.3.2.3 Composite Image Interpretation
The resulting composite image was a two-dimensional matrix of intensity values
which could be interpreted in the following manner: a zero at a given (x,y) position
indicated that no pixels from any frame's solid/liquid interfacial band appeared at that
location during solidification.
Nonzero values were readily interpreted by their binary
representation: a nonzero bit at arbitrary bit position M (counting the lowest-order bit as bit
0) indicated the presence of a pixel from the interfacial band of frame M. For example, a
value of 0d192 = Ob 11000000 at coordinate (45, 63) indicated that the solid/liquid interface
had been present at that location during frames 6 and 7 of the solidification record. In
general, however, interface motion was fast and predictable enough that each coordinate
contained either a zero or a perfect power of two.
The composite image was finally transferred as text to a UNIX-based workstation
for velocity analysis. There, another C routine (see Appendix A4) was used to create a
formatted datafile listing the interface points by frame along with various supplementary
information and calibration values. This datafile completely characterized the solidification
event along the visible surface of the specimen and was suitable for input to the iterative
solidification model.
3.3.2.4 Pixel Intensity Profiling
One additional method of analysis which helped characterize the doublerecalescence phenomenon in Fe-Ni was pixel intensity profiling. In this technique, an
IPLab script stepped through a series of solidification images and sequentially recorded the
average pixel intensity observed in a very small (2x2 pixel) portion of each image. The
result was a time-intensity profile similar to the output of a 40.5 kHz wide-band pyrometer
tightly-focused on a -0.16 mm square portion of the specimen. No attempt was made to
calibrate the observed intensities to true temperature in this work, since experimental
conditions varied greatly from specimen to specimen; however, the profiles were very
useful in establishing relative temperatures and defining successive stages in solidification.
3.4 Solidification Model
3.4.1 Assumptions
An original computer-based solidification model (see Appendix A4) was used to
calculate dendrite growth velocity within the melt from the observed surface propagation.
The primary assumption underlying the model was that the solid phase grew spherically
outward at a constant rate from a single nucleation point in an infinite undercooled melt;
observed surface interfaces reflected the intersection of the expanding spheres with the
physical boundaries of the ingot (Figure 3.5).
It was expected that this assumption might begin to break down either at very small
undercoolings or else during the latter portion of the solidification event, when remnant
undercooled liquid may become significantly reheated by the neighboring solid. Shrinkage
effects might also distort the assumed geometry of the specimen.
It was assumed in addition that solidification proceeded quasi-adiabatically in the
undercooled melt, and that the effects of the quartz enclosure on solidification kinetics were
negligible. In this manner, the material at the quartz/metal interface could be assumed to be
representative of the material in the bulk of the melt. This is another set of assumptions
which will admittedly break down at very small undercoolings, when time scales increase
and heat flow begins to become an issue.
3.4.2 Operation
Operating under the constant-velocity assumption, the computer model first input all
the (x,y) points from a given interface datafile and assigned a relative time value (relative to
the acquisition time of the first video frame) to each point. The relative time consisted of an
integral multiple of Tf plus a fractional value of Tf that varied with the screen location of the
interface pixel, where Tf was the acquisition time for a single video frame. The fractional
portion is required because the pixel elements are not scanned simultaneously, but instead
sampled by four horizontal passes of a 1x16 pixel vertical raster block (Figure 3.6; see also
effect on appearance of image in Figure 4.3). As a result, the relative time value for a given
pixel (xi, Yi) in frame N is expressed as
trel = Tf N + [floor
(
4'+ .xl)
(3.3)
Given the dimensions of the cylindrical ingot and a calibration value (mm/pixel), the model
was also able to assign a z value to each (x,y) pair, thus defining each interfacial point
completely in space and time.
At this stage, there remained two unknowns: the exact coordinate of the nucleation
site and the period of time which had elapsed between nucleation and the first video frame
of the solidification event. Given any possible nucleation point (xn, yn, zn) and time delay
(tn) the program was able to calculate a consequent average velocity for each frame based
upon the location of its interface points (xi, yi, zi):
P
Vf_
where
vi
1
P
for V
Di
ti
Di
(3.4)
Di =(xi - Xn) + (yi- yn)2 + (zi - zn)2 ,
ti = trel + tn,
and P is the total number of interface points evaluated.
The standard deviation of a group of average frame velocities, normalized over the
average overall velocity, represented the degree of validity associated with the assumed
nucleation position and time. The model proceeded to iterate over both position and time
until it reached a local minimum in standard deviation, and the resulting solution was
assumed to reflect the actual nucleation point of the solidification event.
CAMERA
PYROMETER
Figure 3.1: Schematic of apparatus used for pyrometric data and image acquisition.
Figure 3.2: Detail of experimental setup, showing specimen position and geometry
during processing.
Pyrometer Signal Ratio
Ratio-Temperature Calibration Plot
lt.•
-------------- -----------------------I
----------- l-------------------------------
1450
Portion plotted from
about 80-100 seconds
(undercooled liquid)
-----------.
.I..
II
.---.-------
I
-----
50
--
---- ,-------
100
Thermocouple Temperature
1800
1700--------------------------
(D 1400
r-----
a5
E
a) 1350
1600-------
----------------150 ---- -----1500--------
1300
1.2
1300------. ,
time (se
time (sec)
,
***
4-.
,'
1400-----------------------
1200 .
.4O
-
H-
slope: 870 K
1.3
1.4
Ratio
10UU
Figure 3.3: Typical ratio-temperature curve obtained during slope-calibration testing for
pure Ni. Included at left are the time-ratio and time-temperature data for the
specimen which yielded the ratio-temperature plot. In this case, a best-fit linear
analysis of the data gives a ratio-temperature slope of 880 K.
Figure 3.4: Determination of solidification interface. At left is single image from
recalescence event with computer-defined interface shown in black and newlyformed solid bright at top. At right is complete sequence of interfaces from same
recalescence event.
TRIGGER NEEDLE
SPECIMEN CROSS-SECTION
Figure 3.5: Cross-section of solidifying specimen, with successive model interfaces
shown at constant time intervals following nucleation.
Inherent (64 x 64) pixel resolution
Raster direction
(1 x 16) pixel raster block
Image boundary
Figure 3.6: Raster motion during image acquisition at 40,500 frames/sec. Raster block
begins at top left and scans from left to right, top to bottom in four passes, reaching
bottom right after slightly less than 1/40,500 sec.
4. Results and Analysis
4.1 General Observations
The high-speed optical investigation produced thousands of images, with most
recalescence events comprising between 5 and 100 video frames.
A comparison of the
pure Ni and Fe-Ni images revealed several trends which held for all compositions.
4.1.1 Multiple Nucleation
Multiple simultaneous nucleation was never conclusively observed at any
composition, despite detailed post-experimental review of both spontaneously-nucleated
and needle-stimulated specimens. One or two apparent multiple nucleation events could
instead be attributed to propagation from a single event on the back side of the specimen,
during which the jagged leading edge of the dendrite array intersected with various
unconnected points on the front (visible) surface (Figure 4.1).
A particularly striking
example of this phenomenon occurred during related investigation of Ni-25 wt.% Sn
alloys, and is shown in Figure 4.2, in which the "apparent" new grains all have the same
crystallographic orientation as the parent grain.
These results do not imply that multiple nucleation can never occur. Instead, the
video evidence is a strong indication that growth kinetics dominate nucleation kinetics in
this sort of experiment. Moreover, the growing solid does not appear to have any influence
on nucleation in the undercooled material ahead of the solid/liquid interface.
4.1.2 Interfacial Morphology
The solidification interface was also observed at every composition to exhibit two
distinct interfacial
morphologies.
The
envelope
of
solid
dendrites
appeared
macroscopically jagged at lower undercoolings, but smooth and convex to the liquid at
higher undercoolings, a phenomenon previously reported by Colligan and Bayles [3] in
pure Ni. This behavior will be further discussed in later sections devoted to the individual
specimen compositions.
4.2 Lens and Wetting Effects
A combination of two factors caused the diameter of the molten specimen to appear
larger than the 11 mm inside diameter of the quartz tubing that housed it. First, a lens
effect arose near the left- and right-hand walls of the tube when its contents were viewed
from the side, due to the increased thickness of curved quartz between the viewer and the
specimen. A secondary and more dominant effect developed when a hot specimen became
molten and began to wet the inside wall of the tube; there was an immediate and significant
apparent increase in sample diameter.
After some experimentation, it was found that the best way to quantify the above
phenomena was to introduce an immersion oil-soaked section of millimeter-rule graph
paper onto the inner surface of the quartz tube in place of a specimen, and photograph the
setup with backlighting (Figure 4.4). The immersion oil enabled the graph paper to wet the
quartz in much the same way as the specimen, and the wetting and lens effects could be
calculated by examining the apparent spacing of the vertical rules of the paper across the
width of the image.
The unaffected, 1 mm-spaced horizontal rules in Figure 4.4 provided a calibration
value in terms of pixels/mm which could later be used to relate pixel distances to true
distances when calculating velocities. The value was also fed into a small optimization
routine, along with the positions of the vertical rules in the calibration image. The routine
calculated that the data was best modeled by assuming the inside radius of the quartz tube
was approximately 14% larger than its physical value. Figure 4.5 shows black model rule
lines (Rapparent = 6.25 mm) superimposed over the gray observed rule lines (Ractual = 5.5
mm) traced from Figure 4.4, demonstrating excellent agreement over almost the entire
width of the tube. In practice, data was never taken from less than about 1 mm away from
the sides of the tube, in order to avoid the large error inherent to such measurements.
4.3 Solidification Model Behavior
4.3.1 Iterative Convergence
As previously discussed, operation of the solidification model consisted of an
iterative search for an optimum nucleation point, assuming constant-velocity growth of
solid. The variables of iteration were, in order, tn (time delay), zn, xn, and Yn, where the
positive z axis extended toward the camera. The model's ability to converge upon an exact
solution depended greatly upon the true location of the nucleation point; complete
convergence was far more likely if the nucleation point lay on the front surface than if it lay
on the top, bottom, back, or interior of the specimen.
4.3. 1. 1 Convergence for All Variables
Often, a spontaneous nucleation site appeared to be located on the front surface
(metal/quartz interface) of the ingot. In some cases, the nucleant was clear upon review to
have been an oxide particle or a scratch in the quartz tube; in other cases the only evidence
was an initial solidification interface with a very small radius of curvature (Figure 4.3).
For almost all of these "front-surface" nucleation events, the computer model verified the
apparent nucleation site by iterating to convergence in all variables.
The predicted
nucleation point typically matched the observed point within approximately 0.5 mm, and
the delay time between nucleation and first frame acquisition was between 0 and 1
frames-a required condition for a visible nucleation site.
It should be noted that the model was also able to converge for all variables in some
cases where the nucleation point did not lie on the front surface. For instance, there were a
number of examples of needle-stimulated top-surface nucleation that were correctly
predicted by the solidification model.
More generally, in all cases of complete
convergence, the model's optimum (xn, Yn, zn) coordinate was found to lie within a
distance (.05*R) of the ingot's surface, where R represents either the radius (cylindrical
surface nucleation) or the half-height (top/bottom surface nucleation) of the ingot. This
behavior was interpreted as strong evidence that nucleation occurred at a metal/quartz or
metal/gas interface in all cases in which it was not manually stimulated at the top surface by
a needle.
4.3.1.2 Convergence on Cylindrical Surface
In many other cases, the model was unable to reach an exact solution for tn, Zn, xn,
and yn. This often occurred when the nucleation point lay unseen on a hidden surface and
occasionally even when the point appeared to be located on the front surface of the
specimen. In many of these cases, a solution was possible if the nucleation site was
constrained to lie on the cylindrical surface of the specimen and the model iterated over only
tn, xn, and Yn, with zn determined by the other two spatial coordinates and the geometry of
the ingot.
4.3.1.3 Time-bounded Convergence
A final general case occurred when no convergence was possible for any set of
initial conditions because the optimum tn value increased without bound. As expected, the
model often behaved in this manner when the true nucleation point was far removed from
the front surface of the specimen and the camera had not recorded the initial portion of the
solidification event.
Fortunately, it was often possible to impose constant upper- and lower-bounds on
tn in these cases and solve for the optimum (xn, Yn, zn) coordinates and solidification
velocities given the bounding tn values. This was a particularly useful technique in the case
of non-convergent needle-stimulated events, because the Yn coordinate was known to lie
within a small distance of the (visible) top edge of the specimen. The strategy in these
cases consisted of iterating with various fixed values of tn until two arguments were found
which resulted in sensible bounding values of yn--typically, two pixels above and two
pixels below the observed top edge of the specimen. The corresponding velocity values
were also taken to be upper- and lower-bounds, and they did not often differ by more than
five percent.
In all cases where bounded solution sets showed a variation of more than 10% of
the average solution, the solutions were removed from further consideration. This result
typically followed when the radius of curvature of the first observed solidification interface
was unusually large, supporting the notion that the camera had only observed the final
portion of the solidification event and the model has broken down.
4.4 Pure Ni Results
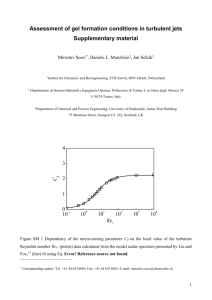
4.4.1 Solidification Velocity
Presented in Figure 4.6 are the solidification velocity values, plotted against
undercooling, obtained from the computer model used in this work.
Comparison with
dendrite growth theory will be made using the so-called "LKT" model of Lipton, et al. [9],
who adapted the Ivantsov dendrite growth model [25] using a marginal stability analysis
(see Appendix Al for LKT development). Shown in Figure 4.6 are both the unmodified
LKT prediction for pure nickel and the LKT prediction assuming a linear kinetic
undercooling parameter [t = 0.40 m/sK.
Clearly, there is fair agreement between
experiment and theory up to some critical undercooling AT*, where 150 K < AT* < 180 K
in this case.
Above AT*, it is difficult to characterize exactly the relationship between
undercooling and velocity, other than to observe that solidification velocity continues to
increase. These results are in general accord with all of the prior work referenced, with the
exception of Hofmeister and Bassler.
4.4.2 Interfacial Morphology
A morphological analysis of the solidification interface at various undercoolings
was carried out in the following manner: any specimen whose images exhibited
discontinuous interface curvature not attributable to the camera raster pattern was classified
as "jagged;" specimens showing only continuous interface curvature were called "smooth."
This analysis revealed a transition undercooling range for Ni of 160-170 K, above which
all solidification envelopes were smooth and below which all envelopes were jagged
(Figure 4.7). This range is slightly above the critical range for morphological transition
previously proposed by Colligan and Bayles. The temperature range does correspond well
to the "critical undercooling" values observed on undercooling-velocity plots both in the
current work (150-180 0 C) and by prior investigators.
Furthermore, a morphological
transition AT* of 160-170 0 C is also approximately equal to the microstructural AT*
observed by Walker, Colligan, Schleip, and others in pure Ni.
The present work strongly suggests that the morphological transition in solid/liquid
interfacial shape and the observed transitions in velocity and microstructure are coupled
phenomena resulting from the same intrinsic shift in solidification mechanism. However,
the physical mechanism responsible for the transitions has yet to be satisfactorily explained.
Various hypotheses point toward either a kinetic attachment limitation [6], the onset of
dendrite fragmentation by remelting [26], dynamic nucleation via shrinkage-induced
cavitation in the undercooled melt [27], or other fluid-flow phenomena.
4.5 Fe-Ni Alloy Results
4.5.1 Pyrometric Thermal Profiles
Two typical high-speed thermal profiles from Fe-10 wt.% Ni specimens are shown
in Figure 4.8. The upper profile represents a specimen which was not observed in the
video record to have undergone double recalescence; instead, the equilibrium FCC phase
solidified directly from the undercooled melt. This profile exhibits a clear thermal peak at
the tail end of the rapid transition, approximately 10 K in amplitude and spanning about 0.1
ms. A similar phenomenon was observed by Piccone [28] during his studies of hyperperitectic Fe-Ni, Ni-Sn, and Fe-Co alloys using the same apparatus. He attributed the peak
to superheating and subsequent remelting of a portion of the newly-formed dendrite array.
The profile in the lower half of Figure 4.8 shows a specimen which did exhibit
double-recalescence behavior in the video record. This behavior is clearly reflected in the
thermal profile by a preliminary plateau at approximately 1495 K (near the metastable
liquidus temperature) lasting for about 1 ms, followed by a secondary rise in temperature to
near the equilibrium FCC liquidus with no characteristic thermal peak.
Unfortunately, not every specimen which underwent double recalescence visible by
the camera displayed such an unambiguous pyrometric profile.
Many such specimens
instead showed little more than a change in inflection, and sometimes no hint at all of
double recalescence in the pyrometric record (Figure 4.9).
Failure to detect multiple
thermal transitions was probably a result of the pyrometer's relatively low spatial
resolution; the view field of the instrument was between 6 and 10 mm in diameter for the
duration of the work, and the output voltage reflected the average light intensity of the
entire field. It is not surprising that transitions narrowly separated in distance or time were
not crisply detected by this instrument.
4.5.2 Pixel Intensity Profiles
The pixel intensity profiling outlined in section 3.3.2 was a much more reliable
method of detecting and characterizing double recalescence events, despite its limited
temporal resolution (40,500 Hz). In general, double recalescences were easily identified
by a secondary intensity transition either on videotape or in an animation of the digitized
images from a single event. Occasionally, the growth of FCC material took the shape of a
nearly continuous interface which lagged the BCC solidification interface by a short
distance.
More often, however, the FCC transition was more diffuse, appearing to
nucleate and grow from multiple points behind the BCC interface.
In either case, the
transition was predictable enough so that, at any given point on a single specimen, there
appeared to be a roughly constant delay time between the passage of the BCC interface and
the subsequent transformation to brighter FCC material.
Due to the consistent behavior of the transformation, the choice of the target area for
pixel intensity profiling was relatively unimportant; an arbitrary 2x2 pixel area free of
oxidation or quartz discoloration was chosen for each event. The resulting time-intensity
profile was generally a much more sensitive record than the corresponding pyrometer
profile (Figure 4.10), giving a clearer indication of recalescence behavior and delay times
between transitions, if not accurate temperature values.
4.5.3 Recalescence Behavior
4.5.3. 1 Characterization
Recalescence characterization was accomplished through simultaneous examination
of the pixel intensity profile, the high- and low-speed thermal profiles, and the video
record. Double recalescences which were clear on the video record were almost always
verified by a double-plateau in the pixel intensity profile and were considered clear
instances of initial BCC phase formation.
