Problem 5 - Chapter 22 problem 62 Q :
advertisement

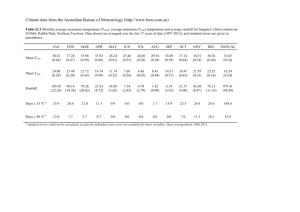
Problem 5 - Chapter 22 problem 62 (a) In any cyclic process the net work done is W = Qh Qc : There is no heat transfer during the adiabatic processes. The heat added and removed to the cycle occurs during the combustion and exhaust phases which are at constant volume and satisfy Q = nCv T: Hence nCv (T4 nCv (T3 Qc =1 Qh eOtto = 1 T1 ) =1 T2 ) T4 T3 T1 T2 During the adiabatic processes for an arbitrary compression ratio r we …nd T3 (V1 =r) T2 (V1 =r) 1 1 = T4 V1 = T1 V1 1 1 ! T3 r1 ! T2 r1 = T4 = T1 Substituting these results into the expression for the e¢ ciency yields (T3 eOtto = 1 T2 ) r1 T3 T2 =1 r1 =1 1 r 1 : Note that everything cancels except for the compression factor r and . Clearly as the compression increases the e¢ ciency increases. (b) To …nd the Carnot e¢ ciency we need the maximum and minimum temperatures. The maximum temperature occurs just after the combustion phase, state 3, while the lowest temperature just after the exhaust phase, state 1. From the ideal gas law the maximum temperature, Tmax ; in terms of the minimum temperature, Tmin ; is T3 P3 V3 3P2 V1 =r 3 Tmax = = = = r ; Tmin T1 P1 V1 P2 (V2 =V1 ) V1 r Tmax = 3 r 1: Tmin The Carnot e¢ ciency is eCarnot = 1 Assuming a diatomic gas, Tmin =1 Tmax 1 1 r : 3 = 1:4: The plot of the two e¢ ciencies is 1 e 1 0.75 0.5 0.25 0 5 10 15 20 r eCarnot is bold, eOtto is thin. Problem 6 - (a), (b) & (c) Chapter 16 problems 32 and 68 (a) For an adiabatic reversible expansion Q = 0; hence the change in entropy for this process is zero. However the temperature of the gas changes via the relation Tf = Ti (Vi =Vf ) 1 : During an adiabatic free expansion, the temperature remains unchanged. Hence after an reversible adiabatic expansion, it is necessary to add heat at constant volume until the …nal temperature is again Ti : The change in entropy for this process is Z Ti Z dT dQ = nCV = nCV ln (Ti =Tf ) : S= T Tf T Substituting the above relation for Ti =Tf we obtain S = nCV ln (Vf =Vi ) S = nR ln (Vf =Vi ) : 1 = nCV ( 1) ln (Vf =Vi ) = n (CP CV ) ln (Vf =Vi ) This is the desired result and demonstrates that the change in entropy via these two processes is identical to that of the single process consisting of a reversible isothermal expansion into this volume. (b) 32. If we measure y as the distance from the bottom of the cable, then a free body diagram shows that at equilibrium the tension at y supports 2 the weight of the cable below that point. This weight is the mass per unit length, ; times yg. Hence the speed of a transverse wave is p p p v = T= = yg= = yg: (c) 68. Since the velocity of the wave up the cable is v= dy dy p ! vdt = ygdt = dy ! dt = p dt gy If the cable is length ` the the propagation time from the bottom of the cable to the top is Z ` Z T dy 1 p ` 1 dt = T = p p = p 2 yj0 g 0 y g 0 p T = 2 `=g 3