3 Lecture Hours ... Introduction to Organic and Biochemistry, Custom Edition, ISBN-13:
advertisement

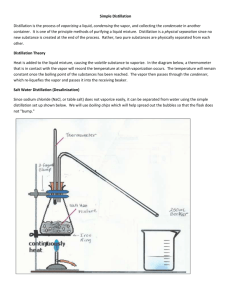
QUEENSBOROUGH COMMUNITY COLLEGE CHEMISTRY DEPARTMENT COURSE SYLLABUS CH-128: INTRODUCTORY ORGANIC CHEMISTRY Pre-Requisite CH-120, CH-127 or CH-151 Hours 3 Lecture Hours Textbook Introduction to Organic and Biochemistry, Custom Edition, ISBN-13: 978-1-305-31493-1 by Bettelheim, Brown, Campbell, and Farrell (Cengage Learning). Customized from ‘Introduction to General, Organic and Biochemistry’, 10th Edition, ISBN-13: 978-1-133-10508-4 by Bettelheim, Brown, Campbell, Farrell, and Torres. Lab manual Organic Chemistry Laboratory Manual, 2nd Ed, ISBN-0-697-33923-8 by P. Svoronos, E. Sarlo and R. J. Kulawiec (WCB McGraw-Hill) 4 Laboratory Hours 4 ½ Credits Course Description This course is the second of a two-semester sequence and is intended to provide a brief, but thorough introduction to organic chemistry and biochemistry. The major functional groups such as hydrocarbons, alcohols, ethers, amines, carbonyl compounds and their derivatives are studied with some emphasis on nomenclature, reactions, and stereochemistry. Several aspects of organic chemistry related to biochemistry are also stressed including units on amino acids, proteins, enzymes, carbohydrates and lipids. The laboratory introduces students to the various synthetic methods for making organic compounds, as well as to purification techniques like distillation, recrystallization and extraction. Curricula for Which the Course is Required/Recommended This course is recommended for students in Nursing and others planning to pursue careers in the Allied Health fields. It may be used as a preparation for CH-251, but may not be substituted for CH-251 and is not open to students who have already completed CH-251 or CH-252. General Education Objectives The general education objectives are to: a) communicate effectively through reading, writing, listening and speaking, b) use analytical reasoning to identify issues or problems and evaluate evidence in order to make informed decisions, c) integrate knowledge and skills in their program of study, d) work collaboratively in diverse groups directed at accomplishing learning objectives, and e) employ concepts and methods of the natural and physical sciences to make informed judgments. Spring 2016 CH-128 SYLLABUS 2/10 Course Objectives/Expected Student Learning Outcomes Understand the nomenclature, structures and reactions of simple organic compounds: alkanes, alkenes, alkynes, alcohols, amines, aldehydes, ketones, amides, esters, carboxylic acids and their derivatives. Attain knowledge of basic concepts of biomolecules such as amino acids, proteins, enzymes, carbohydrates and lipids. Correlate structures and reactivity of simple organic compounds and develop scientific research abilities such as planning and performing experiments, gathering and analyzing data, drawing conclusions from qualitative analysis, communicating results orally and in scientific writing and demonstrating some familiarity with chemical literature. Methods by Which Student Learning Will Be Evaluated The general guidelines for assessing grades are as follows: Examinations, Assignments and Classroom Performance Laboratory Work Final Examination 50% 25% 25% The distribution may be changed at the discretion of the individual instructor. Aside from the above guidelines, the students are mandated to take the American Chemical Society (ACS) assessment test which is administered during the 14th week of the laboratory period (see Laboratory Schedule) and 10% of that grade will be added on top of the student’s final course grade. The ACS test cannot hurt your grade. Attendance/Absence Policy for Lecture Attendance will be taken at every class. The Student Handbook states that you will be considered excessively absent from a course and will receive a WU grade if you have been absent for 15% or more of the total number of contact hours for your course. If there is a laboratory component to your course, you will be considered excessively absent if you miss 15% or more of either component. A WU is computed as an F in your GPA. Students who have valid excuses for missed classes should speak with their instructor and present documentation explaining the reason for the absence. Absences that have been excused by the instructor will not be counted toward a WU grade. If your class meets twice per week: you will receive a grade of WU if you have a total of 6 or more excused and/or unexcused absences. For any lecture that meets only once per week, you will receive a grade of WU if you have a total of 3 or more excused and/or unexcused absences. Accommodations for Students with Disabilities