Apparent single recalescences on the video
record were typically supported by single-plateau pixel intensity profiles; in addition, the
high-speed thermal profiles in these cases generally exhibited a superheat peak on
recalescence of about 5-15 K which was absent in the case of double recalescence.
The video record was no help in the identification of secondary recalescence events
which lagged the initial solidification by more than about 0.2 seconds, because video data
was not recorded beyond this time. However, the low-speed thermal record was assumed
to be sensitive enough to identify these delayed transitions. In cases where the recalescence
behavior was truly unclear, the data points were omitted from plots and calculations.
Figure 4.11 reveals the recalescence behavior of the various Fe-Ni specimens as a function
of composition and nucleation temperature. Filled symbols indicate that primary BCC
solidification and double recalescence occurred; outlined symbols indicate primary FCC
solidification. Briefly ignoring the significance of symbol shape, one can interpret Figure
4.11 as a primary solidification map valid for all quartz-fluxed Fe-Ni specimens of similar
aspect ratio weighing approximately 8 g and exhibiting the same cooling rate (phase
selection depends upon the thermal history of the undercooled liquid as well as the presence
of heterogeneous nucleants [17]).
This work suggests that there exists a window of primary BCC solidification in
hyperperitectic Fe-Ni bounded at smaller undercoolings and very large undercoolings by a
transition to primary FCC solidification. The window appears to span about 150 K at 10
wt.% Ni, tapers to less than 120 K at 15 wt.% Ni, and has disappeared entirely at
compositions of 20 wt.% Ni and higher.
4.5.3.2 Phase Selection via Induced Nucleation
As noted earlier, both Fe and Ni trigger needles were used at various times to
induce heterogeneous nucleation at lesser undercoolings, with the idea that the Fe (BCC
from room temperature to 1185 K and from 1667-1811 K) might induce primary BCC
solidification and the Ni (FCC through the entire range) might produce instead the FCC
phase. As Figure 4.11 clearly reveals, these efforts met with no success at all, with the
exception of three points near 1350 K in the Fe-12 wt.% Ni material.
In those three
instances, FCC material formed from the Ni trigger on a section of the map which
otherwise featured solely BCC solidification.
At all other compositions and temperatures, trigger needle structure seemed to have
absolutely no effect on the determination of the primary solidification phase. Apparently,
FCC Ni is simply not a sufficiently potent heterogeneous nucleant to have much of an
effect on the kinetics of primary phase solidification in the compositions that were
extensively studied. Perhaps a trend would have emerged if more data had been collected
from the Fe-15 wt.% Ni alloy.
4.5.3.3 Delay Times in Double Recalescence
The delay time between recalescences was defined on the pixel intensity profile as
the time between points of maximum curvature at the beginning of each transition. The
resulting correspondence between undercooling and delay time for double recalescence is
shown in Figure 4.12 for the compositions which most consistently exhibited that
behavior.
Figure 4.12 reveals that average delay time drops with increasing undercooling in a
roughly exponential manner. The data scatter may be an indication that there are several
different mechanisms by which the FCC phase nucleates and grows into the mixture of
undercooled liquid and BCC solid material. However, the data is in qualitative agreement
with a theory that FCC formation requires the attainment of a critical local fraction solid,
since the fraction solid immediately following recalescence can be expressed as:
AT
fs = AT
AThyp
where AThyp =
AHf
Cp
(4.1)
FCC nucleation may result from the impingement of growing BCC dendrite arms during
the equilibrium solidification period following initial recalescence.
4.5.4 Interfacial Morphology
A morphological analysis identical to that described in section 4.3.2 enabled the
characterization of solidification interfaces as "smooth" or "jagged" for most compositions
of Fe-Ni studied in this work. Figure 4.13 summarizes the results of the analysis.
Once again, there is a rather sharply-defined transition from jagged to smooth
morphology at a critical undercooling; this AT* is approximately 190 K for both Fe-10- and
Fe-12 wt.% Ni. The exact value at higher Ni compositions cannot be precisely determined
from the data, but it is evidently no more than 190 K, and not a great deal less. The range
of 160-190 K previously identified for morphological transition in pure Ni would suggest
that AT* decreases only slightly, if at all, as the specimen grows richer in nickel content.
4.5.5 Solidification Velocity
Solidification velocities obtained from the computer model are given in Figure 4.14
for Fe-10- and Fe-12 wt.% Ni specimens which exhibited double recalescence
(undercoolings are measured from the metastable liquidus temperature). The two sets of
data are plotted on the same graph because LKT theory predicts a virtually identical
undercooling-velocity curve for both compositions. Indeed, it is clear from the plot that the
two data sets essentially overlap and are well-predicted by an LKT analysis for Fe-Ni using
a kinetic parameter of 0.28 m/sK.
The most notable aspect of Figure 4.14 is the clear lack of any transition in
functional dependence in the range 170 K < AT < 190 K. Instead of the downward shift
observed in this work for Ni (Figure 4.6) and in the work of Barth, et al [21] for Fe-25and Fe-30 wt.% Ni, the data continues to be well-described by LKT theory right up to
undercoolings of nearly 250 K.
In light of the observed transition in interface morphology, the lack of any
corresponding shift in solidification velocity is somewhat difficult to explain.
One
difference between these Fe-(10-12) wt.% Ni results and the contrasting pure Ni and prior
Fe-Ni work is that the primary solidification phase in the current work is a BCC material.
Perhaps crystallographic structure plays an important role in determining the interface
kinetics of these rapidly-solidified materials. Unfortunately, there is insufficient data in the
present work at the higher Ni concentrations to draw any conclusions regarding the
solidification velocities observed there for FCC Fe-Ni.
A more likely explanation is that the velocity transition is not actually governed by a
critical undercooling, but instead by a critical velocity which is comparable across
materials. The transitions in the Barth work appear to begin somewhere above 30 m/s. The
data in Figure 4.6 for pure Ni suggests that the critical velocity range corresponding to
150 0 C < AT* < 180 0 C is approximately 26 m/s < V* < 35 m/s; velocities of nearly 50 m/s
were observed at the largest undercoolings. The maximum velocities observed for Fe-(1012) wt.% Ni in this work were only 30-35 m/s. Thus, if the transition velocity for Fe-(1012) wt.% Ni alloys is near the top of the range observed for pure Ni, it is not surprising
that no transition was experimentally observed.
However, regardless the explanation, it
would seem that solidification velocity behavior and interface morphology are not strongly
coupled in the metastable phase solidification of these Fe-Ni alloys, as seemed to be the
case for pure Ni.
SIBLE SURFACE
CAMERA
SPECIMEN
X-SECTION
Figure 4.1: Schematic of apparent multiple nucleation scenario, observed experimentally
in Figure 4.2.
Figure 4.2: Example of solidification interface propagating from back to front, almost
directly toward the camera lens. System is Ni-25%Sn, -25 ms between images.
Dendrite tips intersecting front surface appear as numerous expanding diamonds
with the same axial orientation.
Figure 4.3: Composite interface image showing front-surface nucleation; initial interface
at upper-right exhibits very small radius of curvature. Note also raster pattern effect
at 1/4, 1/2, and 3/4 of the image height causing apparent discontinuities in interface
curvature.
Figure 4.4: Calibration image of oil-soaked millimeter graph paper rolled inside 11 x 13
mm quartz tubing.
Figure 4.5: Model vertical rules (black) overlaying observed rules (gray, traced from
Figure 4.4), verifying that the lens and wetting effects are well-modeled by
assuming a 14% increase in specimen radius.
I I
I
I
I
I
I I I
I
I
I
I II
I
I
I I
/
/
I
II I
I
I
60
Current Work
*
LKT prediction
50
*
E
•
0
LKT w/kin. par.
40
/
*
/-
30
O
*/
a)
>
20
10
"
"
"
A
1
0
50
100
150
200
250
Undercooling (K)
Figure 4.6: Solidification velocity plotted versus undercooling for pure Ni, as
determined by this work. Also shown is LKT prediction with and without
modification by a kinetic undercooling parameter.
300
Figure 4.7: Interface morphology transition for pure Ni. Shown are typical interface
morphologies observed at undercoolings less than the critical range of 160-170 K
(left column) and greater than the critical range (right).
51
__
~I I
_
~___
1500
1450
1400
&
1350
E
0 1300
1250
1
time (ms)
0
E
0)
L.
E
0-
-
-
-
--
------
Figure 4.8: Typical thermal profiles for Fe-10 wt.% Ni specimens undergoing single
recalescence only (top) and double recalescence (bottom). Note superheat peak at
top and double-plateau behavior at bottom.
Double Recalescence (not detected by pyrometer)
1500
CI)
L 1450
E
'
1400
1
n
0
1
2
3
4
time (ms)
Figure 4.9: Pyrometer profile for Fe-10 wt.% Ni specimen which was revealed in the
video record to have undergone double recalescence. No clear indication is present
in the thermal record.
Pyrometer Temperature
Pixel intensity profile
I/
60
50
*
0
40
30
-
0
,,,,,,1,,,,,-.
2.5
3.0
3.5
· ·
4.0
. ·
4.5
time (ms)
Figure 4.10: Comparison between pyrometer profile and pixel intensity profile for Fe-10
wt.% Ni specimen undergoing double recalescence. Details are much more
sharply-defined in the pixel intensity profile.
54
Primary phase solidification as function
of nucleation temperature, composition,
and trigger material
· _ _· ) I__·
.
1450
•
· _· · · ·
-
.
'S
o
1400
0~
ca
*
**
*.
Sa
*
Ill
,mm
a·
I
FCC; Ni trigger
FCC; Fe trigger
FCC; spontaneous
BCC; Ni trigger
BCC; Fe trigger
BCC; spontaneous
- - -meta• stable Tliq
stable Tsol
a
aS
m
a)
0.
mm
m.
1350
mm
U
I--n
mm
mm
m
m
m
m
0'
1300
S
O
0
<*4
1250
z
1200
I-
8
12
_I _I
I . I I
16
weight
20
,
, II I
24
percent
28
Ni
Figure 4.11: Solidification behavior in Fe-(10-30) wt.% Ni.
32
.....
Delay
time
between
recalescence
events
3
2.5
E,
E
(U
0
2
1.5
1
0.5
0
100
150
200
undercooling
250
(K)
Figure 4.12: Delay time between recalescence events in Fe-10- and Fe-12 wt.% Ni
specimens.
Interface morphology as function
of undercooling and composition
0 _· I _ · · _· ___·_·I
( ~ 1 ~__·_·__
50
o
Smooth
A
Jagged
Unclear
x
0)
100
A
0
"i
150
transition
temperature
200
0
250
0
0
--
300
8
12
16
weight
20
24
percent
28
32
Ni
Figure 4.13: Interface morphology at various compositions and undercoolings. A
transition from jagged to smooth structure appears at approximately 190 K
undercooling.
Velocity vs. Undercooling:
Fe-1O- and Fe-12wt.% Ni
40
I, I I
I
I
I
I
I
....
I " tI
I
I
|
|I
o BCC Fe- 10%Ni
* BCC Fe-1 2%Ni
....
* LKT theory; mu=0.28
30
-- - - - -
-4
'
U-
I
0
m
IdNa
u)
*QD
0
E
on
20
%*O
v
4 -a
a·
0
a)
))
*8
10
04
I1
I%-./
m~lMIN
I
50
I . I,
100
I
I-I
I
150 200
Undercooling (K)
,
I
250
Figure 4.14: Solidification velocities for BCC Fe-10- and Fe-12 wt.% Ni.
5. Conclusions
5.1 General
1.
The undercooling and subsequent solidification of high-purity Ni and
hyperperitectic Fe-Ni specimens were accomplished, and the thermal front accompanying
the solidification interface was directly observed through the use of an ultra-high-speed
digital camera from Kodak. The degree of undercooling was evaluated for each heating
cycle through the use of a two-color pyrometer and subsequent calibration experiments; a
solidification velocity was calculated for each recorded solidification using digital image
analysis and an original computer-based solidification model.
2.
At no time were multiple nucleation sites observed during a single primary
solidification event. Instead, at lesser undercoolings the solidification interface exhibited a
"jagged" morphology, a phenomenon which may, at lower resolution, present the
appearance of multiple nucleation sites
3.
The computer-based solidification model was successful in determining
solidification velocities with less data scatter than had previously been observed using
pyrometric methods. The model can also be made to track the growth of subportions of the
overall dendrite array, in the event that a study of growth velocities observed in different
crystallographic directions is desired. Pyrometric analyses exhibit no such flexibility.
5.2 Pure Nickel
1. The relationship between solidification velocity and undercooling for pure Ni
showed general agreement with many prior investigations; namely, there was an
approximate power-law dependence at low undercoolings which is well-described by the
LKT solidification model. Above a critical undercooling, 150 K < AT* < 180 K, there
appeared to be a partial shift in solidification mechanism, resulting in a secondary velocity
dependence whose exact nature is unclear.
2. Observed at a similar critical undercooling (160K < AT* < 170 K) was a shift in
interface morphology from "jagged" to macroscopically smooth, implying a change in
controlling mechanism at the dendrite tips perhaps related to attachment kinetics, dendrite
fragmentation, or fluid flow phenomena.
The morphological and solidification velocity
transitions appear to be coupled in pure Ni.
5.3 Iron-Nickel
1. Primary solidification of both the equilibrium FCC phase and the metastable
BCC phase was observed in the hyperperitectic Fe-Ni specimens.
The BCC phase
appeared to be favored at lower Ni concentrations (10-12 wt.%), while FCC solidification
was observed exclusively at 20 wt.% Ni and above.
2. A transition in interface morphology similar to that observed in pure Ni was also
found in all compositions of Fe-Ni. The critical undercooling was well-defined around 190
K for Fe-10- and Fe-12 wt.% Ni specimens, and grew no larger at higher Ni
concentrations.
3. Solidification velocities for BCC Fe-10- and Fe-12 wt.% Ni were plotted against
undercooling and found to be nearly identical, as predicted by LKT theory.
Agreement
between experiment and theory was also excellent even at the highest undercoolings
observed (250 K); there was no shift in velocity dependence as seen in pure Ni.
A
plausible explanation is that the shift actually occurs at a critical velocity, not a critical
undercooling, but in any case it appears that interface morphology and velocity dependence
are not coupled in Fe-Ni as they appeared to be in pure Ni.
6. Suggestions for Future Work
1. The structure of the solidification interface behind the dendrite tips has yet to be
adequately understood. The high-speed images obtained in this work reveal an apparent
thermal transition spanning a distance of several millimeters, but no attempt has been made
here to develop a theoretical model for the behavior.
One possible avenue for future
investigation could be to use higher magnification to obtain high-resolution images of the
apparent interface with the camera used in this work.
2. A theoretical basis is needed to explain the transitions in interface morphology
and velocity dependence observed in this and similar work. Of the explanations offered to
date (attachment kinetics, dendrite fragmentation, cavitation due to shrinkage effects, other
fluid-flow arguments), none has managed to predict the phenomena experimentally
observed in both alloys and pure materials.
3. A further study of the solidification behavior of Fe-Ni alloys using the highspeed camera could further characterize the double-recalescence phenomenon and help fill
in the solidification map in this work at compositions outside Fe-(10-12) wt.% Ni.
Experiments incorporating on-camera quenching, with subsequent microstructural analysis,
might also shed light on the microstructural evolution that takes place following primary
BCC solidification.
Appendices
Al. Solidification Theory
The dendrite growth theory used to create the "LKT" solidification velocity plots for
Ni and Fe-Ni in this work stems from the work of Ivantsov [25] and Lipton, et al [9], with
additional contributions by Boettinger, et al [29], Aziz [30], and Turnbull [31].
In short, Ivantsov proposed the first analytic equation to determine heat flow in a
solidifying dendrite tip realistically modelled as a parabaloid of revolution, resulting in a
single relationship linking R, dendrite tip radius, with V, solidification velocity. Lipton, et
al later derived a secondary relationship between these two variables on the basis of
morphological stability arguments.
The combination of this work and Ivantsov's prior
relationship formed the most widely accepted theory of dendrite growth established to date.
Incorporating modifications by the other contributors listed above, researchers were able to
predict accurately the undercooling-velocity behavior observed in many different elements
and alloy melts.
For further information regarding LKT solidification theory, refer to the above
work or individual treatments given by Piccone [26] and Barth, et al [21]. The next five
pages show a numerical implementation of the growth model using the symbolic
mathematics package Maple V 5.0', following closely the developments of Piccone and
Barth.
iMaple is a trademark of Waterloo Maple Software
LKT SOLIDIFICATION MODEL IMPLEMENTED IN "Maple version 5.0,"
SYMBOLIC MATHEMATICS SOFTWARE AVAILABLE FOR UNIX WORKSTATIONS.
NOTE: This is an effort to implement a create a clear, usable
LKT model that will calculate undercoolings, radii, etc., at
various solidification velocities.
* All thermodynamic constants will be expressed in SI units.
