Name: ________________ Geometry Guided Notes 6-1 and 6-2
advertisement

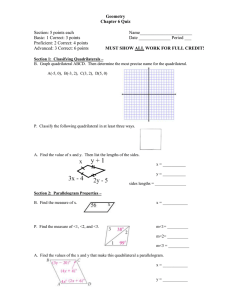
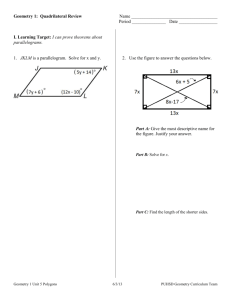
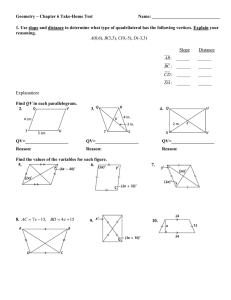
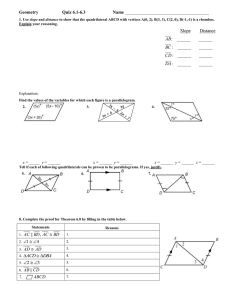
Name: ________________ Geometry Guided Notes 6-1 and 6-2 1. Judging by appearance, classify each quadrilateral in as many ways as possible. What is the most precise name for each? 2. Determine the most precise name for each quadrilateral. A(3, 5) B(7, 6) C(6,2) D(2,1) R (-2, -3) S (4,0) T(3, 2) V(-3, -1) 3. Find the value of x and y. Then find the length of the sides. Solve for x using your knowledge of parallel lines and parallelograms! 4. 5. 6. Find the value of a and the measure of each angle. 7. Set up equations and solve the system to find the values of x and y.