a Responsive release from core- shell assembly
advertisement

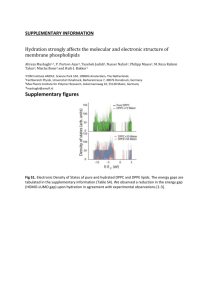
Responsive release from a coreshell assembly Yanjing Chen, Matthew R. Preiss, Arijit Bose, and Geoffrey D. Bothun* Department of Chemical Engineering University of Rhode Island bothun@egr.uri.edu, 401-874-9518 *Associate Professor, Associate Director RI Consortium for Nanoscience and Nanotechnology (RIN2) http://www.egr.uri.edu/che/Faculty/Bothun/lab.html Liposomal release Leaky enough to allow release, but not so leaky that cargo is lost prematurely • Limits range of lipids that can be used • Poor stability L L ++ ++ R∝ ++ ++ 1 DK ∝ Pm = rb t ++ ++ ++ ++ ++ L Activation allows release on demand • Expands range of lipids that can be used (e.g. high melting lipids) • Improves stability and reduces leakiness 1 1 + ra rb ++ L € R∝ 1 1 1 + + ra rb rs € 2 Liposome-nanoparticle assemblies (LNAs) dNP < dcore dNP < ~tb Core encapsulated Bilayer embedded Surface decorated A B C dNP < dSLB D1 Nanoparticle ‘activation’ facilitates release • Magnetic fields – magnetic nanoparticles (e.g. Fe3O4) • Light – metallic nanoparticles (e.g. Au) D2 Complexed A-1 B-1 B-2 200 nm C-1 D2-1 D1-1 (A-1) Wijaya & Hamad-Schifferli, Langmuir, 2007, 23 (B-1) Rasch et al, Nano Lett, 2010, 10 (B-2) Chen & Bothun, ACS Nano, 2010, 4 (C-1) Wu et al, J Am Chem Soc, 2008, 130 (D) Volodkin et al, Angew Chem Int Ed, 2009, 48 3 Magnetic nanoparticles & magnetoliposomes • Maghemite (γ-Fe2O3) or magnetite (Fe3O4) − Small, single domain nanoparticles are superparamagnetic − Low toxicity • Magnetic drug delivery • Magnetic biosensing and diagnostics Permanent fields • MR image contrast agents • Nanoparticle-medicated hyperthermia • Responsive drug delivery Alternating fields De Cuyper & Joniau M, Eur Biophys J, 1988, 15 Soenen et al, Nanomed, 2009, 4 Viroonchatapan et al, J Control Release, 1997, 46 Babincova et al, Bioelectrochemistry, 2002, 55 Pradhan et al, J Control Release, 2010, 142 4 Nanoparticles in EMF fields (at RF) A: Neel relaxation. Magnetic dipoles change direction; magnetic losses heat (< 10-20 nm) B: Brownian relaxation. Magnetic moment does not change; frictional losses heat (> 10-20 nm) ⎛ 2πfτ ⎞ P = πµ0 χ0 H02 f ⎜ 2 ⎟ ⎝ 1+ (2πfτ ) ⎠ µ0 permeability of free space χ0 (POWER DISSIPATION PER VOLUME) equilibrium susceptibility, incorporates ρ = φρ N + (1 − φ ) ρ L , cp = φcp,N + (1− φ )cp,L H0 , f field amplitude and frequency (H = current × # coils per length) € τ effective relaxation time due to Néel and Brownian relaxation € € € ΔT P SAR m NP = = Δt c p ρ c p mV Rate of heating of a mass containing NPs Laurent et al, Chem Rev, 2008, 108 Mornet et al, J Mater Chem, 2004, 14 Pankurst et al, J Phys D, 2003, 36 5 Local nanoscale heating may not be achievable • Water is an excellent heat sink • Bilayer thickness yields minimal heat transfer resistance Ts T∞ , k,c p dq = const dt € dq dT λ∇ 2T + = cp dt € dt ⎛ 1 ⎞ dQ Ts − T∞ = ⎜⎜ ⎟⎟ nano ⎝ 2π krp ⎠ dt R∝ € P rp2 Ts − T∞ = 3k t k € can achieve heating, but local (surface) = bulk volumetric € Keblinski et al, J. Applied Phys, 2006, 100 Gupta et al, J. Applied Phys, 2010, 108 € 6 An example • Can local heating be attained in magnetoliposomes (MLs) and measured directly using an anisotropic fluorescent probe? 12 nm Fe3O4 67% encap. 317 nm dia. dipalmityolphosphatidylcholine (DPPC, C16:0) choline headgroups gel phase (T < Tp) rippled-gel phase (Tp < T < Tm) fluid phase (T > Tm) 0.35 BBBBBBBBBBB 0.30 DPH anisotropy, <r> acyl tails 0.25 J JJ J J JJJ JJ JJJJ B B B DPPC BB B J J BBB J J J JJJ J J JJJ J DPPC + 20 mol% cholesterol J J JJ J 0.20 JJ J J J J J J J JJJJJJJ B JJJ 0.15 0.05 20 J J J J B 0.10 diphenylhexatriene (DPH) JJ BB B BB gel phase (T < Tm ) 25 30 35 40 Temperature, Bothun & Preiss, J Colloid Interf Sci, 2011, 357 BBB BB B 45 BBBB BBBB 50 J JJJ J JJ JJ JJJ J fluid phase (T > Tm ) 55 60 oC 7 In situ heating via membrane order, <r> !.