Enthalpy of fusion: (assumed equal in liquid and solid)
> H := 1.746e9;
H := .1746 101i
Heat capacity: (assumed equal in liquid and solid)
> c_p = 6.029e6;
cp := .6029 107
Solid/liquid interfacial energy:
> sigma := 0.28;
a := .28
Thermal diffusivity of undercooled liquid:
> alpha := 5e-6;
c := .5 10-5
Solutal diffusivity of undercooled liquid:
> D_o := 6e-9;
D_o := .6 10-8
Marginal stability constant:
> sigma_star := 1 / (4 * PiA2);
sigma_star .-
1 1
4 72
Assumed kinetic undercooling coefficient (if kinetic undercooling is to be ignored, set this
to a very high value, such as le10):
> mu := 0.28;
. := .28
Solute concentration in atomic %, i.e. Fe-20 at.% Ni --> "20"
> C_o := 10.4;
Co := 10.4
Equilibrium liquidus temperature:
> T_f:= 1770;
T f:= 1770
Calculated value for entropy of fusion:
> delS := H /Tf;
delS:= 986440.6779
Here is the approximate slope of the liquidus. Note this is a NEGATIVE number, in K/at %:
> m := -2.1;
m := -2.1
Next is the "equilibrium partition coefficient," used in the calculation of the
velocity-dependent slope of the phase diagram of the material:
> ke := 0.74;
ke := .74
Also need the "atomic diffusive speed:"
> Vd := 20;
Vd := 20
> k := (V) -> (ke + V / Vd) / (1 + V / Vd);
V
Vd
ke + -
k:= V-1+
V
Vd
-
This produces a velocity-dependent slope in the effective phase diagram:
1
> ml := m * (1 + (ke - k(V)*(l - In(k(V)/ke))) 1 (1 - ke));
.74 + - V
1
74+ 20
1 -In 1.351351351
ml := -8.076923077 + 8.076923077
1+-
1
20
20
1
20
V
With all physical constant input, here are the equations to predict growth:
First, the definition of the Ivantsov function (a function of a Peclet number):
> Iv := (P) -> P * exp(P) * Ei(l, P);
Iv := P -4 P e P Ei( 1, P)
Thermal Peclet number:
> P_t := (V, R) -> R * V / (2 * alpha);
1RV
P_t := (V, R) -9 - 2
Solutal Peclet number:
> P_c := (V, R) -> R * V / (2 * D_o);
IRV
P_c:= (V, R) -• 2 D_o
Constitutional undercooling w Aziz velocity-dependent slope as per Barth paper:
> delTc := (P_c(V, R), k(V))-> m * C_o * (1 - (ml / m) / (1 - (1 - k) * Iv (Pc(V, R))));
delT_c := (Pc(V, R), k(V)) -4 m C_o 1 -
ml
m(1 -(1 -k) )Iv(P c(V, R)))
Capillary undercooling:
> delT_r := (R) -> 2 * sigma / (R * delS);
delTr := R -> 2
R delS
Kinetic undercooling:
> delT_k:= (V)-> V / mu;
delT_k := V
V
-
Ivantsov thermal undercooling solution:
> delT_t :=(Pt(V,R)) -> (H / c_p) * Iv (P_t(V,R));
HIv(Pt(V, R))
delT_t := P_t( V, R) --+H
c_p
This is the marginal stability solution for dendrite tip radius, as copied from Piccone's Sc.D
thesis:
Defined first are two simplifying expressions, n and g:
> n := (P_t(V,R)) -> 1 / (sqrt(1 + 1 / (sigma_star * Pt(V,R)A2)));
1
sqrt 1 +,
sigma_starP_t( V, R)2
> g := (P_c(V,R), k(V)) -> 2 * k(V) ! (1 - 2*k(V) - sqrt(1 + 1 / (sigma_star * P_c(V,R)A
2)));
g := (P_c(V, R), k(V)) -- 2
k(V)
1 - 2 k(V) - sqrt 1 + sigma_starP_c( V, R)2
Here is the actual equation defining R (here, R_th or theoretical) in terms of V and itself:
> Rth := (P_t(V,R), P_c(V,R), k(V)) -> (sigma / (sigma_star * delS)) / ((P_(V,R) * H
* (1 - n(P_t(V,R))) / c_p) - ((2 * m * C_o * (1 - k(V)) * (1 + g(P_c(V,R))) * P_c(V,R))
/ (1 - (1 - k(V)) * Iv(P_c(V,R)))));
/sigmastar delS
R_th := (P_t(V,R),P_c(V,R), k(V)) -
P_t( V, R) H (1 - n(Pt(V, R))) -2 m Co (1 - k( V)) (1 + g(P_c( V, R))) P_c( V, R)
cp
1 - (1 - k( V)) Iv(P_c( V, R))
With the equation above, we can numerically solve for R given any V value:
> vval := 2.107;
vval := 2.107
> rval := fsolve(R = subs(V = vval, R_th(Pt(V,R), P_c(V,R), k(V))), R, R=le-9..le4);
rval := .4122501976 10-6
This is the real meat of the development; it iterates through a user-defined series of
velocities by a variable amount and computes each of the various dendrite tip radii, as well
as the various undercooling contributions, at each velocity value.
The output is spewed into tab-delimited column format as the file "outfile".
*** NOTE: If the iteration refuses to converge for some conditions, you'll have to alter the
search range defined for R in the "fsolve" step on line 3 of the expression below. The
horrible run-on is due to the fact that I couldn't get the "continuation" behavior to work
correctly for this command, perhaps because the "writeto" function is involved.
> vstart := 0.5; vend := 61; vstep := 4;
vstart := .5
vend := 61
vstep := 4
> writeto(outfile); print(printf('delTntvel\trad\tP_t\tP_c\tdelT_t\tdelT_c\tdelT_r\tdel
T_k\n')); for vval from vstart by vstep to vend do writeto(terminal); rval := fsolv
e(R = subs(V = vval, R_th(P_t(V,R), P_c(V,R), k(V))), R, R=le-9..1le-4); Pt := Pt(
vval, rval); Pc := P_c(vval, rval); delTt := evalf(subs (V = vval, R = rval, delTt(P
t(V,R)))); delTc := evalf(subs(k = k(vval), (subs(V = vval, R = rval, delT_c(P_c(
V, R), k(V)))))); delTr := delT_r (rval); delTk:= delT_k (vval); delT := delTt + delT
c + delTr + delTk; appendto(outfile); print(printf('%e\t%e\t%e\t%e\t%e\t%e\t%e\
t%e\t%e\n', delT, vval, rval, Pt, Pc, delTt, delTc, delTr, delTk)); od; writeto(termi
nal);
A2. Tabulated Thermodynamic Data
Thermodynamic data for Ni and Fe-Ni alloys were again drawn from the prior
work of Piccone [26] and Barth, et al [21], and are given in the following table:
Property
Units
thermal diffusivity, ml/s
Pure Ni
1. 6 x 10'-
BCC Fe-l0 wt.% BCC Fe-12 wt.%
Ni
Ni
5.0 x 10 -6
5.0 x 10 6
solutal diffusivity, Im/s
D
6.0 x 10-9
6.0 x 10' 9
enthalpy of fusion, J/kg
2.98 x 105
Hf
heat capacity, cp J/kgK 7.34 x 102
solid/liquid
J/m
0.2 5
interfacial energy,
2.40 x 10 '
2.41 x 101
8.28 x 102
8.29 x 102
0.28
0.28
[none] 1 / 4C2
1 / 472
1 / 4r2
m/sK
0.28
0.28
10.4
12.5
17 6 9
1759
-2.1
0.74
-2.1
0.74
marginal stability
constant
linear kinetic
undercooling
coefficient, p
solute
concentration
equilibrium liquidus
temperature
liquidus slope, m
equilibrium
partition
coefficient, k,,
0.40
at %
K
K/at %
[none]
1728
A3. Tabulated Experimental Data
Undercooling-velocity data shown from other work was obtained either from a
table supplied in the appendix of the work (Piccone [26]) or by scanning a published figure
into a computer and using image-analysis techniques to determine plot values from the
resulting images (Bassler, et al [8]; Walker [2]; Hofmeister, et al [6]; Schleip, et al [12];
Colligan and Bayles [3]; Barth [21]).
The error inherent to the scanning technique is
estimated at 0.5%.
The following tables summarize the undercooling-velocity values both of the prior
work and the current investigation of Ni and Fe-Ni alloys:
Part I: Pure Ni
Piccone
AT (K)
velocity
T (K)
Walker
velocity
rn/s)
m
(m/s)
6
6
8
8
10
12
0.03
0.03
0.06
0.05
0.03
0.04
12
0.08
16
25
32
0.14
0.16
0.32
46
0.15
47
49
60
61
114
115
132
141
0.14
0.20
0.35
0.67
20.0
69.8
29.3
35.7
27.6
40.6
47.5
53.2
59.8
61.2
71.2
75.8
80.4
86.8
91.3
95.2
99.3
100.3
109.8
108.9
111.9
121.9
120.8
1.0
1.7
2.9
4.3
4.3
6.2
7.3
7.2
8.3
11.1
11.6
11.7
13.4
16.0
14.1
15.4
20.6
19.5
20.0
15 2
169
1 c,
17E
19C
194
19E
213
215
26C
263
264
271
277
277
279
280
284
286
286
286
288
288
289
294
298
302
304
306
307
308
309
315
316
317
317
13.
62. c
56.6
64.7
43.1
45.5
46.2
54.2
46.2
44.6
50.8
102.3
67.7
60.4
68.7
62.5
48.1
80.4
55.6
48.4
84.9
54.5
52.9
50.6
83.3
69.2
52.6
62.5
55.9
70.3
52.9
59.2
55.9
49.5
62.1
123.3
126.1
121.3
133.6
130.4
138.8
143.0
146.6
158.7
154.1
158.9
166.7
171.7
169.2
182.7
182.5
191.8
188.6
169.0
177.7
188.4
201.9
200.5
232.2
223.3
214.2
248.9
211.9
233.4
218.3
241.1
i
20.8
20.9
24.6
25.1
26.9
27.6
29.0
28.8
28.9
32.9
36.5
34.7
36.2
38.7
36.6
38.5
37.6
40.7
44.7
45.5
43.5
41.4
44.9
33.1
41.1
47.6
48.7
61.1
64.4
70.0
77.7
Hofmeister
Bassler
velocity
AT (K)
velocity
AT (K)
(m/s)
(m/s)
133.0
9.7
132.0
145.7
10.1
12.7
149.5
13.4
153.6
167.6
165.7
169.4
173.0
182.7
177.7
178.9
180.3
182.5
184.1
187.2
190.3
200.5
13.6
17.8
18.3
18.3
18.4
18.1
20.3
19.3
20.7
19.0
19.8
19.4
20.6
19.4
201.0
19.8
206.2
240.8
260.4
266.9
293.6
292.5
295.7
294.3
294.3
297.7
300.8
19.6
20.8
20.2
20.2
21.7
20.8
20.8
20.0
20.3
20.2
20.3
28.1
30.6
34.7
54.1
2.7
3.1
3.8
9.3
71.5
9.3
80.6
11.5
82.4
112.1
125.8
145.0
148.4
156.7
161.7
161.7
179.5
194.6
202.6
214.2
254.4
267.4
289.6
298.5
239.5
11.9
13.3
14.6
15.2
15.7
16.1
16.5
15.9
15.9
16.0
16.1
16.6
16.2
16.4
16.3
16.2
34.9
- LI-
Colligan/Bayles
AT (K)
velocity
\T (K)
L ~p---
Schleip
velocity
(m/s)
(m/s)
23.0
31.2
36.4
42.4
46.1
54.2
60.2
55.0
68.4
63.9
79.5
80.2
79.5
78.8
76.5
78.0
80.2
78.0
89.9
95.1
95.8
102.5
94.4
112.9
115.2
116.7
118.1
116.7
111.5
115.2
117.4
128.5
130.0
1.0
1.5
2.3
2.8
3.5
4.3
5.2
3.6
5.2
5.8
5.8
6.3
6.6
6.9
7.7
8.4
9.1
9.3
8.4
8.9
9.2
9.9
11.3
14.5
14.5
14.5
16.7
17.5
17.9
17.9
17.9
19.6
20.0
67.6
81.4
89.0
96.9
106.0
108.9
118.6
128.2
0.9
2.2
3.7
5.1
5.0
7.4
8.6
7.8
130.6
8.0
134.7
136.7
147.5
148.1
151.1
9.9
12.1
12.2
17.4
17.7
151.6
18.5
156.0
174.2
17.8
31.1
182.1
34.4
184.7
185.3
200.2
202.0
188.8
209.0
34.2
34.7
40.5
40.1
45.1
39.9
209.0
210.2
208.7
221.3
228.9
238.9
253.5
264.1
303.9
41.4
43.,
47.6
48.4
44.(
50.E
56.(
55.E
66.(
1
323.21
72.2
Lum
(this work)
AT (K)
velocity
(m/s)
262
41
296
101
143
110
128
187
113
93
287
283
285
284
280
203
273
74
131
119
122
236
46
11
19
12
20
35
11
8.2
47
50
46
45
46
32
38
4.6
28
16
19
38
Part II: Fe-Ni
Barth FCC
Fe-25 at.%
Barth FCC
Ni
velocity
(m/s)
AT (K)
73.3
77.1
88.5
93.4
96.7
97.7
110.8
122.7
144.4
150.4
153.1
174.8
179.2
180.8
196.0
197.7
198.7
203.1
211.2
217.2
,.,,~mmw
MM "
AT (K)
Fe-30 at.%
Ni
velocity
(m/s)
2.7
1.9
2.1
3.0
6.0
5.1
4.1
9.5
14.9
17.7
10.8
40.2
30.3
32.3
32.9
30.2
32.1
31.5
30.9
34.4
69.4
85.1
93.2
95.9
101.3
118.7
137.1
139.2
142.0
147.4
172.8
175.5
189.1
202.1
203.7
205.3
4.1
4.5
4.7
7.3
5.3
6.2
14.7
11.1
10.6
13.5
22.1
16.8
26.5
22.0
33.6
31.2
Lum BCC Fe-lO wt.%
Ni
velocity
AT (K)
Lum BCC Fe-12 wt.%
Ni
velocity
(m/s)
AT (K)
(m/s)
238
243
240
181
237
233
132
154
227
143
135
244
145
91
185
155
62
185
229
170
32.8
27.5
31
19.9
27.91
35.3
6.5
11.4
33.3
12.4
8.6
32.9
14.5
4.2
21.1
7
5.2
16.4
32.7
13.9
195
117
114
22.8
13.6
16.5
104
138
8
14.6
222
167
186
113
110
28
17.1
23.7
14.1
15.6
214
26.3
195
226
228
167
136
95
152
195
161
25.4
37.2
32.8
19.8
15.9
6.1
12.7
20.6
14.7
A4. Computer Code
The following pages contain the original computer code developed by the author
during this work.
Listed first is "Nic-xfer.c" (three pages), the routine used to translate Nicolet
oscilloscope data from an older, unwieldy "LOTUS 1-2-3" format to a more usable tabdelimited column format.
Next is "mk-cyl-data.c" (two pages), the program which input text-style
composite interface IPLab images and produced datafiles suitable for input to the
solidification model (cylsiml l.c), simultaneously querying the user for various calibration
values.
Listed finally is "cylsimll.c" (seventeen pages).
This is the computer-based
solidification model which made possible the determination of the solidification velocities
of the Ni and Fe-Ni specimens investigated in this work.
NNNNN
**444
N N
0a0-,0e0
CooDCDCCDC
H, mH,
H H H
CDCDCDCDCD
r
CDCDDCDC
rr:E- ?t:ý
G
:3- (D 0
CD
H.l
r-C
)
0
-E
D
'
C?'0
Dc
C?
tQ (D ID
Di'-
tott
0D MI U) q-
'0-
Di'0'-
F
1r-. *3
ID
C'9
It-I
h,
Di
qO~c
0
u
nCw
00
too~
I-D
CD
C)
.
I)
rrwCD nr, r) tr
SH-.
CDVCD
·
-CDM
D
(D
030
(C
Ok,
I-
(D
1-?
I i
DC·-
'-00 f.
Di.(
Cm
C-0-
-
-t
H.
t3'
mt-h2
CD'-?,
CH.CrIC
-
0CDC~
0
cDC~
oc
k
o
I--C? H..H
I-
OCD
iCD
DC
C
1)
cDCCD
'-c,
C)0n
DDCD
CDCC
CD
C
H.
I-
-,CNh
CI
c'
a
CD
DC
4iC
tnn
c~
-
H. Di
O C?
0-cD
IC C~ l
0,?C
nX~
zy
C
N
C
-
CDC
l-'i
H.,
DCDCC
CrD
CD
'1
0-D
0.
0-CC
N
*
*,
Di?
CoS
Hi
U)
CD
cDCD
I-C
COD
CD
C?
H.IC
*
'0
ID.,
'---C
CDI-
CD
CDCm
IC
C)(0
c,
CDC
C)
0,
CD-I-,C)
s
0-
CD-
Hi
aI).I
N
t,
0'
,
CDt
0,
rDr Fl
C?
cD
-.
t
DiD~IC
CD
D
1-?
I--
lt-
C
N~'-,O
CD
H C?'
·.
lDC
CDCDH
0-C
)
C).CD-
CC
*
*
Di
C
iDCoC
-
II
~~I_
D 'w0'
(D
D:?-HI
:Dr .
II
-C'Di)
Srr
(D
II
I-h
,
m
0.(
-0
'0CD
(D :1CD-I
(D
C
II
~
rT
C
..
rr rt
.1
CDC '-D--C
'0
'0
'0
II
II
-'
-9
-
U~~"
H'C
II
-
t-
-*
-
DI-*
IDh
II
P'
II
II
II
00
1-)
-
-
"
d
H
-
\D
- H, C? C
,H,
C? H
C? -M
, H,.
C? dP
H, H,
C? H,
C?
H, H
C? d
H,
5- H,'.H,
000
-
C
*0)
C
flO-
C(D
''0
Cr
II
II
II
ii
0C C
0
0
HHHH
--
--
CHa
0
r
, C?
CDODH
P
t) CD 0
H C?
(D-C(D
CD H- CD H
0
rr
4•C
H
C
H
t-h - I,C F"(D0 m ::IW :J M ý
DC
DJCD
0drt ,. M*dp :j' rt ,. ttj.odp 10 10,
44o•'p
CD C
'1 '(D c..DCD
C? r'C :C- (D
(D
3 (D .D ( FD,
IDcDC, .Dw
(DH-(D
H
o QD (D
W
R
CDrR'1•-.
?CCC?1-I'(
c)EDCD
r'-j
R'CD RCD
D)
H'H'-'t4
C(DP- "'CD
(D
++D
HID
NN'
'
rr rrrr
r)
0f~
HC
ii--f
I cJS
-*
'
P-H F- H-HP-
H)
'0
-3
0 D '0'
rCDCDfDCDCD
rT
rT rr rr rt
r M M M t-h M
n~
3QHO-3C)oHC
4
D)
)IH
-
rr r
D')C
)w)f
*3C
(
-*
(
t:j
F
'
H'CDO
-
*
I-
w 0
0
O
-
C) '(D 38 CD-n o'
(D
" 11H-LnL--
H'-0
CD-
0
'
00
*
aa
J
u.rg
)-
(DM
0-H
-
1
CD
*
4.0
*CD
(D
(D'0
'HzH
,
0.0
C
Ln
1-)
ý-o
(Dr·w
Ln"
D
*Cti
-·r
-
'
rt0
DO
.
C?
CD 0
H
0
rr
CD 0
C"
Ch
-
H---.
CD
H,
rT
tC
..
C
Jrri tCD
o Dto
1(D
D
10
rr
:
37
D
rD
H-CH'D
rrT o
rr(t
CD
D H
CH'C D
DI
-
CD
00
*DC
4
CD
onro (D
*0D
4DC
*CD
C)~
CD
'0
CD
H'
0
0 (D
0
O~ (D
H,
C
C?
CD
mmn
ICC CC)
C4'~
CD
DC D C
000
HH
, -,
Ir::
r
HCI
*01
CD
i
1
-
4
z z zIU
k(
\o
I
rrrs
OH.
rmD
4
41)
· i.i
N
o
O(D rt
(DODC
DCCD
tn
*
*
4
ri 0
,0rC
0n
Di
eCH.
(D
0
1*1i-
Di
-
'0
I-.