Any student who feels that he/she may need an accommodation based upon the impact of a disability should contact the office of Services for Students with Disabilities in Science Building, room 132 (718-631-6257) to discuss his/her specific needs and to CH-128 SYLLABUS 3/10 coordinate reasonable accommodations for documented disabilities. Students should also contact their instructor privately to discuss their specific needs. Academic Integrity Academic honesty is taken extremely seriously and is expected of all students. All assignments must be the original work of the student (and partners or group, if applicable). All questions or concerns regarding ethical conduct should be brought to the course instructor. “It is the official policy of the College that all acts or attempted acts that are violations of academic integrity be reported to the Office of Student Affairs (OSA). At the faculty member’s discretion and with the concurrence of the student or students involved, some cases, though reported to the OSA, may be resolved within the confines of the course and department. The instructor has the authority to adjust the offender’s grades as deemed appropriate, including assigning an F to the assignment or exercise or, in more serious cases, an F to the student for the entire course.” (Taken from the QCC Academic Integrity Policy, 2/14/2005.) Laboratory Policy All lab policies will be explained in detail by your lab instructor. Three (3) or more absences from the lab earn a grade of WU in the lab. For CH127/128, CH-151/152, CH-251/252, a WU in the lab results in a WU for the entire course. The first and last lab meetings are mandatory and count against your total attendance. There are no make-up sessions for missed labs. Policies regarding excused and unexcused absences will be explained by your lab instructor. A full lab report is required for each experiment and is due the next class period. Your lab instructor will describe the format for lab reports, as well as requirements for entry into the lab. Students who arrive after the pre-lab lecture may not participate and will be marked absent. Safety in the lab is extremely important. Therefore, the ACS safety video must be viewed during the first lab session. A safety quiz must be taken and passed, and the safety declaration sheet must be signed. A student that shows up for the lab, but who has not seen the safety video and passed the quiz, will not be permitted to conduct the experiment, will be considered absent, and will receive a zero for the lab. There will be several additional showings of the safety video during the first three weeks of classes. A student who has not viewed the safety video and passed the quiz by the third lab session will have accumulated 3 absences in the lab and therefore will not be permitted to continue in the course. They may either withdraw or receive a WU for the course. Required attire: Students MUST wear safety goggles in the lab at all times. Shorts and short skirts, tank tops and cropped tops, sandals and open-toed shoes, untied long hair, and any type of food or beverage in the lab are forbidden. Students who fail to follow these rules will not be permitted to perform the experiment. They will be marked absent and will be given a zero that lab. CH-128 SYLLABUS 4/10 CH-128: INTRODUCTORY ORGANIC CHEMISTRY LECTURE SCHEDULE CHAPTER TOPIC HOURS 1 (10) Organic Chemistry 1 2 (11) Alkanes 4 3 (12) Alkenes and Alkynes 3 4 (13) Benzene and Its Derivatives 2 5 (14) Alcohols, Ethers, and Thiols 3 6 (15) Chirality: The Handedness of Molecules 3 7 (16) Amines 3 8 (17) Aldehydes and Ketones 3 9 (18) Carboxylic Acids 3 10 (19) Carboxylic Anhydrides, Esters, and Amides 3 13 (22) Proteins 3 14 (23) Enzymes 3 11 (20) Carbohydrates 3 12 (21) Lipids 2 Exams 6 NOTE: 1. The approximate hours per chapter are guidelines and are at the discretion of the instructor. The instructor is responsible for making assignments and scheduling examinations. The Final Exam date is scheduled by the Registrar. 2. Numbers in the parenthesis under ‘Chapter’ indicate the chapters in the 10th edition textbook. CH-128 SYLLABUS 5/10 Lab Report Format The pre-lab report (#1 - 5, up to “Experimental Procedure” section) must be completed before coming to lab and the final report (#1-9) should be completed and submitted by the next lab period. Prelab Report 1. Your Name and Partner’s Name 2. Title: The title of the experiment. The title should be simple and specific. e.g. Synthesis of Cyclohexene (not “Synthesis of Alkene” because there are many different alkenes) 3. Objective: (1-2 sentences) Why do you do this experiment? What is the goal of this experiment? e.g. “to separate benzoic acid and p-dichlorobenzene from the mixture using the solvent extraction technique” * Do not mention the procedure or the background information here. 4. List of Materials and Equipments: Include important properties of all chemicals (molecular weight, boiling and melting points, density, etc.). Do not include standard laboratory reagents. The only time the reaction apparatus is discussed in the section is when the apparatus is not a standard apparatus. Please keep in mind that beakers, Erlenmeyer flasks, funnels, graduate cylinder, test tubes, etc. are all standard laboratory glassware and they do not need to be mentioned. 