%! <! 5??@54 1 kW, 330 kHz, 250 A 5?? /! %! !.$! $! 54 !.#/ SAR #! &)(8)736=7)*+5>- 012*34',567589*+:7;- !.$/ Cannot distinguish between local and bulk temperatures at the fluorescence time scale 54 !.#! ! "!!! #!!! $!!! %!!! "! &'()*+,8 An approach inspired by biology Transmembrane proteins hydrophobic core and hydrophilic ends Brownian and Neel relaxation Spin/oscillation Estretch ∝ κ a d = 6.5 nm € Ebend ∝ κ b adapted from € Wi et al., J. Phys.: Condens. Matter, 20 (2008) 494211 Y. Chen, A. Bose, G.D. Bothun, ACS Nano 2010, 4(6), 3215-3221 9 Bilayer-embedded SPIO nanoparticles DPPC 5 nm maghemite (γ-Fe2O3) stabilized in chloroform with oleic acid (Ocean NanoTech) B A C 1 - 10 mM DPPC DIUF water emulsification REV at 50 oC 450 mbar, 20 min 300 mbar, 20 min 200 mbar, 10 min 10 mM DPPC DPPC/NP lipid/NP 10,000:1 (0.9 mM FemM, 2O3) 2 - 25,000:1 (0.5 5,000:1 (1.8 mM) 0.08 mg/ml Fe2O3) 3 - 10,000:1 (1.2 mM, 0.19 mg/ml) 4 - 5,000:1 Stored at 50 oC (2.4 (fluid mM, phase)0.38 mg/ml) Solvent phase Dipalmitoylphosphatidylcholine (DPPC) SPIO NPs (as received and washed) 10 Structure and embedment via cryo-TEM DPPC DPPC/NP 25,000:1 ϕNP/L = 0.002 DPPC/NP 10,000:1 ϕNP/L = 0.005 DPPC/NP 5,000:1 ϕNP/L = 0.01 Scale bars = 200 nm 11 Embedded NPs exist partially as aggregates THINK BIG WE DO A B C 200 nm 12 Effect of SPIO NPs on DPPC phase behavior gel phase (T < Tp) rippled-gel phase (Tp < T < Tm) fluid phase (T > Tm) 1 50 Cp (KJ/mol.°C) 40 30 2 20 L/N ratio ΔHm (KJ/mol) ΔTm,1/2 (oC) Control 34.3 1.2 25,000:1 35.7 3.8 10,000:1 37.2 3.5 5,000:1 41.8 5.6 3 4 10 Experiments conducted at 0.1 mM DPPC using a TA Instruments Nano DSC 0 30 35 40 Temperature (°C) 45 50 13 Controlled release from dMLs 25 oC diffusing probe (carboxyfluorecein) CF Leakage (fraction) 1.0 3 0.8 2 0.6 2 4 0.4 1 1 1 2 3 0.2 (1) DPPC (2) DPPC/NP = 25,000:1 (3) DPPC/NP = 10,000:1 (4) DPPC/NP = 5,000:1 1 2 lipid bilayer total release 4 0.0 0 1000 2000 3000 1 Time (s) 1 2 14 Enhanced stability & non-invasive #!! w/heating, optimum 1.5 1.0 € 0.5 DPPC/NP = 10,000:1 30 min heating 1! %2+3(4546(+,7. Initial leakage rate ( s-1) 2.0 0! Hf < 4.85x105 kA m-1 s-1 /! $! 0.0 0.0 0.5 1.0 1.5 mM Fe2O3 2.0 2.5 w/o heating, stabilization ! ! "! #!! #"! $!! $"! %&''()*+,-. 15 Thermomechanical leakage mechanism τN ~ 1x10-9 s τB ~ 4x10-7 s 7 nm w/aggregation, τN decreases f-1 ~ 3x10-6 s, Neel + Brownian L/N = 5,000:1 Before heating L/N = 5,000:1 After heating, 250 A 500 nm 16 Conclusions • Small hydrophobic nanoparticle “triggers” can be embedded in lipid bilayer membranes to control liposomal release – May overcome stability limitations and poor cargo delivery • Release achieved at non-invasive EMF conditions – Transient burst-release mechanism ("$# • Functional and stabilizing lipids • Cytotoxicity (w/o PEG-lipid shown) !"#$%&'%(%)*+ • Restructuring due to embedment ("!# !"'# !"&# !"%# !"$# !"!# !# $# %# &# '# (!# ,-./0.)1&2-.+3µ45+ 17 Bothun Lab & Collaborators Graduate Students & Post-docs Matthew Priess (PhD) Anju Gupta (PhD) Qian Ni (MS) Ashish Airan (MS, Amgen) David Bello (MS) Yogi Kurniawan (PhD) Bastian Weinekotter (MS) Yanjing Chen (PhD, Post-doc) Undergraduate Students Julia Roder-Hanna (ChE, REU) Mark Bicknell (ChE) Paul Spinner (ChE) Eily Cournoyer (ChE) Jeffrey Hanson (ChE, REU) Sara Eldridge (ChE) Ashley Cornell (ChE) John Alper (ChE) Sean Marnane (ChE) Robert DeLuca (CMB) Summer Interns Heather Kumar (Eng, CCRI) Alline Lelis (MB, Providence College) Amy Rabideau (Chem, Syracuse) Allison Boyko (HS) Emily Murphy (HS) Collaborators Arijit Bose (URI ChE) Christopher Kitchens (Clemson U. CBE) Matthew Stoner (URI BPS) Robert Hurt (Brown U. Eng.) Funding National Science Foundation (CBET 0828022, 0914331) National Institutes of Health NCRR RI-INBRE (P20RR016457) NASA RI Space Grant Consortium Rhode Island Consortium for Nanoscience and Nanotechnology (RIN2)