CD-tnrBi
(D -i'-
H( D
rca
ht:r~
CDCD
I-
H-D
*DC
CD
C·
0,
I1I CII
I
'.1
0
N N
Crrrt
H
N,
LLL'O'0
(D (D
t'i f-
Cr
'0 -'0 - H-
"0~
-CD
0000
++H
H-0HP. tL
Crrr (
'000
tI-H -(D1
M
Sm
A
0
Cr0Cr0--H
CD CD
rrHH'' r"-Hr :j
0 0Cr
'0
'0
P.
00
P.
0(
LI
P..
rD
o~ H
HO--1:
0 H- a 0H D-CO rCCrr
0C
rrr~r
Cr
CrH
rr rt
H·-
M
rr
140
Cr0
CD
CrLI.A
*z
*0
*
*
"' Inl
A
1C
I
0
P.
0
CDI
0
*
*
H
0:r
*LO *
*0
*
CCH
*
*'LI
*
H
-,,
.II
m) h
C)L3.D
+ WHH IIt
0- mU
CLp
H* 01.4 -CC'~lO-CC
.
-CD +t
dP dp
x.
*0-'
*t
CJ H
c
0
-
0H
5
-..H
H,
*0(
*0
*j
U.
IH
H
H
H
-H.
ra
LCD-H-
*
*0
0
0
+
n
+
14
H-
C
H
L
CD CD
.
./
.---.
t:1
tj DL CD
*0
*0
Cr
ro
I
-H
CDCD00--....
.
t OHC
Y,
o
•
*I
CCLI
*00-
*
r Cr
r·
r
.
U P.--
. 0
-Cr-
0)Ill
*00·
Cr
pl-hPh0
I
--
0-0
0\
(D'0
0
C
-
'0'0'0
000
0
H- H
000~r
Cr C C
CDo(I
rH
It CCT
r
" 11rtCDC(D0
:: :ftjH- if HFý 0 -r : - rr 0- f,- rt
0p
*0,10
*0
CD
*00D
*
CD~'
*" II H
*
Hc :HO
m
'0'0'0
000· r
ML
H00
rt
*
--
C
*0
*
0
05n
'*
0
000d
*C
00
0
F-1
CD
C
CD
C
Cr
0
C
CC
-
0
L
Cl
0)
0'
*
ii
CtO
CD
to
CD.
0
N
1:1Cl
D
Cr
I
H-3.
z
0D
,,•
+
n
HH
0I
H
CDn
Pr
~IC
(D I
*N 1.C
CC
Cr
CD O
If
(D 0C
ItO
0-•
rm·
00
0
CD
0
d'U
o
CD
0
0
0
Cr0-Cr
CD0
00lr
-CD cD
OCD
0
CLC H.
-(DB
(
tCrr
H
r0
'0 rC
H~n
rr
01
'0C
Q0
0
0
0CD
0
'00n
-
•
0
ICD
01--
'
-CD
rr
m t
n
H-
0 Z C
I
o0 LI
En r
HO
(DH
CD
*
CD -or
CD
0.
CD
to
0
vOC
Cr0.
0-o
CO
CD
CC --
0 0
'0*
0
HO0
0
H
CD
*l
*
C
1
r- (iD
1- a
rH
i0.
*
C
rto
Crc
KUCD
CO
C/lCD
*
F-hC2l(D .*
0
*
D rtH0 *
Srrr0*1
**
0
00*
C-CDC
*Ci
*
*
*
*
OnrCD*
On
CD
00-00*
ClrCOCD
CCI,
*
CC
HH.HCD
0cCD
CC
iCC
00
(D7
0-HOCD
I
0
0' 0~
n
0
CDO
-I
CDOa
0m
0
0
r
*0H·-CC0b 0CD .
*
0r
'L
0
C-
O
'0
car0
CD
CD OC
-'0
cD
0.
CD
CD
CD
OCD
HO
I-he
HOCDIO
.CD
n,
C
H
Of
H
l'O
N
'0*(I
0?
CD
~'t
r
i.-
CC '0Hr 0- CD 0
CD
oo
H
0\
O
H
-
•p,y
cu000
rt
CD.
CD
H.
m
'00
10Cr
(D
tzj
m 11
- rr
tooa
o-0
03
o
I"
H
-
0
CD0
to
0
P"
r
\0
cD
0
01
CD
0
CD
CD
CrC
*1n
CD
09
0.
h
CC
CCC
Cr0
CD
CO
CDI
Cr
H
0
OC
C
CD
0n
0n
CD
'0
CD
CD
CC
CC
CC
CC
CC
CCCa
OCCHC
CC
000
CC
H
0
0
00
D D
0
0C
CDr0-Ci
Ci
C
CI
C
~
tCd~C
(a (n
(CD
0 rrry
.
(D 1
r: r
rtCD
"
n - 11
rt
0
~ 'lCD Sb~
:
0C
D
D
-r¶
(D U)
-lid1
r)00
CC
tr 2'l
F
11
CDI*
C-C
00
Zo S
(D
C1
r
(D
D
FI
C-CC-
C)
o
--
CD
C
nC
r
CD
0o
o
C-rC
0D
C--
(D
t.
CD-
ICO
10C91 ,
(D '
C-i·
CDD
0
Cl,
tzj
0
ýo
'rt 'I +r C)
ft
rfl
CD-D
-~C-+C
:j
rr
rIIt 11C bCD
f
ý W0(D
tC0
ýl
-
(D
CCCCqC
H
-
CD
CD
0
O
CD
(D
·-
-·
a
m
Zm
~
. c
C-Cl t-h
no
rrDC(Ds
C
--
C-Cr)
Orr
'
N
8CD
n
CD
C
0
CD
O
80
0DC
*C C-
C-i,
C-
-
(D
Cl
--
0.
0
D0
C"c
~q
100
CF
(D
-
rr
92:
CD
II
2CQ
In3
(D
CD
C
CD
C-t
(0
CC
0
(D(
0
o'10
h.CCch.
Fl
CDCD
0
CD
O
01
0
+
C?
+ rt
(D
tl)
D(Q
Dr
+0
C33
cct 0C
f
'
rt
Ii
0*N
c
"D rt
r- ++
(
0I
-'
"'
I
1
CD
CC
C-i,
CD
C-
~
C'00
4*NN
3t
:w
'r00
0
C)N
9) rt
tcr
(2
rt
00
oo
AA
Di~o
Drta
N0 H
1-4ý
N
Ct.. f)
:
00CC
0
N. N.DC
N
nD
0
N. N.
I-Ic-.
DC
Lao
CD Ib N.D
I
t3
)~t
00
-Co
I
1-
t3U
:3,5
rD (DCr-
ION
O
0 a~
CotoN.
Co·i
DCDi rtktc
:3
O nHDi
l
rr
(D C1 rt
nO Ci)CDC(D
DC.O
0' N
-0
txo
N.
Co
-o.
En
r-CO
a,
nnC1t
(D(D
036
v
CD
000
00
Hr)C
IC
On
(b
CoDC
rD
CL
Di (
0DiN. n1
Co
ý
CD
OCo
CO
Co-
N.-.N.C
o-Fl
Ofo
C
0-00"
-i
Coo
np
rN(n
(D Con
ti0
o
0I
COO
ýo
Cl
N,
00~
nN
Cn,
(DO
N.o
ON.h C)
IOCCO 0-rrC).
N.0
co
OCT
(D~b
C11
C
N.
CDo~l
CoO
Co .
Oi CorCo
(q rt
O
H
I-I
Co
~COCcO
(DDi
0a
CoO-
CCCm .~D
to
SC
cnO
N.
c
C
N.
Co
00
COa
ICC)C
H
C
'It,2OHu1
'oC
,
C)
13
QO)
CO%
Coro
+n
COCO o
nýl
(
Co tI 0-0i
0
N
N. Coho,
H,
DC00,
C-1'-0
Co
Co
cD~
m9~s
CloC
Co
0 Dron.
(D(
CO'0
Coý
N.
-CoS
0~
CD 40 )0C
.
COn
'ao CD~t
co
10
Col
c.oro
1
n RC
,
CDD
(r
D N-·
C)
~ Co
ctoO
C)Co
t 7 :
!Zo Co
'-3Cuo
(P
tr
N.c),
O0C
tzl
QODI
oCO
Co
o
Cao
o
Oq
Fl D
(OO
riO
N,
nz
100
Cl
nDCC
CoO
Hc
C) t'3 >4'C)
H'-4D
0-Co
00.rr~(D
Sb
~icoOco'
0r)
Oao
13
0
C
Clno
Co
nO Con
0-CoO DC
13tr3
Co
Co
0?*3
C)
Co
'-
tri
H\oCO
A
'-Di
NNN
0m
0
(i
r
(D
)tlI-'2)
.1;c
Pli
L.
Fh
(Dv >3
(D
0
O
N
C
I
..
N- N- Mi-·u
r ,
Cozi mj
to 0
1):-CoI..,-
(D
P.
-rtN-O
f:1 rA
cDi
0)CFN-
f..N·r -NC)
'Ot'0
Co
Di
N- 84 C mi
(n
0*3
OC
ý3
COO
m
00
+QS
++
-
CO-
ti
R,
-(D
0
Co
Co o*
OD
Con
(D (tD
iC
M
(DMi
C)
N*
C)
N
t-hr
CC Mi
Ho
MD
-
(D -
((D
tt
(Do(
t-h
cos oko-
(D
is
(D-
>C.
u 0
Co
Co
N-
1-
+ P.
>1-
-C
(Dc
0 to
Iti
H
rr
'
'
N-
Co
5r
0"CC
Co
i
'1
CC0H
CCC
4
rtr
Co<
r,
(o~
>0
0
0
to
H
'-'
3 !;2:
·
Ct0
'4
O7)
Cn-
a
(D-
00
(D-D
C
H
o C)
c)2
(D(D~
Co
r-
+
01
o Co DC)"
0
Co
D.
. wi
(D
in
(DC4
ON-
Co
cr
0
C)CC L
Co C).
rr
Co
C
(D
N
XCC C).
(DC)C
N-
g.01
0-N
N-(D
(D
0o
r
0'
H
00
0-diN
m
CC
0'
HD Co
rr3C)
IC Crt
tý
t-hOnPiN-A
+ n0.'
'000
-C-C-Mi-.
-.
P.CC?-h~
dp
3 -C)Com
i OH
tCC C
(D(
tzi
130
Cori
o't
X-rC
rtC
Cj-Cf
::1
CoCoo
LO Hr
Co
---
0 Enh
I.
S3
CO
-C
Co
~-
H
-H
'00:
wti
t~j
a a an a an Ccaaa <
m m w
C -- +
rCTC +
-Ch ?h
'0
dP
-d
-- nd
ap
VA
CCp
-
191DR
(- D-j HCCPa
T
I'l
R,
CD
aca
a
H,--
na
M tti~t
MCIH
~
tDo -w
)
H'
l
(D R
0
H
**
~
CO
a
P
C
E
- F-t aO (D H "D
(D -(DO0
mi
wa a
rC PIH*D'
l<C-Ca X
~C
P.~
t§X
C.VDNC0C1 0
iC DR,
CD
(D
:F dz
u z
0) a Id
ID
C
ý-l
N
+
01+
-•
a!
H-hSD-- (D
(DCD- tr:-F(D--
(D
-CC
ilr-rHCD
rt
rr
CTHf-
nCD)
(D
(D
H
En
r-
H-
XCD
CT
(D
j
C-h
(D
wD
(n
0'
0D
mt
En
na
OH(
C-tD
0H
0D
(
a
F
0
0r
CC-'
0
w )C
HC--
CD
C
(D CADInT
CTý
H
(tD
CDDn
aCDDC
H r
CT
M' (DC(D
(n
H-HHr--(D
CCt0-(D
P- L< pD
-Ci.H
--C
0-l
'
(D
(D
(D
:E "C
10
0- 0D-
CDH
-wD Q
11 CD
CD
1-1
-D
a=
H
a0
"
CD
i0-
0g "
aD
CD
aC
0-C
rD 0 F. Q
-H--(D "C
D) i-C
CE; -(D
HfaC
CT(D (D
Cr-
0Cn 0 C)
*1
IlC
1a
0
CDC
(D
OX
F- I
int
OCT
D
C
In
P.+
ai
C-tD
CD
InH'-0
w
0D
CDH
C ýO
CN
ýuý
t
ýCD
0-
0cC
0-Hl.
13~
:in
tr
CT
(D
c
0
IOan
i
CD
D
(T
DD
CDC"
O
-
CD
(D QC
CDU
CT j
Hr
P.
(A
CD
F
---
C.
N
I--C
N
CD
CD
t
t-,*
(D
• 0
-0 z'-H
CD
+
+ 0
-1
En Q
I0
FHD
a
0p,
to
Cb
CDC
ý'au
(D
O~
Ca
a 0Cio
"
a
OHi
C7r)I
CD
t-h
a< w
<0
rtiI..
(D 9t,
"
CD w
(D
~
0-C
-.Ill
z
0
<OC
Ha
D
CDV r
(0D
(D
aoa
CDO.
w 5D
ýl
D
C
0-'
P.CEnt
In (D <
CL
a WCD
(-
O
H-C
(D
rt (-D
C 0C
(H- PV
0 CDX
tVW(
ýl
DC
P. Wh
CC
CT
CCO
H---
CD
DD
C
0DC(D-C~
P.
C
H
n*OOC (CD
.
11W
MDCT
:YiWC(D 0C
CDC (D
CD-
C
OD
D~a
P*-)
C(H
-n
Ml
a-
, 0CaC
D F-1 x
0- N0(D
0n
nth0)
I,
P)
on
CD
at
-CCr
CcI
H
CDH -C(DC(D
Z
1D(D- 0
aH
P)~
Ci
-MOCCCD
r
ID
P.
(D
CD
CD
CD
0D-t
(D
CCH
oj
t- H-a C
a-CC CA
CDa
'0 C-
H0 CT CT
00
;j
(D
--a
A
-
CD
0-
dp
A
If
CD
V +
CCiCe 0-
CeO
0
-Co
C
-CTC
CCC.a
A
CD•
CD
11
--- "CD
-
X.•
•
-'
-0
C
(D-C
C
CC D
0
CD
INNNN,
NNNNNNNNNNNN1-1NNNN1111NNNNNI
N
N
N~~1
Y
*1 *1 *
N
L'i
t
N
nN
(D (n
Q
~I
to
h.1cn.
'
1*-
r).
0.(D
(
0T
Zi
0-( n
000
:z0
I
.l
0
0D
(OnD
D
ON.(
N.
Fj11
I
0
nno
N.jO'
0.1
-.00
Z)
000
000
(D0
fl
n0LQ
Qn
ri n
C0 0
110N
(0
00(
0.n1
0D
0
-.101
(n o
000000
N
Nr
*0
0
U,
10
0
NNNN
010
01
~
(0
'0
I
(0
0
''0
'(0
('1
'0
0
I*~
(0
'0
IN,
it~
10
(..
I
I
I
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
* I
*1
*0
01
*0
*0
~
01
~(0
*0
'-SI
*n
(01
0
*0
I
I
0
*0
*0
*0
to
01
*
'1
0
0i
01
'0
*0
*0'
*0
0'
1
00
o
On-O
0r
0
'0
(
0r
N.
(0
I
I
I
I
0I
(0'
N.
0
0
0-
1
(D 0
10*
-
NN
NN
-1(0
"n3
0-ON o
(01
Q(D(
00
011D
)
0[On(
OnNan-.(0
-
00l
OON
0a
n)G "
0001
00
0
lo
QO
00011
0.0
11
I0-0C
0D(0
00n
00
ý
0(D
nj
:31
nO
0
01n
Q.'-0D
I
NrNj
Ni ND Ný N
00
*D
*t
nO
*
N
n11
Nl
00 r
(nr
Dý (
0
t
t*
f
(D
*
*z
(nD0Ot
0
00
Fl
0(n
'
*
00
l(
r-D
00
1
) w~.
D.
*D
0D
On0
0
O
01a0.
U)
n1
OJ0'
l'
(00D0
.
0
D*(
Ok,
n<
0j
a
t
NNNNNNNN
DN Di
N, N, N1 NN
0
0
0
0
0
NN
N
NNNNNNNNýNN
NNNNNNNN
N.
0
0
n
0
0
0
0
0( 0
.
0'
`4
0 r
0
(
1
'( 00
00N
(0
0
(0
N.n
11
r-t0
r~t
N.N.
n1k-01(
00
r 00()
z- -
0
(
(0nj
D
I
I
I
I
I
I
0
nO0
N.(
(D.0
0
Z3
00'
N.
ý
N.
0'(
0-
0
N
n
0-NO
.0
((0
0
11
0
0
000
rtn.
o
0-0r
.(
n
.
n(D
0 n
10 NI.
nO
n
0
on
nOn
0Nt
00
0
0
S 0
N.N
11 nO
N.ON.F
(n 1 (D
(0 0-0
nO 0.0
ON
i0..
0
0
(0
0.
1(D
(D D.
0.0
rtN.(A
nN
0
000I
n
(D1
:0011N
(00
0
1 0N
( 0 N.(
0(0
.
*N.
*10.
0t
N
'1 •
n
00 1 (0
11
(0N.
(0
Q
On3 ON.r
u
0
001
0 t
DNi-
0.N,10
00n0
N.i
r 00
011 N.0
0
00(Drl
*
*
*0
N
D
01 0 0
0
Q0j
*0'
Q,0nON
11
*17'
*0
*01
D
0
0
*
*0
0
n 3
00-
'0
:b0
t
0I.N 01 O,<
N
n
0
0o
ko
E.n nb 09
'to
0-'
~0
N
NN
UN.
~I_
-. IN
t*l
(010
0
0
NN
*
0*-
(0
D 0o
0
w
.0
0
NN
m'0 a*
'0,0r- ,
0M.
n".0(0'
N
00
r1
p0
'11Ci
(n
NN
0
W00(
0
N.
Di
0
On
O 3
(D-'0
*3 nn
F1OO
'(D 1
in.0
.
0 (D t
t11 (D
n(D MO
n
0.
0-h 10-l
011'
:Z.
0-
NNN
0
00
2(0 t. tl-(
'(D
0
nOrt W''
t 0 (D 30
fl000'rr
000
0
03 (DnN
00 1100
00-1z
01
00-
IDn
(0.
(Q00*1
010u0
00-N.i
.
0
01
'
(D
.
0r0
(0000
0CN.
0
0
(0 M
(D0n
11
.
'
<
N
11000
O
00
000(
'
~
n
pi
()00" I
o1
n
,a0
0' 0(
0
lN.N
00101
0D
n
(D
DrQ (0
nO0n
n-D
t.n
n -(D (0DN
(0N.-
N
0.
0.
0.0
(V(V1V.V(.V.V.c(.(.
0.
.
.