5. Experimental Procedure: You will need to write an accurate procedure that will permit you to complete the experiment. Use numbers or bullets for each step, and make each step simple. The purpose for having the flowchart in lab is to ensure that you have read over the procedure ahead of time. It is not safe to begin lab work without the adequate preparation. The experimental procedure should be written on the left half of the page, leaving the right half for the observations section. (Example) 5. Procedure 6. Observations (1) cyclohexanol (10g) is added to a 50mL round bottom flask. (2) Na2Cr2O7 (10g) is dissolved in 10 mL water in a 125mL Erlenmeyer flask. (3) ………….. (4) …………………… (5) ……. (6) do simple distillation…………….. (7) determine the bp and calculate the %yield * Instructor’s Signature _________________________ CH-128 SYLLABUS 6/10 Post lab Report: 6. Observations: Write down anything you observed (color change, formation of bubble, weight of compound used and/ or obtained, m.p., b.p., etc). What you write here will be used for the “Results” and/or “Discussion” sections. (Example) 5. Procedure 6. Observations (1) cyclohexanol (10g) is added to a 50mL round bottom flask. (2) Na2Cr2O7 (10g) is dissolved in 10 mL water in a 125mL Erlenmeyer flask. (3) ………….. (4) ……. (5) ……… (6) do simple distillation…………….. (7) determine the bp and calculate the %yield 9.934g cyclohexanol added 10.1 g Na2Cr2O7 added color changed from orange to brown The distillate was collected between 90-100°C, total about 8mL b.p. = 98°C, 7. Results: Summarize your results here. Record the amount of reactants and products, the appearance of your product, the melting point or boiling point, the percent yield, graph, etc. Describe how you calculated the results using the proper units. Although you write your data and results in “Observations” section, you have to re-write them here using complete sentences. 8. Discussion/Conclusion: The experimental results are always discussed in this section. Describe if and how the experimental objectives were met. If tests were performed to confirm the product, describe how you made your decision from the test results. If you have unexpected results such as low or high % yield, etc., explain and justify the results. 9. Questions: Answer the post laboratory questions assigned in the laboratory manual. CH-128 SYLLABUS 7/10 CH-128: INTRODUCTORY ORGANIC CHEMISTRY LABORATORY SCHEDULE WEEK TOPIC EXPERIMENT PAGES 1 Check-in; Introductory Remarks Laboratory Safety & Equipment; The Basics 1 1- 4 2 Distillation - Simple & Fractional (p 33, #1, 2) 4.1 & 4.2 29 – 32* 3** Recrystallization of a Solid (p 71, #1, 2) 6.3 66 – 70 4 Separation by Extraction (p 65, #1) 6.2 63 – 65 5 Dehydration of tert-Amyl Alcohol 6 Synthesis of t-Butyl Chloride (p 153, #1) 13.1 151 – 152* 7 Qualitative Tests for Alcohols (p 182, #2 and draw all the alcohols on p. 183) 14.2 180 – 182* 8 Oxidation of Cyclohexanol to Cyclohexanone (p 225, #1, 3, and a quiz on percent yield) 17.1 223 – 225* 9 Equivalent Weight of Organic Acids (p 205, #1a, 3) 16.1 204 – 205 10 Synthesis of Aspirin (p 215, #4) 16.3 214 – 215 11 Synthesis of Acetanilide (p 257, #1) 19.2 255 – 256 12 Preparation of Phenacetin (p 186-7, #1, 3, 6) 14.3 185 – 186* 13 Lipids – Preparation and Properties of a Soap & The Hanus Test (p. 307, #1, 2, 3) 23.1 23.2 305 – 307* 311 – 312* 14 ACS Assessment Test 15 Check-out handout*** NOTE: Questions to be answered in the lab report are in parentheses. * designates labs that have modifications to their procedures. See the next page titled ‘Modifications to the Laboratory Procedures’ ** If the Safety Video has not been seen before this experiment, you will not be allowed to continue in the course! *** handout for the complete lab is attached at the end of this syllabus CH-128 SYLLABUS 8/10 MODIFICATIONS TO THE LABORATORY PROCEDURES EXPERIMENT TITLE DESCRIPTION OF THE MODIFICATION Distillation – Simple & Fractional For Part II, step 2 (p 31) of the simple distillation, collect only 10 mL of distillate. Do the same for the fractional distillation (step 2, p 32). Synthesis of t-Butyl Chloride For step 1 (p 152), do not heat