UNIT 2 NOTES 59
advertisement

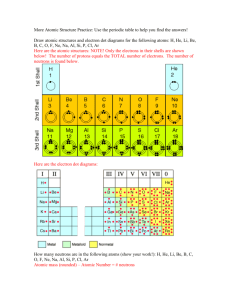
UNIT 2 NOTES 59 Electronic Configuration The location of an electron is described by 3 terms. 1st Term: Shell (n) shell 2nd Term: subshell (sublevel) - designated by s, p,d,f ( g,h,i ) - refers to the shape(s) of the area in which the electron can be located. - also designates an energy level within the shell. The first shell (1) has one subshell (s). The s subshell is spherical in shape and has 1 orbital 3rd Term: orbital Each orbital can accommodate 2 electrons subshell - designated by s, p,d,f - refers to the shape(s) of the area in which the electron can be located. - also designates an energy level within the shell. - relative energy: s < p < d < f s subshell: spherical 1 orbital p subshell: pair of lobes z x y x z y d subshell: double dumbells (note not required to know f or g subshells or name) x y xy xz yz x2-y2 z2 60 orbitals in subshells Sublevel s p d f # Orbitals # electrons 1 2 3 6 5 10 7 14 Remember shells are numbered 1,2,3, etc. Three rules are used to build the electron configuration: – Aufbau principle – Pauli Exclusion Principle – Hund‘s Rule Aufbau Principle • Electrons occupy orbitals of lower energy first. Blocks in the Periodic Table 61 Aufbau Diagram Order of aufbau series shell filling -Pauli Exclusion Principle (Wolfgang Pauli, Austria, 1900-1958) -Electron Spin Quantum Number • An orbital can hold only two electrons and they must have opposite spin. Hund‘s Rule In a set of orbitals, the electrons will fill the orbitals in a way that would give the maximum number of parallel spins (maximum number of unpaired electrons). Analogy: Students could fill each seat of a school bus, one person at a time, before doubling up. 62 Aufbau Diagram for Boron Aufbau Diagram for Nitrogen Notations of Electron Configurations • Standard • Shorthand • Orbital notation/diagram Standard Notation of Fluorine Number of electrons in the sub shell 2,2,5 1s2 2s2 2p5 Subshells 63 Shorthand Notation • Use the last noble gas that is located in the periodic table right before the element. • Write the symbol of the noble gas in brackets. • Write the remaining configuration after the brackets. • Ex: Fluorine: [He] 2s2 2p5 Electron Box Diagram Notation • Draw out a box for each orbital in each subshell • Apply the rules for distributing electrons Blocks in the Periodic Table Our Orbital Picture of Be The first shell (1s) is filled. The 2s orbital has 2 e- present. The 2p orbitals are empty. The 2p orbitals have room for 6 e- 64 Our Orbital Picture of C The first shell (1s) is filled. The 2s orbital has 2 e- present. 2 2p orbitals have 1 e- each. How to draw a Bohr Model. Examples below 65 Electron Configuration Practice Worksheet In the space below, write the unabbreviated electron configurations of the following elements: 1) sodium ________________________________________________ 2) iron ________________________________________________ 3) bromine ________________________________________________ 4) barium ________________________________________________ 5) neptunium ________________________________________________ In the space below, write the abbreviated electron configurations of the following elements: 6) cobalt ________________________________________________ 7) silver ________________________________________________ 8) tellurium ________________________________________________ 9) radium ________________________________________________ 10) lawrencium ________________________________________________ Determine what elements are denoted by the following electron configurations: 11) 1s22s22p63s23p4 ____________________ 12) 1s22s22p63s23p64s23d104p65s1 ____________________ 13) [Kr] 5s24d105p3 ____________________ 14) [Xe] 6s24f145d6 ____________________ 15) [Rn] 7s25f11 ____________________ Determine which of the following electron configurations are not valid: 16) 1s22s22p63s23p64s24d104p5 ____________________ 17) 1s22s22p63s33d5 ____________________ 18) [Ra] 7s25f8 ____________________ 19) [Kr] 5s24d105p5 ____________________ 20) [Xe] ____________________ 66 67 FLAME TEST LAB In this activity, you will investigate the colors of flame produced by solutions of metal salts. When a substance is heated in a flame, the atoms absorb energy from the flame. This absorbed energy allows the electrons to be promoted to excited energy levels. From these excited energy levels, there is a natural tendency for the electrons to make a transition or drop back down to the ground state. When an electron makes a transition from a higher energy level to a lower energy level, a particle of light called a photon is emitted. Both the absorption and emission of energy are quantized – only exact amounts of energy are required. An electron may drop all the way back down to the ground state in a single step, emitting a photon in the process. Alternatively, an electron may drop back down to the ground state in a series of smaller steps, emitting a photon with each step. In either case, the energy of each emitted photon is equal to the difference in energy between the excited state and the state to which the electron relaxes. The energy of the emitted photon determines the color of light observed in the flame. In this activity, q-tips dipped in solutions of metal salts are heated using a Bunsen burner, producing different colored flames. By comparing the color given off by an unknown with the known metal salts, the identity of the metal salt can be determined. Flame Tests Activity Materials: Bunsen burner Q-tips Solutions (1.0 mol/L) of the following metal salts lithium chloride strontium chloride calcium chloride copper(II)chloride sodium chloride potassium chloride UNKNOWN Procedure: 1. Light the Bunsen burner and open the air vent to obtain a non-luminous flame with two blue cones. Be sure to avoid a yellow flame. (Why?) 2. A each station. Carefully place the end of q-tip that was soaked in the metal salt solution at the top of the inner blue cone. Record the color and intensity (bright/faint) of the flame in the data table. The color given off by the salt is the initial color observed, not the yellow-orange color produced by the burning q-tip. (To avoid burning the q-tip, wave it through the flame rather than holding it right in the flame). 