(D (D
(D0 (D (D (0000
<>4
j
M
WZZ
t7'M
-
I
r w
1
'-I
*4.w
A A
H4
*r (Q00
En
(D4V3HV
curt
(t.* ,
V
o
m
40
(VT.
*0tn
UnC)L
0
0
0000
nj.
00
0
0
0
0
tQ
Uf0('3
N
()%V(
.r
0
4
0
00 Q)0
0
ij 4k.
0
4
0
NJ
N
~
t
.
IQtjýQ
.
.O0N
0l(D
C
" " 04.
HH t(
13
t2
H
HH0
NINH
*
*(Vfn:
*(VVt
N
0N
N
H(~(
t4H
*04
'0C
D~*
r)t
C) Q( t
DI..
4-
.0
4(V
(D a
HH34
"1
H
4
01
(DOH
tr30
n
4.
t
00 (D
(D:
Fj
0.
En
I'
"
0
0
0
1
(V
00
0D
(V
(V
t~
O
4t ct
ft
t
PA
0
H
~1.4~~
~~ ~ ~~~~~~ 1~~,gP~ ~~ ~ ~
~~ ~~~~~~~~
~ C rn ~H Hm ~
0
Or4
(V
0
0
r-(-'(1
Z-
...
.
O
O
PA(V*
rt
H(0
Or0HP.
H FA
*D
CD 0V V
.
000
0
<
0o
-
(
(4
0r
00
0
0''c
0.0
~
(D
0
Hl
0A
0(
0
0
1
-.
0
00
0r
1
Wf
t
w
N
##:f
t
N
9
0
D(D(ND(
.H
4011 -
(VOrt
0 r-
4
0
O
4
.
(D .
02
.0
44
W(
N
0-
.
0.
1
(0
4r0(1-
4r9
~
o
(0(V
(D rt
rV02~( 4.
2:~r
(10
4
(D
*
4.~~F
4
0
000
(1
.
4
00'
0
4 (n
~84
(V
t, 000
00
0
En
(
&
(0
4.
C
HZ
H
013'
00(V*
N
t
-H
(-..(V
40
wn
010
(DtO
:.* lH.
~ ((.V
t3
*0
H
m
0
HO
40
C)~0
NtnC)t
H
(D (D (D (D~~~N
(DD(
D(
~(D
(
~ P th""rhth"m"
N
(000
.
.P
H3
.PHD0.
D
:3 t0 CA120
Z
*
(V( 0V
;1
HHc00
00
N
0.
b,
#f
4
(Vb
0b
0
t( QN'
H
N
'3
C)~
N
.P
D(
.H
3ý j:
jý t ýý 3z:
m m m m NmO
m
m
m
m
NNNN
((1
(
0.0
N*N
0
0~
0
(1
0
00
*(
PAgQ
00
0.
0
0.
~
(
-.
D
(D*0
H
0l
'0
.F
lý
m
-
-I-
NDfrIt j:
m
1
x
0
o.
0
~
~
3
Hs
0D(*wZ
0
a-
C)
CD
ý0
0
4u
n
rt
t
ri
9(1
40o ()
*Z
N3
t0~H
O
2
404j
4z
t
N
HC
IN'
N
OHO.
N
t"H tiN
4tz
En
(Jl
Q
a,4
.
41
)
0030
Q
(*I(
o
4000
4,
Q) t
Q
0003
H
0
00
.'
(NJ
0C)
U
(jM
w4.(In.V
42:
1'
.
(
(V
*
.
(
0
4)
4(bVI2:(C
Y
0
co
(4.
0H
t-(-- 4
0
00
0
I., C>
oN0W4
1 Ln CJ0(OO
a
*D
(V 0oJU(~
0 L(VL
(DOC
IH VUO(.(Vý
Ln 0%"
4
0
(D D
N0
4
*.0.4.
0 00
P(4( .( . . . th4M M(
*PH PH F- PH P- P P-H- H-HQ4*ý4*
Y,
I
..-.-
4(3
00
WTýC
2
-.
00
~
C
-l0
3
0
H (
H
021'
O-110
000
H.-..
H
0A
0
0
c?
0 000
0
0
0 :rW
'C0
<0100 P) ;
W
rt o 0
9XC
-
0
HCJD
0
C-0
x0 0
0C~0ID? 0 4
-
o
.-.
LODC
CO
Cr*-.
--
XCD
01003H
pi
I
(D
~
ý- 0
;4-z4
t
0PA
0
(D
Ir
l,
0
D
0W
*
-
*
(D
(D
00 0
10
0
0
(D
0 0'
N0
0,0 0
PA
(- (D
0)
o0Cv
(D
0
L<0
W-J0r
-
0
-0
0-<
CJH
'I0C
?WC
t-h
CD
*
O
**3-
0
C
*-
0 D0co
D
*
0,
3D~
CD
xN
-
(D D :---
0
0-
nD- w
HC0
-
'OC
OC
0
-
-
-
0
0
'I* OC(
0
0-D*
00
*0 t
-- b-
0D
C0'
C; N
OCD0
D
000
Na
000
0 0
0
*,
0~
0D:
0
9:-'
*00
H. &
(D0(
000
00
oD
N
CDC 0
-C
*
CDnO0
*CCD
0*-.
-0
(D
0
b-D
-- 00
0
zj
CD
0 0H
0
Fl <
00
00
0
00
(DCD
Z
DCD
0
0
*
*0-C
C?
'000
P.
00'
*3D .
UC--'
0O
-
(D
0
0 MCD
0
0 0
r -
*
0
N NN
N
loCa
H•-OW
CD
*00CCD
CDJ
D0
it
'C'
00bFt
C?'
Q)-
'-0
?C
i
-~0
:
'C
C
I
0
(W " D
,a
-l
-'0t
t1C'
(DCq
0
0
00
C
l
C?--D
'-Ci
0
C
rr
*0*
HCH
o
CD *
Pi
03
HD
0s1I
HI-
C?
C
(DO
I 00
rr
.
(D -HrtC
Do
o(
-?
(D
0
000'")
P.C"
'
'0 .0 *0
t0 CC
H
*
00
C?D~
0C C
C
0l
OWN00-
0 0
0 00
Hr(
0*
0
Di
NC?
c?'
H-C?
Wc?
OW
'< (D
.00
O N CD'
- H
..3
0T C?'0
H-D
0'
(D
0(
CD
0,0)
C?0
0. 0A
0 CO 00
0 0 (D
('D Z
3-'.
00
0
00
**
D'--
-r00
-(DC- rr
ýj0CC?"HC?0-1
'00r-10
0j0
-• 0
z (tOr 0 0 CD 'd0
S?
r-,.-.r
(D
H H
(D rr -H-m
CCCC?
D
NN
3--D
H r) C0
0. .0I0.
0
00r
-i0
otlr
'00
'-*
P0
C?*CC
C? 0CCDZ
d*
'*-C?
-O
00-
D'0'
CDHH
P. 0 Di0 0. 0n -- 0)
03-.-
W-*N0
(D
0<
-0W
11
CD
'0
0)
OWýr
C?
0
(D
0, -
CDH
0,
(D
0(D
tn
N.NN
"N
N
N N N
0
0 4'-'0
*0
40
CD 0'r
C1
0 -000
CD
l
Di t
"
1140
114 W1 01
*
40
4 CD.
0
0t
dP' (D
'0-
*DJ
4-4
(D
D C
r_
i
-
11 Wi
I-10
4'''"
0·-00 CD
D, -'Di
00'
Sb
4C.C 0
CD 0
XD
m
rt
t t4*D
ýL~~~f
oDCD
-(
0
~Fl(DO a tý0
:1
0'
0
1ifiC
DP,(
D
C-'
0
CD
C-'L'C
0
CD"
(D'
~00
114
CD
+
ftC-
CD
(D 'fCO
4 CD
4C u D'
CD
0 0
0CD
(D-3
0r
"
040)
(D ttl(
(r.-
~~P
*
0
0
(D
40CD
0
0'-
V
4'
4
4'
r'
ir~
4
ft
(D E
0C0ODC
0-..
9) D
(CD
04(
~
040 :3C
~
(D
C0
x00
(DO
c40
(n" C
-03
011
"CD
C'In
r-
0'
0
:1 C-'
(D
(D
0
CnD
F- WC
(DD
-- 0lý3
(
N N
t C(DO
(D
"
-'00'(
:D D
CD"44CC--Of
r-- <
(DD"D-
0
ft
('D
CTDOý
4-O44
(DC-
400'
0l
(CD
0AQ
0
(D1
(DC-tCOCOD
?I W
ft
* 'Fl C
(D'
5'ý
W
00-CD-(D
Ftl CD CD
Di
.3CrWt(DC
0i'
-~
(D
4
N""
(tD
0t
P.C-C
(D
ýs0
ft-C
l<'
C- 0
0~ CD.
M4CM
40
o4 +i o
C-' (D
4
('
D Wi H- 0 Z
:
w - 000
C- t M:3
DCOO.
"
w4
'i-'
D
'ICDA
0o
-C-'C-'C-'C'C
rv CD En .3
40
40
40
4
(D
0ý
1o'0'''0'''
000000
11~C-. P
C '''
00t
N N
.3
Di
CD
(DE
I
N N N N N
0
(DN
*
00h
*
W0'W
0
(DNNN
D
i
Dthr
0
C-"
w
I
- w
I -r
0
-'
0
-
w
C--C(D
f-0
P- '0
( m O
-0-
t
ftC(D
00
M114
CDD
rrD
<0
OCD(
4COMOOCD
1
0-0400
- ftr
D
d
11 rz0C
l<fT
* F- do
4(
CC
.-1ý-' 0D...
ft0'
r
(DC(D
0
ftO
0 r
th'0
6*r
rr
t-.C(D
~
4-'
H
<J
ft0
rD
*~
0 C
0-
0 1 0
P.
i
P
f--M
00
0
04CDt
0
~
*r
CD
"CD
CD-ft
4
a
0 0~tDCC
-
CDCD *
t0
*Di0CD00'O
D
rrD OCD<f
0
Di4C-l
00'
:31C
O
(DO
Of
D
D
00
(D
P-Cr
'00ý
00
00
CD
H
0D -
Di
*
0
D0 000
OC D ftttff
4
4
CD CD- *
04CD 4
CD.
.3
0'*
*0
*0
0CD
4
ftO
CD
*0
4
*OOCD
*OCDO
4
CDO
*CDCDCD
*00
*
4
4
4-4 ft
4
*4--'
0
404
0
*CD-.3 0
*4- 4 CDCD
*4-.
ft
*0
04Di
*Di 00
* ft 0 ft
CD
~ CD.
4 0 ft
4
*0
4
*
4
*0
*00'
*00
*4-00
*0400
4
4
4
4
4
4
4
4
4
004*
40
*ft
ft040
0
CD
CD
4
4
N
-
C-hC CD H.
(-
'P.
ftC--OfW
C-ft-
ft
0
4CD
0
0
04*
4
*CD
40
o\'0(D- (D
O
D 0
'0 OQ
n' (D
-o ftOz::r N
CD
FO0"CDC(D
C-<
t- ftO DC "
~
4
4
4
4
4
4
*
*
ftDi
4*
Di0 *
~0'CD-CD
*
*OCD
*
4ft
C--. *
4
CD-CD. *
CD 4
4.30
hCOftft 4
*
OCD *
*
n D
4
*
*OCO
4
C-
CnC
fCN
t
0th 11
04
IP
*000
*0
0'CD
~0Di0
ftO CD
*0
00--
UCD0D
ftCD4-.C-1,04CD
<'
<0
040~ 40
CDD
00t
0 1'0 10 10'
pliOO.
CD0
I-'
-'
a00m
*
*
*
*
CD.
4--4 4
"44*
N N N N
-00
.th
0'
0
N
NNN
N N
N
N
N N
*1
41
0.(4
419
1-4~1
411-4
0D
*
*141-*
*4 H. 4
a
HHH
041
414141
*
*
0(4*4*4*?
1( H.
*
*
*
*40
4*40
*1-441
41 *1
441
41>41---. 41 41
0 -. *4 41 .H. *1
r.i
41 =.- I(D
1--I
*410
*
*
*
*1010
*
0
*
*4
*41-
*
(4(44
*1
-*4* *4
14-0
1~41
41
•o
0-4
U-
14-0
*
-(-(4
*
*
4
H..*1
4•
(4*
(-(10.4
4(44
41
4
H.
*40
*41m1
(D
(4
1I
4 H.
4(4
4
=
(44
o
4,.rt
*
Z1
'
-41-
(D
0*
ti(
*~0
0.(( W
(4 (D
4
41
04
--
H1
-1
Y*.0
ID
(I
-(D
*
41
t
4(44
*
*
tr
-- 0
(D 41
*0
441
-rr
(4
(D
*0
0
41
(D
(4
41
4
(D
4(4
(4
*
(4
*0.
41
*1
(4
*0
*10
0-41
0
0
*0-
toof
0(4
M
ýL11 (D (
- 404
1*
*
LQ*.H(
1-4
(4
(4(
(D
0.
H.
41*0.
*00
M
*0rT
*N*
II(
0
0-
rtl
41(D
-.
I(4
0*1
41
&
(4
*1
41
41-14
04
*
0
*41
*0
41-.
(D1
*4
4
41
4(4
4*1
4(4
*
4
6p
41.
(4
4(4
40
4(D*
*44
0
En10.--.
N
*41.
*0
4 1 rt rT
041
41 I-.
1-1•
*4(4
H.
.- a 10
1111 *
If l
*410
0
4
4 H..
4(4
*
41H..
41-0
(4•
(4
N
10 -In
*41
0
v(4
(4414
0
(D4*
41*1W
*41
*41.
4(44
(4414
(4*4
* H.
441
441
44141* *4(4
(441j
0
0(*
*0.
*
(40I
x410
2)
*1410(4
41
41
N
*4141
4
m "
*0
*0
*0
(-4
*N
*1
*
0
*1041
40
~ H.
(44
4)
41
0.41
)--,
t-'
44
4 H.
41.41.
o
*1-i
41 *4 *4*4*4
41-4
0-41
0•4
*4(4
0-N
41.
~*4
41(11
1•
0*4
*1
0*
'1*
4
4
41
NN
N N N
~
N
NN
N NCND
P10--
X
RS
*1*4
.P- (DI
4100
>4*
rr rT
mx
t1
10
(11
(D
10 P.1*0
C)
r'r
(D0
40 *4* :j-ZN
410f--0Q*<
10- H-3 ND
1N
0*
*4
*04
41
0
-(4
*'4
0
(D1 H0
4(*41
*(
*
*
D410*41414H.
*41
:r,
(D
P.- H.
rj
0
(4
41-.
(4
*0
*
*l
r~r
::r
400
C:0.
4*
1(r
t
>4
rr0
(
If*
41
*0
0
44
41
0-
*0
(
44
4100
0
0(D
41
0
t3414
*
1<
r~r
r-0.
10
0
(D4
4
*04
* 00
* *H-
*1 *
r'4
04
N
0
0
0
0-
-(
0*4
D
(44*0
0.1 4
*0
*0
(D44
41
(D
41
H-
04
4
t4
0~
0
41
0
N)
If
>
(4*4
41
*l
*0
(D
*
(400
.+
0D
X
*4
0v-
-17
lb. (D
0.
*1140
Ft4
I4
WNO
*0
pv
it 4(4 11 (D
l<
L<
+ (A,
L<
0
m*
0-f0
10~
(44
r-41D -D 0
(Dc
:0
toX-h -0
0
N'H
0 *
410 0
go
-E
:10:10101010rTri
*1*141*1*1r
414141-111
*44
d'm
0
S0
~
I"
j
4110
101010101010r
I"H-
rr
I7
*D
(4
4•
(D
0x
l<
0+
*0
0
0
*41
*Aft
~ ~ ~0OO
~01 00zzt
*
X
ý
*
H'
r
*0
H.'CF"t
F!1
*
*
0
H Di
P.l<
oD(D
I- - (::,
L ,<
+11*
*C
1
t1ý
**C- ** 0+.
0'
H
Mt
*'-**
*0
*
*l
0r
E
0
+.w)"--CD
0,.<
1-p
" 0F-I'( (D
d-C0,FR
--. oz4En
l Ii, D - DO
(C0
0'
0-H
404
CD
0
**.4
4:
4
RI
t
0
4C
CDo
P
0
Mt
-'
*
,4.*
o4
4
,,0
0
C.)
'-'
4 ,o
N,-C- -'
Cl
0
M
c)
)I'P.
F0
S *+
4D
*
..
C -. FI4*tt
.
-
'
- Ci-,C
*F-0
*l
+* 0
*
•-~~ (D-CD.
4FD
r 't
(CD'
~(
Dj En.
4
(D0
Ni
-.
1
0
0*
1Q00
- 0 r'0CDC0
*HO'
CDt
0' H0,*t
W (D0
CtH
ti (n11CDCD'0
*00
P
. CF
0'v
0'CDFNa 1*
0
*OCDF-I
03
D(C*ý WCM0C*OCDO
100
z
q
10
t<
0'
0*
'3
Cl * 04 000
*4
rt (D
H'-3
o
'' X'
"
H 0
(D CtCb, ;I 0rr V 4*c'F.tt
+LW
C
z'X
*
4-00
Z
'0i
'
Vt
*C1C)F.
CPV*
l0CD
'
0
*0
F-0,
0D
0r
'N
*C
O C
0
ZO)
0.
4
-,
1-0 PV
0
"3
0
*0,
0,1*
i +'
'CD
*0
1- l
C
*- En4
-'I
*00O1t
o'
.
r- *I 1*
0"'
0
*
-0(OD
0,-'~
)
O*1
HI0 C
H'Z
Ct'-0 0
0
0
(*DC
(D
0
*014*
t<4 0'
0,
*
40-0D
+D
piPo-+
FF
'0
:-' (D
0
00
4
Y,
4
*
(D
**-
4t
4-
*4
.
0
00Z-
0
*
*('D-*
CD
*
t
n
4'
CD
*4<
*+
H3
-"1
0
0
(D
Zl
4
~
C000
Dt
*0CD
*0
*4-0 En- * CD01 :D
*
*
4
*Q"
40r t1
*'F-'C
W
0
0<
0
*CD
10-C
*~
*
00
0
r
40D0
(D0D
0t
'-D
-'
*
*0
0(C~
*D-D
*
*
*
'*
4 1
0
*r
4
04
*
0C
1
CD
00
v
*
1"
++
-+
'
+N
11
F-
"3
C
D
F''
00
DCl
'-1
'31
N0J10 P<
F0"-H
1'
0
0n
Int P.00
CD
FF ID WDF0,'
0
C
04'
04
04
04
C*Ft0*D
1CD-40*0
4r
D
1
Ct<
*0C
*0
4*
C
(D0OcrtCD
0
(Dtr*
Mtt
F
F *CDC
*D If'
C
"<
CF
-1<0
r*CD
<0 *
Ct
(D,~
00
10
*'
Di *
004
rt 0
CD
4*,
4
00*OD
Ct t*
0'
0
0000(D
"-
1
14
D0
0
(D0.
