Final Exam
advertisement

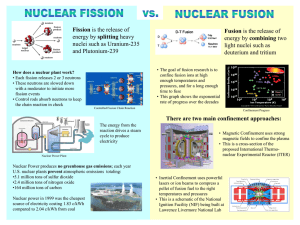
Final Exam • Wed 3/18 from8:00am-11:00 WLH 2005 • Will cover all the course material including the last week • 25 questions –multiple choice. • You are allowed to bring 1sheet of paper with equations on both sides. • Bring a scantron form and a picture id. Nuclear energy Nuclear Fission Nuclear Fusion Energy consumptions vs gross national product Oil Production in the US has peaked Energy Use USA World production is close to peaking 1 World Oil Resources Alternatives to oil Coal Nuclear Energy Fission Fusion Hydroelectric Wind Solar Biomass Curve of the Binding Energy fission Natural radioactivity Many elements found in nature are unstable and decay emitting radioactivity. fusion These include Uranium, 238U , Radon and Potassium 40K. Carbon 14C, 224Ra The half lives of natural radio-isotopes are long. Not useful as sources for power. Low Power output. Energy can be released by Fusion and Fission Induced Nuclear reactions Fission of Uranium Can result in short half lives- fast reactions-high energy density Fermi (1936) Combining nuclei (Fusion) D + 21D ⎯⎯ → 31T + 11H 2 1 Fast neutrons + Energy Slow neutrons Bombard uranium with neutrons Found induced radioactivity Neutron reactions (Fission) n+ 1 0 235 92 U ⎯⎯ → 141 56 Ba + 92 36 Kr + 3 01n + Energy Neutron Source (radon+Be) parafilm uranium “Transuranium elements” 2 Fission of Uranium Liquid Drop model- Strassman and Hahn (1939) Irradiated Uranium with neutrons Detected Barium Conclude Uranium nuclei splits into smaller fragments n+ 1 0 235 92 U ⎯⎯ → 141 56 Ba + other _ products Nuclear Chain reaction Explained fission due to the instability of the higher larger nucleus. Lise Meitner T1/2 ~10-12 s n+ 1 0 235 92 U ⎯⎯ → 236 92 U ⎯⎯ → Ba + products Critical Condition 235U (Fission) n n n (Escape) 235U n 238U (Capture) Nuclear reactor Chain Reaction binding of 1 neutron releases ~3 neutrons Each neutron can initiate another reaction Enriched 235U Reproduction constant K = no. of neutrons that produce a new fission event K=1 ( self –sustained reaction) Centrifuge separation of isotopes Natural Uranium is a mixture of 235U (0.7%) and 238U(99.3%) 238U is does not undergo the fission process but acts as an absorber for neutrons. (neutron capture) Most Uranium nuclear reactors use uranium enriched in 235U. (2-3%) Nuclear weapons used highly enriched 235U. (~90%) Enrichment done by mass separation. Gaseous diffusion Centrifuge process. Laser separation centrifugal separation gaseous UF6 3 Nuclear reactor Nuclear reactor fast neutrons must be slowed down to react efficiently. Moderator- slows neutrons to thermal velocities. Control rods- neutron absorbers to control the level of neutrons Critical condition. – When each neutron released initiates a new reaction. Plutonium Plutonium is a fissionable material created in a nuclear reactor. 238 92 239Pu − U + 01n ⎯⎯ → 239 94 Pu + 2e can be made into nuclear bombs. Pu can be chemically separated from U in spent fuel rods from nuclear reactors. Nuclear Fusion High energy required to bring charged nuclei close together +Z v +Z +Z +Z Nuclear Fusion Fusion of small nuclei releases energy D + 21D ⎯⎯ → 32 He + 01n Q= 3.27 MeV D + 21D ⎯⎯ → 31T + 11H Q= 4.03 MeV D + 31T ⎯⎯ → 42 He + 01n Q=17.6 MeV 2 1 2 1 2 1 Plasma Fusion Magnetic Confinement Plasma is a gas of ionized atoms Heated to high temperature Confined by magnetic forces ~10-15 M Requirements for fusion High Temperatures (T~ 108 K) High density (n) for long time (τ) T~108 K Lawson Criterion nτ > 1014 s/cm3 Princeton Tokomak long times low density 4 Laser fusionInertial Confinement Prospects • Nuclear energy by fission is currently a source of much of the electrical power (~15% USA). • The problems with nuclear energy – Radioactive waste disposal – Atomic bomb threats Deuterium pellet Short times High density Lawrence Livermore Lab Nova Laser • Nuclear fusion reactions promise an unlimited source of energy. – Controlled fusion reactions are not yet possible. 5