past 55-60 °C. For step 5, substitute sodium sulfate for magnesium sulfate. For step 6, just decant the liquid instead of using a gravity filter and do not perform the simple distillation. For step 7, please check with your instructor first. Qualitative Tests for Alcohols For the Jones’ oxidation (p 180), warm test tubes only if the color does not change. For the Lucas reaction, heat only if the test tubes are not cloudy after 5 minutes. For the Iodoform test, use only 5 drops of both the positive test sample and the unknown and perform each test in a beaker since larger quantities of KI/I2 are needed. Oxidation of Cyclohexanol For step 9 (p 225), use sodium sulfate instead of magnesium sulfate. For step 10, decant the liquid instead of gravity filter and do not perform a distillation. Instead, weigh the liquid and calculate the percent yield. For steps 11 and 12, check with your instructor first. Preparation of Phenacetin For step 2 (p 186), 0.50 g of iodoethane should be changed to 1.95 g). For step 6, use ethyl acetate instead of ether. For step 8, use sodium sulfate instead of magnesium sulfate and decant in step 9 instead of gravity filter. For step 10, use only 4-8 mL of hot water instead of 10-15 mL. For steps 15 and 16, please check with your instructor first. Preparation & Properties of Soap and the Hanus Test For step 1 (p 307) use coconut oil instead of cotton− seed oil. For steps 4-8, use a bar soap to make the soap solution since the soap in step 1 does not provide enough suds. For the Hanus test, for step 1 (p 312), use 5 drops of each oil instead of 10. Then check with your instructor as to whether you will do either step 2 or step 3 (only one is needed). CH-128 SYLLABUS 9/10 DEHYDRATION OF TERT-AMYL ALCOHOL (2-METHYL-2-BUTANOL) – WEEK 5 H2O 2-methyl-2-butene H2SO4 OH 2-methyl-2-butanol Procedure OH2 + + 2-methyl-1-butene 1. Setup a simple distillation using a 100 mL round bottom flask as the reaction flask and a 25 mL round bottom flask as the receiving flask. Keep the receiving flask in an ice bath to reduce evaporation. 2. Place 10 mL of water into the reaction flask and cool in an ice bath for several minutes. 3. Use a funnel to transfer 6 mL of concentrated sulfuric acid into a 10 mL graduated cylinder (note: sulfuric acid is corrosive!). 4. Use a pipette to transfer the concentrated sulfuric acid dropwise to the flask (from step 2) while continuously swirling. 5. Slowly add 10.0 mL (8.1 g, d= 0.81 g/mL) of tert-amyl alcohol to the flask using a pipette. 6. Add several boiling chips and then heat the contents. Collect the distillate that boils at 30-43 °C in a receiving flask (25 mL round bottom flask). Continue to collect as long as the distillate is still flowing into the flask at temperatures above 43°C, but stop the distillation if it goes above 50 °C. 7. Discontinue heating and transfer the distillate to a separatory funnel (using a funnel). 8. Place 10 mL of 10% NaOH into the separatory funnel and swirl cautiously (release the vapor immediately). 9. Separate the bottom aqueous layer and place in the designated waste container. 10. Transfer the top organic layer into a small conical flask, dry the organic layer with approximately 1 g of anhydrous sodium sulfate, and then cover the flask with a watch glass to prevent evaporation. 11. Use a funnel to carefully decant the liquid from step 10 into a round bottom flask and set up a simple distillation (avoid the transfer of any crystals of the sodium sulfate into the distillation set up as they contain water which should be removed). 12. Collect the distillate in a pre-weighed dry vial that is kept in an ice bath to reduce evaporation. The bp range of the mixture should be 30-43 °C. 13. Calculate the % yield of the mixture of alkene isomers. 14. Add 5-10 drops of the distillate into 5 drops of 1% KMnO 4 in a test tube. Record your observations. Add 5-10 drops of the distillate into 5 drops of 1% Br 2/H2O in a separate test tube. Record your observations. CH-128 SYLLABUS 10/10 Questions 1) Write the reactions that represent the addition of KMnO 4 and Br2 to the two alkenes that were formed during the reaction. (for assistance in answering the question, refer to p 123-125 in the laboratory manual which uses cyclohexanol instead of tert-amyl alcohol) 2) Explain the purpose of the following in the above experiment: a. Sulfuric acid b. Simple distillation – in step 1 c. Sodium hydroxide extraction d. Anhydrous sodium sulfate e. Ice-cooled receiving/collecting flask f. Covering the flask with a watch glass in step 10 3) Give all possible alkenes formed during the dehydration of each of the following alcohols: a. 1-methylcyclohexanol b. 2-methylcyclohexanol c. cyclopentylmethanol d. 3,3-dimethyl-1-butanol