0~~
4 0
C
0
0,
0
-'
0
*
4
(D
<CA
(D,0
CD
" U00
,
0 4D
0
4CF
4-D
88
(D-0
't
1-
(DC
0
F'
0
-C
D
--
-'• * +
0
n
C0*''
D
(0
Ct.
CtDF)
ýo
m0
li
+.
.
i
*
C
0''
4o
F-M0t,**
0'
i0',.10 ,, C .',
0+
*
-
O110*4
uII
Ctt(
q-(D..
'
Ct
*
'
In' (CD *
In
() Oat
CD
ý
..
""3
4.r"
•0 C.
3
C) **'00
C+"
CD
....
0::1
*
t
'-
Mt
00
0
r*
0
4
z t-
0*
DC .D
OC
• ...*tC CC
0•
0
0
0
0
l1 I X<++M:
+•+
C F-l
Ct
1
-. ~ ~~~
+,,
<
4
"1
04 ..
"3
(Q
rC0'
T
*-<
*
*
Q-Q
r*
"
W
-
*
n,
(D- 0
CD 0
4CD
+.-+
*
Hn
(D
*
~
*.
CD
-.
00"
C04 ,,*
(D00 .
a 0CDF-0
4".
"
*
+
rr*04
++-4 * :
FlC
0C)
(D0'
0' F
0n
Di0'0t0o
(D
'0
t- C I-
nN
411*
(.D
uC
IF
C
R
0
*<
++C
(n 4
4
'*
(D+ D, +
I
+D
:0'
0 Z)* +
CD0r
'00
0 m
,0-F
rr
D
+
0 F-C.
n 4'*0
C
C
CD
1*.
D
*0)
*0'Q+
Cl*
C
*
*0
+
G)4
-' a0' x02
0
*04 0'O
*
*04
04
D4
'- *0-Fl
14
0 -C
•.
0
41 *
'+
. .,-.r
r
.'
m
0-. N,
EnI.E
<r
CA
+'-t
+ *1to Ent-.
1D-4~
I- W.4Il
'- Mt
PiD
tCD
**
(-b.,.-+
a
:ZI :c Q")t
011-CO
"•
o
t<C
:O ..i.
if.-.
In
.o
0"
''
1<
I_0. 0
4 0 0
I
Ct0'-.
0+
.
* "<
404
*
-'C
*D
'-'A
r*0
H-N 0E
C3 *000
.
-0
0)
4"
+ .+
40
i.
C
F---D+*
'-<4
(
~~~~~ t
(D
**
,.,.0
FO-'
-<1
0 +*0
0:
A404
Z4D(D0<1r
*
*~~~:
2-4 ,-,* Ct "'0 0
4
H+Ct
* 4D'
'Mt
+(D
(n L<• *
+
*
+I +
4r
0
O1
:I t
CDD
0*
..
+0
*
:
*
4
*
*
Ctt
0
00)
0
04*
M00
-c
w
0 00* +
""A.
+'+0 *j
**0'
(.
(D *ý
•(D '-<o
+ C1+ +00
WIC'0)'~
ZI I0
0 r
(D')'C
*
.
+o ++"t
++
C D
Mt(
44-4~(
4* :3'
r.-
Cj,+C 4
01- 1" +
ý3
*+
0 *
0
+
0 caC
4l
rttt
*
CD W- 0
0
'0r
*CDT
CJO-
*
*F-*~r
Ho
-
+i
(D
w<
*
0
P.
+ZE
Z"
I0 "
Ct'-o
+ MF: I , ;,-O , *
D
t'*O
)
0rr
-
~0
4
*
4
CD D t3
*0
*4
0
*D*
0CC(0
11
0
0'-<
, :,
Ct
-
*
l
.
lb *
CDCD*.
-Ct+
0
-'
"
(D
CC"'0
1-0
vC D. 0 I CC m
D'
v C)+-,
-",0
0 z~
IV
+•
ti
P 0j
F
(D
z0
H
< 0l
00
0CnZ~D(D
"<04'D
D to
0 '-O'000't(D(
.0 Or
0
CD-'
0
0 t ::1 C
'00
0
F0 *,x*'",
(DO 0
F00
* t tt
Di' H 0
0 Oli
Ct'100
O 4
C :
Ct
0
<
*1
CD
rt *+
0,<,+
(DC~
(D (D0P1 +
0
H0
DC
0
0'
<
0
0Ct"*Cr',+
0
-'
*ý
r?
0
0
0
CDn
00,F
R
F
4:Z,1C
H '
QCD
M
CDI
4004n
(D
40
,I'
-'DC
0
1CD D
-C
0ý
0001<0zr
R
0
M' 0 *
W
o3 ()t+
Ct
P.
000*
::
H
51
*00 0
Ct
0-"0
(D
0 U 0
4,
Q)0
C
* F11-D
4
D
<
4'
*0CD
+
w
DC
04
0D
0
*0
*0.0
FM l
**3
0
0 *3
:: *t
*.
Ct00
. C4tL
1<Ctl
r' 0D
t *ý
-'H-I
0*0 *' (D '0*
*3
~
(D
(n
CD
*00
(D
*000
(0D
0
*11
C
*
F--)t
*
4
CD
'0
H
C
_
N N N
-~
0
p1
0
0
01
0-.
0
S
N N N
N
4
*
*
*
4
*
1-i. *
4
*
014
*0-H'
4(011
+IS
S00(
0 0
11
PSI +PV
S
0
00
0 0+
+- 02
+
0<
x00
0i --
11(0
in0.-.
14
(001
00.4
00
020
*
0
01
'11
0 (0
4020
* -- 02 U..
(00
4
4
11
H.
rv
z1
*
*
(Drrt
0100
0.10(0
N
*
0-0
(004
*
U
'014
0-
1 4
*
11014
Or-i. 4
coo
4
4
*
*
*
4
4
4
4
4
4
4
4
4
4
4
4
4
H0.
1111
010
*
4
4
*
4
4
*
4
*
.10
10
H- H .h.0
F.
00
In(D
0
-
11
r't
-0
10
*
002
001T
0
(D 0l 1l-i
i
if
lo0,0
co1-1
0.4
I<
N~
(0.
110
-
*1
0
0.
1(D
14H.1(000
0
0.
*l
0-1
-A
U.1-. (0
0 1-.
u 0n1
1-i
In(f141n
I-h 10 NU.
4*
0U..4-.
0 D
I
41
4 I
*1.
U.0
U-a1 H2Pk(
k-ti
(000.(0
Q1 01<01011
I
010
(0k-.
(0
4
(0k
D(001
(1
4
r-. +
Fl
1-. --
-Q 01
00
(0D4(0
141-i.
0--.
(0
01-c,
0
EQHU.
M011--U.
Qj(
-(0
11
1401
100
(0
+-1-U.
00
:I
(- 0
01
1--
(D~0
(D
10
U--.0
0
10c-i.
-1012
j (00
1-i.'
t-,
(D t34 I'D
-.;N-U
0 U.o
0.11U."
0r 10'
.
0
..
U.1-,
Z (1
k-C4
U-.-.14
1i-.
0-0
(0
0
0.
14
010
14(0
(001
11~c
0
(D
0l
0-112
0110
0 0ID n
011
0
(00.
(0
n*H
0 U--.
0
(D-1i
(n
0 01
rr-
;,
a
U*0
W111
--.
<1 U.
0
40.1
0
to
0
D
.
0214
U.
H-
1-1-14
I
rr11
00
10(00
-01
-0
0.
0
(0
(0
U-.. 0
00
1-1-10
(D0I
01c I
01.4
'-"(0
.4
S.g
W -h0 1011
4<0
* ..
rrI
0
k--
4
1014144
10 -h
A-021
E0 --
I
Jo
0.0n
ri.
0(0
H- 0
*0.11
(0
-..
I
k--U-H-I(ll(
I.
0(DH
En1
0114
004
04
11
01(0
(D- 00-4
0.
U..10
M-1-if(D
401--~
*r1-4(0
0U..(0
- :3 (0
m0(101
w1i-
4400
(D
1
-
4
*
4
4
*
4
4
4
(04
HO
U.
*
4
4*011
440
4
4
11024
02144
0-.
HH
11
*
01.44
0-.
(0U--.*
4
~-0
0-.
01 U.11 c-i.
(04
(04
U---
14*
(04
(04
Z3 (D (-.
(D
0t :Z :Z
U..H
a m-0(D11 -,
Ok.1U-
o
0*
Q00
(T)
0
j
1-
(01-1-D.0<
a4
0 -0(((
(04
0(0
(0
**-0
* t)
--
0)
0*
Q-1(1
1
0 U..
004
U..
;110
U--.c-~
(0 U..
4*021
01014
4 a4 1i 1-1-k-.
*
.. 0 U..
(*
(Dk-111 1)
(D
100-4
0(0
*
4H
4~'4
(01--)
0?
4
(01-1-4
40-0ý
U- w- b.i
(bc-i.
U.-.0-
Oco
(D
14
-,-a
.
~0--. ~C1
*
1-1-Ui.1c
4
0 Z)f-.b
((a0)0010
1-1- *
-404 *
0-0 *
ITNNN10144
01
i 1
U.
104
11
41-4<
*11 I-'
021H0
101
w1 0
00
I-
*-0.
1-1- * --'I
U..*
0
0 4'.0
((
U..
4(0
40
0
40
U.-. 4
k01 *11
1< * *01
Ur~
0I.
+ 02
- U- 4
11
4
01
0.410
(0 *1<
cu.
11 (0 0
M1
0100
U.10 11--
0. II
41001
010
NNN
U.
H-
4(01-cc
(04
4U..r
0
zk-
-
14 *1<0.
U..
k-.
U..
0
10112
U-I-
I
(
(D0
0-010-
01-111 U..
0.0
(D-0
-0.
4
P11*
U) 0--i
r-c
0-
*
*
0-410.
m
(00
-. 0 '4
11(D-00
T
4
0--11
(D21
.4
0
0-11
f-. 11
14(0
0
00
N
'4
10
4<0
(0(0
-. 00
(D
In-(D
tal
4-
0
(0
0(0
0
U.n
(D1i.
(D
1-r- "0
f'4(10
14U.01
t
ci0 a
X, tN
41.
(D(002I
10
0
0-H
.(
4
*
4
1- :1,1 41
0o..
!
00
0.
1iii
144
NN
0-0
100
(0
4
-
w N
.
000r :1
-
01
2).0 r
N N N N
N N N N
,-
1
10-.
10'4(
020(0*
N
v
141-1-
4
141-i.*
-
x0
4
*
4
(0114
11
(004
0H4
(0
4
4
4
4
4
4
P.•
t-1-
U.. 1-i.'
*1-i.
*
* U..
Cr0
H
o.0-(D
40
tn
400
(D
.44
*
4
4
4
4
4
*
*
4
0b014 X.
1
H.
f10 (D
0r
:3-i1001
4.4 1-i.0
4(0001
*U-l 0-k-.
40.
U.-.
00
0.o
1-1-U.. *
U.. *
00
14
bH
.
0
r014-1
U..f0
*
*
*
*
1011
4
(0
X
02
02
*
*
*
*
*
*
4
*
*
l'< 140-
0W
410
4(01411
x >1
002n
*
*
*
n'4
N
40-02
0.-
141.1
11(0
"I
N
"
41-i.
*
*
* U---.
01 *
4(0
U--. *
~01 U-'-1-( *
*r-i-00
*
* U.. 1-i. U.-. *
*0(001
*
*0111-i.
*
*
U-,,10 *
*1001
*
40011*
* U.. (0 0*
*014(0
*
*
*
0
"I
*14~H
*0-10 0~
401
-10
4 c-i. U.. 14
4
U--.10
(D (D If
*010
*0140
S+
S
04 001 5z
I.
14l
P-i
H1410
*
*1i.
-
N L< 14
0 0
*
A-
N
,0
(01<
*
*
*
0101-.
0204
140.4
U..
(01-t*
0 H-
*
0.10
4
(004
0.4
104
U.-. ~
01 *
14
1-1-10
011
01
NN
NN
-
N N N N N N N N
N N N
N
NN
"
'-N
).. En10 0
I."
an
a -m 00I1
C
, Y'
aa0
P2
- 42 P- 0 1-h CT ri
ma
mr
-42
*
a -
"--
E in E
-C
0 1
*&
*0d
a)411
0
I
0--
:faOL
0
0
- *++-g
*3
-*
0;a
S0DI
*
*
..
0
*
*
*
PVt
ID
p1
.4244
0
1En
N~a
42H
1-4
0
4
1<
C
P2 CT0
<ar
El
442
(D
r
(a
42
-
a
42
0 rt
oD
r4-
-
''
422Il
p
(D*
4wo
tr rr0
4..
4:r
0~
42
(D
421
rt
XX
4
(ID
(D
dp(D
-
a;440
444
ma,
*
*
a-a-
mm
m
m
Ct0
2
m
a
-4 -4-
I
0
m
6mm
0
-ia
N 4
4
4*
m
'
(D X
Xrt
D00
0t~
-0
W
(D
?0-do
*+ (D
m
•.
mo
" n
f-
042i
0
r'
:j
0C
rt10 -
42
*
r'r
a --
ýu
-i
0-
0.
am
*
-a
*
(D
(E<0"
421
r 0)
*4-t
a
L
42
.I-.tI
*
*
mn
a
42
L
0- n
~44
"14 .
(D a
m
0-0-
al< mal<. L<
ma
~ .am
0.4
0-a
W
Cr11
44 Ct
l P.
"I
LhI a
0-
i-i- I-~ a
40 -0
*4-.
44
*o~11 a
*
0
rrt
afR
0
0
I--
-aH
N NO
M 04
m 0
*m
*0
*0.
*..,
N
ml
a
0
.o a
IILO
*00
*F--.La
44
*0.110
*
*
-
N
4-. *~
*
*
*
Cr
*
0.m
CtLL-
N
-04-)
*1< 4~,0
*0
a
* I-. i-i- 44
*
*42
Cr
0
0-H
4241
N N N
iox
*
F1.
4
*
0t
Ht
M 0
-4
*
11
o
.*
Ct
0
0
Ct
t
-42
I-I
In
"i
0"<
-
S
0
•1lH
H- H-
0
Ct0
T
t
La•a
"
00
Ia $
-rr rt
0 0
0- rH-"+
4*I
(
H
+
II~
Mi
*
x
(Dl 0
"
11"(D42
H-0
0
10
*4.
0
0 (D *.
* (
.
-r
N
*42D--D
(D.
00-•
( M rT
H-
-3
4-.4-.42
0 0
0
+• +
g
*0
44
0 (D
. •I-'--442
rl
a
0
ý- +
g
40 .
*. 4-
Ow
amr
- +.
OMC ACt
4-
+H
*C
*4
-- 4444
2-4
*
0
nI-
L
rr
rt
10H
-
(D
1
f0
Ct-
rr
042M
Ct
00
"} :3
r-'t 00n
r-
000
N)
I-,.
0
I'0*;-
mar"
*04
0 1.. -'-
4
44244
r-r
0
4ZI
4
I *1
"
b
*00
H-
*04
42
02.
v- I0
II
*j0 a--
1)4*4
44.0
0
4-i44
0t
4
444 20
4444.a
0
H.4
+--0
0W 1 +
-- 2-P-
*
-
C
1 1q
(4o02*
0
*421
.*0.
*-
a
(-D
440
0 aI
o t
-Ct f.
0*)
n0-
O It
*42LI
a44
+*+(CD34- +'+
*
F--•
r+-h<-rst+, H
rr
0.
4
-(Dtl*
*44
0
42--
40044
rr *4442
rt
101
4+
-42
*
ID
IID
024
La
4
444-44
*42--*42---
42
-0C
a42o
*0
*04
424
44'0
*42042-t
*0420
0
4
-1 t0424
- -l-
+-42
*D
*4
""0
*Z 0
@o
a-
4
- -34~
04
N
0- 0
*
40R,
040
42
(0
< tt
-
(D (D
0 4.
a'ao
N
NN
u
'-1*
CD
~
*
"
HNz
CCr
04
CD*
o
CD
I
o- -
.
'l
"
*
Di
*
0
*
(D N
C*D O
CDI-C
1×0 -0 •04
00
K'
'0-m.-
to
rrt
A 41
QI Qi
-.
+ rrrr
7144.'+
0P.D+
0 0 0
0 N.C)0
r*
t
r?4
0(D
P)V 0
0
?
(D
0 0
o
CD
? 0'
to o)
D
-
D0
0D(D
4r:
0 0N
:
CCC
C•4
*
*
o
r-
Z~
ln
0
*
Di
m
*
•4
CC) 4
0
*
4
-r- +
oYmOCW
oC
p rýjn
0 z'
0l
C? (D
CD
CD
ýj-
•
+
RI
*0 "
0
rrý
l0
++
nC?-+
'+
0
C?'
<
CD
CN0
ý1+
rr• +
+00+
+N*C
0+0
+
(
+
' n4,
4I 4, CC
4, 4,C
,4
0x
0
'
0-
0
Di0C) N.C
•C)
4
r I
?N
DIOC(?C??(??
N'' '0
D +CD
+
00
0r
0+
+C::
CCC
CD
(D(
44
(D
0
I-
04
r O
*0
I4
0'OC
0C?:
DCO
*-
1
O?
rCC
*
C?'
r,•
0
0 00
H
C?
Cl
m
0(r
C::
.
-3
(
.
(U
D
(DN
wX (.
rr
0
NCDC
C-.=
00
N'C
C?
Zý (
CD'
CD
0
(D:~j-
CD
Di
(D N
DlN -C
0
CDC(D
0 0
0 0
C).D
44D
w4C+ (nti+
0rC
0 +44
Fý
+
0'
00 +*
r +
CI
+:0C+ (D
4'-N 0'
0-'CD
04
N.
r rt
-
CD CD-CD II
CD
. rD
rt :
rr z1*:Di0
" ++ (D P. + 1I I C
E
-n. ol
• C? C
+C:
+
*C
C• :"
C
..0
+
lC?
En m
C?'
D P-+C
DH-+
F-0
0
+ (D0l+
CN+
CD "
+
C)
CD w00,
'0
0'
o
+ H-(D+
'*0
CD
0
?D 0D
+ 00
+ II? C?,
II 0 C?
11
a IICI
N000''N
N
0'
C+
-Z
+
l-N' 0'
N' (DU
00
'<C
D
CDm-+
En +
1-40 -'4
(D 0+CD
+
N4N
0 C -0
CD:Dj
+ q C: +
00
04
04
0
r-
(-D
0
r
CCC*
CD
P.
C?
tr-L<
-
"0
CD
0:
r
1C--
0
00
C?
0'(
0
-X0
+
'0
+
::3D+ C?0'
CD i+
:CD5
C•4
0
0
CDCDt
(D0
04
o
C?
0-
•
CD P-NI-N
F
0
1 000
?C +:
0 C 0
?0
4rO
-
S0
X44i4""4
0 00
*
5
"t 5l
<t
N<
0r+-+
C:
+-'--
0
N N
0+
I
NNN-'
NN
N
VV
0)
Di
0000
N---A
*
CD
rr
'
0•
'-1*
N.
00-
*
04
*D
Srr
C:
0*
04
-
4
En
rr
CD
D 0l
00'lIJ
0
0
CD
0 0'
0 (D
0
1 Vi
'' :':
,=(D
(D
00
'1C00
0O
CD'(D
CD
OCD
(D
C
40)4
04
CD
*
C:
*
*04
*CO*
*
04
0*
CCC *
404
40)
I-
CD
XC1
4'
0 0
iO
(DV:J D
*044
4
CC)~
H-7
CT
-
0
N-
0
t0
44
V
• 44
VO.
0
CD
CD
P
C
(
40'
*CD
0'D
C
CD
I
IN'(
(D
o...
D'J0
r
0)0C?
I
. 'CDC
4•
~ N.
*Di
NN0
D
C
*Di
"*C?'
4•
*CC)
w :E4C?4'
:N-0 *C?
n.CD
(CD
CD'
N
-3.
*
CDD
D L<
(-
C:0'C
44-CD C?0(DC1
W
In W0
t
VO)
C??
_a
(D
N'0 kV)
040- Q
(D
:
N' 0
ýH-"'4
DiOO
. m (
::
-i
F-D
C 0
+0+(
0 W- PCI!
,'-
-,
-
cn 1-
I
ol
o0CDrN-.
. (D
LoV0
(D4 0 10D
OC
4•
0
0
0 J
CDDN
0
*
~~44
r
40
-
0
0
44-.
N
N
*
N.
CD
N'--C
4Di
40
4CD
*0)
40
* N.
*0
40'
CDDD 40
CDC
CD
0
N
CD
N
40
*0
*CD
CD
(D C
CD:
*0' 0C:
N
DN
0
QD'
4 1-.
0O
'-004,:
N'
*CD
D
•
CD W
*
0
*0
*0
,Di
40
*0'
CC) *
N
0
N' rr
r 44 0
2) N'0
C?
PC (D0:
4-.44
0*
04
CD
0,
*Di
40
40
*Di
C?(D
*
0-4
'•4
D
*
*
0*
04
C:
P.
rr
t3
CD'(DC
(DDIC
ON'
(D
N0
01
Ot
0
04
04
-
~
m
-.
C0
444
Di
1-0 rT
(D
44
N
CD
N
N
NNNp.
*
*
Mi
*
0
0 0
II II I
IIi
*
*
*
*
*1-ti
*0
*0~
*CD
*0
*0
C?0D
Xo M.
F*CDH
W1
I-.C
*D
CDOOC0
tA
00I(D
10
~(D 0~(DCD0
C
x
1
00
P-
-CD
1MrA
C
1010(D
0*
CD-
O
Z'1 0
-
(D -(D
(D
r-
(.?D
(D
C
C?
*0
*10
*1*
0010
*
r00
CD 0
*00
*0
*'
* CC
*
* H
*~
*0
*0
*
*0*0
*
*
(DI'
4- .
0I0
-o
*
*
*CD
*
*
*0
*
*0
*
P.
C?
-.
CX
:3t
0 0D CD
*01-4.
o0
*3<
*
*CD
*
*10
*
*
*
*
*
P.
*0
0
N*
'00
F
0
+
W00
*0
*0
*
*0
*0
*
*
*
*
*
*
*
*
*
0
CD
j .0
*
Srt~
*< .
*
10IC
r :3~CC
P
t-I
(D
*0
-I
In?'
CDl
rt00
D
0
a 00<D
ODI'0
Pi0 (D
(D.
*01-,0C
0
*C
+
1
x0 -CT3
P-C?
(
(D +(.D
+
+ +
(D
0+
0.D1--' rt
CDDq
P.
*D0L
(<D
0 CL
+
*CD
*
0-.
In
H0
:
I
* (D * (D V
tD
tl
(n (
*
0
< X *NN
< P
* 1*
*-
*A
*0
ID 011 (D
CD
(D
(n
*
*00*
*
+D
CD H
CL1
0
0DC
0.
P..
*CD
*0
*CO
*0
*'-
N
N N
N N N N NO
N N
0110
p.
H10
001010 IQ
w
w+
p.(D
.0 w
0. I I 0En
01
00
1`C1 00
( DI
A•
0
0t
S(D
+
-
<
+
In
10
(D-
~
cn0
00
rr
IC (D (D (D (DC )
InO
0 0 u
1100(
11 0
:<
10
0
:
0Q( 0•
t-
100I
*+
*-
000
pi
(DIl
rr+1
010"
CC (
ec"
co*
D(D
0
(D
a-
<+0.
CD
101
(D000C
-- 01000-
(D0
<0
-CD
?
*C?0
*000
*00
H
*1-.
* CD
0-
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
*
-0
V
0
0
0
1-.
*000
*CO CD
*000
*-'CD
*
'<CD
*
CD
*1'
CD
00-
00 00-t0 "?1W
0
LoC (D IT)C
i1 L1010I
0DQ
H
*10 0
0 ID t3
M
.D p.
0-
)00
0.0.000
0-5
0 1
(DOCCO
(D
00
(D0(D110
0
*00(
010 Z-. C?0 DO
0
0.P-
510-C0
* 0P.
)
*00
1110D
1
(D
-0
"
ECn
i
*
DCO 0
(01-
C
<10
A
z
100
0
+?
In
*
CD C
*
*0
*0--
10
FClD
(D0
*0
HO
U00
*CD
0' i
(D (D
*0-D
10CD
0<
CD
(D,
*CD (D
If
P..
*00
v
E<
IN)
(D
CD
0.
NN
*CC rCP
00P
CD
CD3<
P.
0C-..M
*
1<
COP00
C?0
0
In D
(D
3<
00
00
-CD
0
4In
IXX In ( 10
10Z D - L1
HOD
-
CDCD
*
*-h0
0
00'<CD
CP.
0l 10 0
0 ý
101D1(
P.O
00
*1C?
Fl
*0
*0
0
10
-30
*
*
N
*
D P.
En00*n C?
*
-- 0-
CCD
P-CD
00
CD
'-'CD
Nq NNND
M
0
0'
tol
*
0
010*1
0
0
*D0
+ 1
0
-
(.n
r't
CflCC0
1
+
00
rtO 0
Vt 0
•'
E
0
PCC.
(D
0-•
0
P
00
U
0 CDO
--
0
(CD
EnD
4T0
D
C:3,
C-0
1-
0
00
DCC
+ +00
0f 10
0 ( 0-
(D Cn co
z* -
0 0
w1
(D
CD-a
*0 a
1
CO
mm0~
000-0
-tO80 10 8~
9 0
H0-
+ *
Di0 3I
-
CO
D
0
0'
0
0
+
< O
(D
0 A
C
1~0C
Ct
5
(0D~C
0
0
H'0 0-o P
0
c-0 0 0'
0O
-CD 0
*-C-l
<DC<
0J-1'DC z ' "
gP- EA
g.gX 90
w
11
rt0
CO
<
00
00
w
(D
plr
Ct
D(C
0
0(D l
fl 0
COO
(
CD
0
01t
R9 9
(D y
W 07
C
(
Ill
(D (DC
<0
zt
+
0
0+
ýO0
(DCDlt0
(DOPORD
D-
(DC
H
Ct
D(0ZOC
'0-0
DH HC
Q0'
(CD+
OH
-
D)
H H'
A ZD
CD A
(D-
-0
Ct
(DO
C-X-
CCCC
0* 0
(D rr
+~
CtD
0D pD
0
0-"I
*0
0C
ý-<
0 0 "t
CD
I
0CO-CDCCO D
00X
*(
100I
4
0(D
0 *
D
-)
<
CD
C
-
-1C0
MCI
0)
(DC(DC
(D0
(D0~
(DC-
tj
t
I
-0
'00
(D0
<0
CD D0
-
CD,
N
Q
(D-
lzý
Ei
Q
H'
Mt
CD CD CD H
rtrCE0- EnDC
-t
M
(D000 b'(t 0D
-.
DCj
H*
rCD (tDC
001-.C 000
CD
00
-
CD
Sb
m-OCDD (0D
Mt
MtC
0 0 0 H-.
r- H- H- 0C
(D
(D~C- 0(D
0
DO
0) 1-,. in
tn
(D CDOr
FlC
D k,
Di
Mt-
a)
DH
(D
*
(D
C-
-C
a
CtI Ct
-DH -
P1Cin(
CD
CC C0
0
MtI
HCD"
fHo0EnC
CC
(D 11CC
En
Fl (nCHDC-
H
Ct rftCD
*
1--.
CDC
*
I C-t
M
0
0 w -
<O1-tOý
t0F
t0
CD M 0 M0
0
0týOH-
0
0
IIt
00
H
(D
Ct
b-HCC
CD
Ilm0 0
0r
MD
(D•
txj
00
:Z
f-0
f t
cr
CD
(D t-%
(D
CD
(D
0
H
r-
-3
th (D
O
CD
0-
CDC
OD
OD
HOC
O
1
HO
00A
fHH
(D X MtI H
5<0<
D H- PCqP
D H <DD<W(CD Cý
(D
CD(DHC
(ODOCDOCDOCD
Ct
0
0
"OH
"=
L" v P. W 0) I<h<
< CCt<<
0 CD 0CC
CD
CC
0
CDt
0
00
CD
w'
fm
0 ti 0 C
(D
t0r
00
Hý
" 0
T0
0
(D0
00rtE
Mt
CON CD C-C<O 0InH
0(D - CD
<D N3
C
o
0
0-0 CD-'0CtC
C
1-1
0D
0(D
0
0
(D
43N<'X
D1
0,C
0-
(n~
OC
ý
Ct t'0
(D
0
En~CD
CO
Mt0
Mt
ZH-
(D
tD
t-0
SCD010
"
0
flaHO
H
Ct
X
D
0--Cm0
(DC- 00D
wl,
- 0
1---
-OOC
OQ,
1-0 Ll
0 0z; rr
(bCD'
0,
(
1CD-H---C
(DO
0 m
I-,
0t
CC
0
-3t
Clrtm
D -,
00 0
*
*:
(D
C
MCO
<-
D
Ct
-
Ct
C
0
H
0
CD N
M
(r-0
"
0D
0-
0
(D
*Dlb
(DC
CDn
CD
Mt.C
0
HH0-C
t0
D
C- (D 0
-
D
0HQC
0
- 91
0
-D
Mt(D
(-DI 0 i
00-C
(D0
CO0
CD
0
CD
'
N'-.
,.,"
H
CD
WC
0.D
N r? oN ) tr
CD
<
C
"C+
' '0
'-I
F-4
CD
C-.
C-
-
'0
N0
t
(DCr
CCD
rC H-
-
0
DC-
C
C-)
(D D
*0C-CrD C
C-,
-,
(D,
ka
Crt
'C ,' 1
4-
CC
'0
0
4' Z1
9
-
fDCD
4CC)
*DC40
0(0-
(CD
0DDC cn CC
C-C r-
r'CD
4 C-.
40.
*04CD
C
44)
0
*CD
'<
CC
CD
rtD 0.
Hr-r
*D
C,-. *
dpD
CD4
DC
-lC'
.
*D
D g-
C
a 00
(-DD
(D (1) CD
r
l-
DC-*
0.C (DCI,0.
Cr'
'0'
oCoor
4
4
C )'.-
?
C-C
NCC
44.
00D~
DCD0
C-C)
C-) CtiDC
*H-
C.*
C--.
C--.
-CD
*DC-
*DCC-.
C--.
DC-,C
(DD
:<4
DC
X-
00.
C-..
4DC
*DC
4DC
DC
*
*CD
4
(DCC
C-.
4
v
CCrqt•
.D
*
*
4
4CD
(D Z3.C
:3
D i--.
t-h
It
C-.D 0-(
Cr
(DD
ON*
P.II
II
,rCD
(D
r CD
'D Cr-.''
Cr
D-+
(
l<
4C
4)
'0C
CD
lCrODD
*
EnD
OI,-C
*CDD
OC
* '<
n
::s*Ct~
DCCD-*0(D
(0DC
(b0 :3-3rtDC+
* C- CDC
D
0)CD N-0
Y
D (CD
0
11r
*0
*
If
DC
*,t
C-C
• C.
-0
-
H
dC
I-
DC-. )-.
C-
C -C
(DCDD
40-D
00'-
Cr
,+*
(D(rr
00N
tr~r
c,
~II~~
N
N
N
N
'0
DC
II
DC
C-I,
0.
DC
CD
<0(
CD
0.
CD
C-C
<
CD
H.
I-.
CD
C-C'
Crt
CD
CC
'P
0.
CD
-C)
DC
4*
CDD
DC'ChC
CD
H
1-CD
NNNNN
H
CD
CD
0
<
(D
Cr-
(.D
(D
'D0
C-
0
H
CDr
DC
CD
0
m
(DC
<0
CD
x
D
l-H
-1"h
(D
CDC
CD -'•C CD
D
Cf
D))1
CD
CD-C•
ft
- Cr
CD-
C-C
(*
CC
0
(CD
H
* (C
N.
C-- D)
(C
D Cr
1--'
C-
CDC,
'0
-C
N<
-r
*0•
N<
Cr
Cr
C
t--'
:1cr
DC
0
CD(r
D
(D
'C
I-
CD
0CD
SD
<D '
CD O
C-l
0 CrT
DC
''
00
0
CrX
CN
DC
<0
0.
D
''
0
r
DCC
'+r
(DC
C-'D
<
H" N
H-C
H-D
--+
N
<0
4N
-
N+
Cr
CD
DCrr
Cr0
--
CrD
CDr
CC
-rr
-3y
0
0.
CC
P- H
C-..
0-0
(D rt
DC
ICD
D 000-
D CC•CD
H
H
Cr
(D
(DC
-CD
CD
0
CD
(D
<
H
(D F~H
-l Cr
r
<C I.+ 4
11
CDH
ul
(DCC
<-
CCC
Crt
-
CD (D
N
CD
*
t
CD-0
CD
D
(~i CD C
(D 0
DC
H
z --
<•
I--, <-2-
1×
CD
C'- '0
,
<
CD
(D
C
<
(D
DCD
0
t0' 0
0.D
(~DCD CD
*
-.
CC HO
NCD CrT *
(DCCCD
Cr
(DC
DC
DC
CD
'0
0.
CD
'C)
0
D
Cr
C-I,
(
.D
0
II11
(D
CCC
(1) ..
N
H-
CD
--
0.
-Crr
(D
C
-D
CD
DCD
4 0- r+ {b,0CHrtk,
*4'0
.D
*"'C) CD
Cr
I
-
C1. C-' r.
N
I_ 1t.
10-'
*NN
0N
N
--
t
*~
01.
C. C--
*
r0r
-.o
ll
*IrNI(C
*
*O(•
*
D n--
0
INN
C--
(DC~C
o
*CY
,-
0
0~t1
N*0
0
*N at
:NN
*
-*0C(DN
H.
lD
~
C
~
N
CL-N
C
*NC-l.'0
*
Di
*-.
*Ofl-r
()-(-0
0 l
NJ
-0
t-4
*0TX
Zit mH0-tOr
*0
*
(D
*O N
N
(C
N3 ONNl
r_-
C-N.(D
0
e.
.
0
'N
NQ
C-C N
(CD
00"
*
NOn
0
0
ND Fl
*
tiN
0.
C-.
a
lCD
'00
ft
W
C-,
N
I'tO
0- 0-0
ND
NC--
0'm
m D0fC
t3-.n
C-
Nt
1-00
00.
-t
CC-.
10
0.
I
a
EnCo
(*
'0•
frt
N
*
:1(
rN (D
N
NO
-
.. rt
0tN 0-0
*C--.ON0HC-U
g- 9*
O- -0
'0000
C
0r 0lt11
1-.0
N,
*b
~~(
*NNN
N
N
I f
(D*
*%0
Nl
H
*00
-.--
N
.0
EO*
00
If
ON
(N-D
0
0D•E
'
NrC00-00-,a '
11nNvONý1
OfT
00(D(
0.NON 0
C-C-.
C(n
ft
ft.
~
(0
0
0
ýj (
~
C
fN-O-*0-
- -
N
-. --
0
~ ~fC
N- L IQ
0~
*NNC.N
NNIn
CT
N
0 *'00.0
r
a rCtNO
*C-0-f-~
N
0
0
O
0
N
I-.N.
0
II
ND'd0'0'0
N.
(D
N
*• .
f
:NC-10
(q-.f
*
N
0
(D
!14
C-H-H-H
ND
""ý3
ND 0.N0)
HCO
0
L
•'r
D
(D C0000
(DN
-~
rtN
'N
(
N
*N
*0
*
*
*ON
*0
*N
*
100
0H.0
crrI
C-.
0
N
C-fx
N'
,0-t
N0 N.H
N..
In
N
N0.-(DC
x-
H-ND
(DC-
0D00
0Fl-930
0
NO n I
<
NN
ncl N
<1'- '.
NNN
ito
-CC
C
N
N
F-LQ
H-
14
(DrT
*11
0.
rt 0'4
C-. C-
C-
ft
t
(D
C-C
('0
0I
C--dp
lt
0
x ."
N
HO .
NOD
N"
NNN
t
N
0 C0C0
-+
<NC
(D
En
H
*0-
.-
fC-OH-
C-
0C0)-(
*0-
r•
Nl::
+
CNN-
N•
(D
(D
ND
-- +
,a
0j
OCr-
x
* n
Ill
N
(D
H
H
N
Of
<j m
0
flN
C-'-.
Nr
C-
NHCC
t-.
+
<Nrrrl
n-
C--
t
r- 0C
C--tp0
-
N
CO
rr
NO
-
tNiN
-- '
0
0
(D(
N N N
NN
4
4.34
*
ul 0
(31--..
0*
0*
04
~
l
(U
~Q
04*
0
*
(U 4
44*
U)
(U
1 -
I
(D *0
0 tS
0bl0
:3 o0
a~b
001.
lb:3t *
4
U
me
4-.. *
0*
??O *
0'?? *'0
(U 0' *0
(U 4(U
*
(
4, w
4-.. 4
U) 4
C)
0
R
*C?000En
0
)
t-tn(~
pC
)
U)
0
(-'0
004
-'0
0
4?
4
) (1*(En
D
43' (U (U
D
040
4(
0
(
U
iU
'al
(U U (Ql. U
'0*n
40
(Ur
: (D0 (
(D.
t'3
(3(4)r
Oa 4
I
4
4
~
0
lbC
--41
T
*0'??
ul'
.'
1.4
3,
4-.
*
40
411
*01 w II
I-h <
*. <II
lb-lbm
4
*
*
*
C? C? 4
lblb
4
4-4.(4' 4
0(U
0*
04
lb
0'0
(UU)
4
004
(-~*0
*
4-44 4
*
*
(--4'
4
4
1--. 4
0lb
lbO
04
04
lb
40
*0
lb
0U)
(U
04
4
ml
C?0'
r(G
44)4
4-.. 4
U)
N
(U 40.
044 *(U
0?? 4<
0.
40
4 *0
40
U:U)
(U?? 40
(U 4-.
0U)
*
044'4
U)
4
4
4... 4
U)
m
(U
*
0 4-~ 4 0
Obs *0
lb
*
4-.. ~
0*
(C)
1-.
0
•4
(U
0*
*
a
*
lb
f-.
(-44
N
4
N
*
U)4
lb
(U4
*
U)4
444
N N N
NN
N
*
N
N N N N N
4
N
0*
NI
N,N- N
"1NN
4m
0
0
CU
CU C
I
<
D)
U)o
(DH
+U1--0(I
+
< 0
0
,5 ,,
H
X
r'-'
U)
I
(I
-
0
II
+
+
*0
*lb
*0
+
n
0s
CU CU0
0 0 000 0r
CO0
ýl 0
:J 0ý
A
A
+ +
C?(U
0-4
N
4-44(U 4
*
*
4
*
*
4
4(U
4-444
4(4
4
1-. *
44-44
4~44
*0
4-'*
41-1
4
*
U)0
*(U(UU) 4
4
4lb0
4OlbC? *
0.--
*
(-'(U
44-44
4(-1C?•
+
*lb
*0
:j r
rrtr
r'T+
+000U
+
4-
04
4
4
4
*
*OlbU:
*
4 C? 1-. 1-'. 4
*(U
0 C? 4
*0440' 4
*(U
4
*0.C?•
4
40-lb *
4
(Ul-.
~
4
04
4
(U 4
4
U) 4
bi
r'.
N
N N N N
lU(D)
D 00
1--0'0(U
w-Cr(U004o -'
1-j.
C? 0
00 II
lbw
'
0'4-.0'0 )
In'03r.
C(D00
x a)
t0h
1
.C(D
rt-
0.
C?
(D0
'
01'0(U(U
000P.
0
(
D (0
U)U
U
1--.4(
Ol(Cl
M(D(U
0U:U)
00
(U I
0b
(UW
(U
M-,?
lbO'C
(D
lb'
HO H
(,
HY)
0
j
H 0
r'
0y(
04
1-. 4
*0'•
C? 4
4 (U (-..4.~ ~
4C?0.
4
4
4lb
4
4
0'•
4(UC?
(D
+
lb
*(UC?0
4
4 C? *
*U:
*lbOO
*4403'
1-.
0'(U
0
<)
(-.4
*
4
4
4
*
4
00-00'r
rU)00
0
x<C?-
*
4
*
*0'4--.0'
NJ34
4
lb4?
0-U:
4
*
4
4
*(U0 44
lb(U
41-0
40(UU:
(U
40.CU***0
U)
(U
(U '4
*
lbU)
44-440
0
U)
CC0
40.00
*0
•0."
I
4-'U)
*4-4.0 (U
4•
4(U
C?
o
>1
0
'0 0
v
+
+
(I
?tl.4
-(D
a1I...
10
0
C1-U
1--
D'
0
0D
O0
OON
0
0
lb (U
00UbD3
U
U)0lf
(-00
'0?
C0l
1-lb0r
U
C
0.
· - N"IN
- N, N, N
"INN N N N
N
0
04
44
44
g
0
CD
W
CD (D CD 41
(D
rt 0
0
CO
(D
0.0
c40
*n4' 41'C
Di
(D
0.
C
I'-
0 4CD:
CDCD
*
4
Di
CDCD
4
00
(D
0'? CO4(D ul
0
fI-.
CD 41 1
N
NnHCa
H.'
CCD
0.4-4
~
CC?
00
CCD~
HHH1C:4
CC441
r
0 C?
4
0*1*10.Nb
(D 0cn
I--
4Cn
(D:4 C
(D r
0.00D
*
0.
0
0
*
0*41
(D
1
(DiN:4
4
b
**0
Di
CC
14 (D (D
-'
0
(*CODCC
C
0T
C:
Di
~
14:
CD
C:
N
CD
H1 4"
4,4-(D
0'
44
~
CDD01CC
(
14
(D
N
H
0*
Din *
CD4
i0.4
*
0'
CD~ *
N 1-'. 4
40'
0
*
0.
10n *
0
C14-0~
4
4T0 *..0
*
0
4
C3 (
nO. *
4-'. *
(Di (D 0 C 4:444
1
*
H, *
D
r-CDCD 4"
DiCD 4
41*
OCDI
NCD
*
4-1D1
rt*
004
0C?*
CD
*
0*
NCD 4
CD I'..,C
4
4-C??
Di'-
a CCA
0
H
0
Ib
rt1H
C
(D
4110Sb
CD
(D
SCDi'.0
CD
P
KOO
4
*
1-. 1-'. 4
00
*
COCD 4
44*4'
Il
r
:11 (D
t3CDI
(n
'a
r-'0
4
**4'.nH
(D
*t
04:
4-' CD
0.41
r
0'
CD
00
A
*C
C
(D 0
4*
~
0
H1
H1
41
D(*,C
00
(04H4.D0
,
*0'D
0
CCCCC
0
tA
-
(D
10
I,t
0 0
+ 0
4
444
mi
r4
**
0'j0
(D
(D
lt
0l
laO'
004
0.0.4
DiN
41
0.0
*
1-.-
*
4
4
(D
*
4
4-1~4
0.
*
CD
4
4:
*
4'.
4
Di
*
0
H4 0
4*
41.
NNN
NH
X
0
0
0
t-0.O
l<
N
x
Or r
NN
(DH
ý?
m
0(
MM
X
O
w-''<
n00n
0
0z
(D
(t
0×
0CV
0
r 0 xCCHO
0 00
OO
000
<00
0'
I
rrv
0'
04
-4
W H
Ni
CCz
0H
C?
44 0
I(D
0
0
0400
oo
00
''0
(D00
M
I--
p
OC'0
CC
DC'
C
C
N
400 P
YX
(
t-h M
t A
( C:
HHD
00
C?
mo
OH
P)D
00
014
CD'
(D''C
l''0'-*
0
Z3,
I
CD
r.
vl0.
0<
'4
et
W( ;
+
010
H 0
HO
H
CD0.-C
H 00
0
En\
+00 ?
0
0
O '
CD H
HO
000
CC
0''
ND
0-,
(?
D
'CD
0-'
0)
z'-h
CC
0
'-
C
o
d4
(D
10
0
0p
N
H'
M (D
?
(D
CD
C
0
0'
0"
0
(D
A
<
II LQ 10
CD
01
0
I
0"
'--0(
6p w
(00D
0
*COOCD
>4 OH CC
ON
0
31
C-
WOA
N..
0.
0) C?: +0I 0P (DC
0<-(04' 0
C?-'.
0
C?
Cr
-I4 Cd C? + ZC
-C
C?<
C?
C1
CD
0
10
0 (CD?
4l
(D-
4-
(D
(D -(D1
tt
CC
m
0o
OCD
D
00C
0"0
CCC
,
N
P.~
C?
O
0
0'
ND
NIh
0
<a
(D (D
o N
(D<(D
t
(D
<C
(D'
H(D
H+
IIHC?<
C:D
N
N
0.
o Ht
0ft
00
CD0n
*
*
0,
D
~
ý3 F,
0 In M rr
rb
It
i
00
0
0
*C In Qii01j
M
.H
rx (D
*DizH
(D~
*HtIir
It
N
(D
0
*
R,
-D
~ ~ ~*CI~0
( -.- D
-.
I-'
-
ft
CD
ft
CD
ft
ft
.
H
P
0t
0
0
-r
0
(
ft,
Mi
D
01
-
Dii
I- H
D*1-1
ft
P
Hn
H
H D
tD0i-
ft
CD
-3
0m
~
~
01,
1
LOO(D
I-- 00
ft*0
w
t-h~-2
00
H
H
>i
Vi-0
0
w-D
00
CD
CD
CD
D- CA
Nx
H-
D
H
p 0-d
8*.
0'0<
- -.
VV
t
- 1-5U
P-
.' 0
(DC
H
-q
0D
H
mmi
V
0
0D(- (D (D D (D(D(D(
l(
i-H
0,0
ti.
0
H
01
H
m w
HO0HO'O'
- - -
V
~
000
DI0
b--C
riO 0t Wr
0x
P.HO
* *00
CD I-1:1
00
CD0. m
IDIHO
I-.i-n t0 i
0.
H'*
CT<
0 0
,f,,
trt r00
0
H
w w
HO
- --
r
r-0
0C
End
~.
I-r
O
0 0
-
Di-j
F1OAD
(DC(D
'.
Q*
rt0IrC W
y t3I0.
CDt DICDi-rt
M tj
t
H*r
-0.0
CD
, --0 :3
-
vi 0 0
Z Z
0 rr
NO
*
*3
1-3
0<
X
-,0I'-r0
N
m
0~
fA
0t *I-,T
O-'
0 r
L0':1
00
(D
0('0
0 C
r
H
0r
N
0
H
Mih i
Irl
iD
'I
.H
r
Dift
1-< (D
(D*
DIn In
0 0
m
(D- r(
*C~iO~iDiEn
0 0
-ii
0.- O
r
H H
M00
0.
itCDO '
0l0
i..a0r
Mil
D.i•.CDH
+"f- i
•.+,0
0(D 'a ft0IiO
H
it I
M
rl""
P- C
(D 1-t
fC
i(D
D
I-,.
:3
r't(M
I
0ft
DCD
C
(lift (
I- rr.-,
U
.+
<I
I
Ift
-
'0r
-- 0-'
CD-C (Dt
•
(DII--
(D
-c
'0
-
Ent-it<
0(Da
D3
CD
HCD
I--H.
f
HD (D "
0
I'Di
<CD -
ij
13
(i6*~
D
CD
10ftH.0.0.
(D0-i-*
C
- ts•
'
-q.
(
X.11. 1 ý
X'
H-H-M
~
ft '0,
Clt0
H,..
0
H
(D
0 0 1l
OCD
X0 0- ft
H.
ft V- HJ
Di
0
0
H--..-.
0 M Mi
(D --
C
ft (D H-
0
+
P.
ftC
0
-Di
O
) (ft
cc5
00
CD Di
(D
it i.
Di.
0
0tC
I-'D •.D
C
-ft .D- VHCD<
Di.
Di
0 ft 0-,'0
(D
0
ft
rC.
.
-,
it
DiH
CD
(D
Di
(D
CDD0
<<0.(ICI
Mi....
.H- D
ii
CD-'r
4
w
O•
Q-L
D .<
Iit
-T
it
it
CD
OC
Di.
Ft.
CD
ft
10rt
I--
f1t
ft
P.
(iD
*
Di
f
CD
P*MZ
-
O to
CD
--
00
---
toC
D
0.
(iiiD
N
mi
A
Ol
(CD
<
(n
(D
0
ft
0
ft
P.
-0.
r"
(Di
ft
0
ftf
-A0
CD
05H
-
0
CDH
-- 0
(D
(D000
dp
(D CD 0
rt
(iDi
+0
CD
Z1H0
0.0r
"
W.
(
i
CI
H-
i
~T
rr
0 0• 0
Di
(D
In
OH-0
X
0.0
H- (D
-g
*
w
CD
H'
00
f
0C
I-i +.I-
0
' 0
'0
CD
a
N Mi0
H
ftI.-L<H(D
0n
N
CDD
II t CD 0
0
N
S000
0
H.
MI0t
(CD
CD
(D
F0
D 0)-
OIl
CDC(D
(DO
i--'
0 0
OH
0
f
b0
D
ft ,
<-00C(D
--
'
D
Hi
(Di
HD~
I-i
it
HH
0O
C0ft
0 - tfl-
ft
(iiI~
D
CD
0t
ftI-t
• I-0.,
Di0 -r0
(t--D -C(Diw
ft
0D0
ftr
0
1-C~( rt C)OCD
C ()
I-b
it
0- -0.
CD
Ht
M00
f
00 x
(D WO
C
CL..
00
t-
lM
ft0'.Mi+MiC
S--h-1
00
HO
10.0
0
HA
CIA
0
.
1-
CIll
It
HIt
et P. r
P.t
P.c
P
0
(ID
0
C? P-- C? H- C? Fl
rt
(D
-(D
-- D
CCHr
CC00
(CD
I-
(D-(D--(D-(D--(D--(D
+I
It C-
"-0- 0-
1:-4
11
0-
11
+
rt
1<
0
l<
H
CFC
0
r
Cr
•
C-
r
(
f-
.
-
:
*t
~C) CDtr
CD
*D
-r
00
-
(D
C?
0
'1
o
1• 0
0
DC
C?
x
C?
(D
0
CC
o
0
*r
tH
DiD
4.C?*
0~
?D0
(.~
1.
(-' C D
MDDx. r
CD
CD
0(D
- 41
CCC
rr
*j
IC C-l (-
W
C
S(
ICCDCPOH
0
DO
4C
rr b.1
(DC
zC
CD (D
1
CDI
DC
DC 0
0(1
rCC
0
CCC
?.h
H *
CCC
.C(D
Fl U
.CC-
Cl D
C~
(DCC~
fl C ?
C'ri-
D
4
References
1 A. D. Van Riemsdyk: Ann. Chem. Phys., 1880, vol. 20, p. 66.
2 J. L. Walker: Principlesof Solidification, ed. B. Chalmers, John Wiley and Sons, Inc., New York, 1964,
p. 114.
3 G. A. Colligan and B. S. Bayles: Acta. Metall., 1962, vol. 10, pp. 895-897.
4 T. J. Piccone, Y. Wu, Y. Shiohara, and M. C. Flemings: Solidification Processing 1987, The Institute
of Metals, 1988, pp. 268-270.
5 R. Willnecker, D. M. Herlach, and B. Feuerbacher: Phys. Rev. Lett., 1989, vol. 62 (23), pp. 2707-2710.
6 W. H. Hofmeister, R. J. Bayuzick, and M. B. Robinson: Rev. Sci. Instrum., 1990, vol. 61 (8), pp.
2220-2223.
7 K. Eckler and D. M. Herlach: Mater. Sci. Eng. A, 1994, vol. A178, pp. 159-162.
8 B. T. Bassler, W. H. Hofmeister, G. Carro, and R. J. Bayuzick: Metall. Trans. A, 1994, vol. 25A, pp.
1301-1307.
9 J. Lipton, W. Kurz, and R. Trivedi: Acta Metall., 1987, vol. 35 (4), pp. 957-964.
10 J. L. Walker: The Physical Chemistry of Process Metallurgy, ed. G. R. St. Pierre, Interscience, New
York, 1959, Pt. 2, p. 845.
11 G. A. Colligan, V. A. Suprenant, and F. D. Lemkey: J. Met., 1961, vol. 13, p. 691-692.
12 E. Schleip, R. Willnecker, D. M. Herlach, and G. P. G6rler: Mater. Sci. Eng., 1988, vol. 98, pp. 3942.
13 R. E. Cech: Trans. AIME, 1956, vol. 206, pp. 585-589.
14 T. F. Kelly and J. B. VanderSande: InternationalJournal of Rapid Solidification, 1987, vol. 3, pp. 5179.
15 Y. Kim, H. Lin, and T. Kelly: Acta Metall., 1988, vol. 36 (9), pp. 2525-2536.
16 D. J. Thoma and J. H. Perepezko: Metall. Trans. A, 1992, vol. 23A, pp. 1347-1362.
17 M. R. Libera, P. P. Bolsaitis, R. E. Spjut, and J. B. VanderSande: J. Mater. Res., 1988, vol. 3 (3), pp.
441-452.
18 Q. Zhao, T. J. Piccone, Y. Shiohara, and M. C. Flemings: Rapid Quenching and Powder Preparation,
Proc. MRS Int. Meeting on Advanced Materials, 1989, vol. 3, MRS, Pittsburgh, PA, pp. 597602.
19 D. M. Herlach, B. Feuerbacher, and E. Schleip: Mater. Sci. Eng. A, 1991, vol. A133, pp. 795-798.
20 Y. Chuang, K. Hsieh, and Y. Chang: Metall. Trans. A,1986, vol. 17A, pp. 1373-1380.
21 M. Barth, K. Eckler, and D. M. Herlach: Mater. Sci. Eng. A, 1991, vol. A133, pp. 790-794.
22 T. Z. Kattamis and M. C. Flemings: Mod. Castings, 1967, v. 52, pp. 191-198.
23 G. J. Abbaschian and M. C. Flemings: Metall. Trans. A, 1983, vol. 14A, pp. 1147-1157.
24 M. J. Haugh: Theory and Practice of Radiation Pyrometry, ed. D. P. DeWitt and G. D. Nutter, John
Wiley and Sons, Inc., New York, 1988, p. 911.
25 G. P. Ivantsov: Dokl. Akad. Nauk. SSSR, 1947, vol. 58, p. 567.
26 M. Schwarz, A. Karma, K. Eckler, and D. M. Herlach: Phys. Rev. Lett., 1994, vol. 73 (10), pp. 13801383.
27 K. K. Leung, C. P. Chiu, and H. W. Kui: Scripta Metallurgicaet Materiala, 1995, vol. 32 (10), pp.
1559-1563.
28 T. J. Piccone: Sc.D. Thesis, Massachusetts Institute of Technology, 1990.
29 W. J. Boettinger and S. R. Coriell: Science and Technology of the UndercooledMelt, ed. P. R. Sahm,
H. Jones, and C. M. Adam, Martinus Nijhoff, Dordrecht, Germany, 1986, p. 81.
30 M. J. Azia: J. Applied Phys., 1982, vol. 53, p. 1158.
31 D. Turnbull: Metall. Trans. A, 1981, vol. 12A, p. 693.
100