3. Repeat with the other 7 stations. Be sure to record the colors as ―precisely‖ as possible. Data Table: Metal found in the salt Flame Color and Intensity Lithium Strontium 68 Calcium Copper Sodium Potassium UNKNOWN Identity:_____________ 69 Flame Test Analysis 1. List the colors observed in this lab from the highest energy to the lowest energy. 2. List the colors observed in this lab from the highest frequency to the lowest frequency. 3. List the colors observed in this lab from the shortest wavelength to the longest wavelength. 4. What is the relationship between energy, frequency, and wavelength? 5. Do you think we can use the flame test to determine the identity of unknowns in a mixture? Why or why not? 6. How are electrons ―excited‖ in this part of the experiment? What does it mean when the electrons are ―excited‖? 7. Explain why we did not see distinct lines (like on an emission spectrum) when the metal salts were burned. 8. Why do different chemicals emit different colors of light? 9. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted? 10. Colorful light emissions are applicable to everyday life. Where else have you observed colorful light emissions? Are these light emission applications related? Explain. 70 Development of a New Atomic Model Connections… • Last time- Democritus, Thomson, Dalton, Rutherford • In 1900s, new model evolved as a result of studying light absorbed and emitted by matter Wave Description of Light • Electromagnetic radiation (ER): form of energy that exhibits wavelike behavior as it travels through space • All forms of ER together make the electromagnetic spectrum 71 • All forms of ER move at a constant speed of about 3.0 x108 m/s (speed of light, c) • Wavelength (λ): distance between corresponding points on adjacent waves. • Frequency (ν): number of waves that pass a specific point in a given time, usually one second. • Unit: Hertz (Hz), aka (1/s) or (s-1) • For electromagnetic radiation, frequency and wavelength are related C=λν • If λ increases, what must happen to ν? Does c change? Photoelectric Effect • Photoelectric effect: emission of electrons from a metal when light shines on that metal • Experiment: no e- were emitted if light’s frequency was below a certain threshold, regardless of time shone. • According to wave theory, shining any light long enough should supply enough energy to eject an e- Particle description of light • Max Planck, 1900s, suggested that object emit energy in small, specific amounts called quanta • Quantum: minimum amount of energy that can be gained or lost by an atom 72 • Planck proposed a relationship between a quantum of energy and the frequency of radiation E=hν • E is energy, in Joules, of a quantum of radiation • h is Planck’s constant (fundamental physical constant)= 6.626 x 10-34 J*s • ν is freqency of radiation • 1905 Einstein expands on this idea. ER have dual wave/particle nature. • While light emits many wavelike particles, it can also be thought of as a stream of those particles • Einstein named the particles photons • Photon: particle of ER having zero mass and carrying a quantum of energy • Energy of a particular photon depends on the frequency of the radiation Ephoton= hν • Einstein’s Explanation: ER is absorbed by matter only in whole numbers of photons – For e- to be ejected, must be struck by single photon possessing at least minumum energy – According to equation, this energy corresponds to frequency – If photon’s frequency is below minimum, no e- ejected The Hydrogen Atom • Ground state: lowest energy state of an atom • Excited state: atom has higher potential energy than ground state • When an excited atom returns to ground state, it gives off energy it gained in the form of ER. • Production of colored signs (neon) is example 73 Experiment • Pass electrical current through H gas in vacuum tube at low pressure • Emits characteristic pink glow • When the light was passed through prism, separated into series of specific frequencies and therefore wavelength (… equation?) of visible light • These bands are Hydrogen’s line emission spectrum Problem? • Classical theory predicted that H atoms would be excited by whatever amount of energy was added to them. • Expected to observe continuous range of frequencies of ER, or continuous spectrum. • Why had H only given off specific frequencies of light? • Quantum Theory 74 • Excited H atom falls back from excited state to ground state, and emits photon • This energy is equal to difference between initial and final state Implications • Since H atoms emit only specific frequencies, difference between energy states must be fixed. • Therefore, e- of H atom exists only in very specific energy states • In 1913, Bohr proposed a model that linked the atom’s electron with photon emission • Energy is higher in orbits farther from nucleus (like a ladder) • Based on the wavelengths of hydrogen’s line emission spectrum, Bohr calculated energies the e- would have in the allowed energy level for H atom. • Bohr’s calculated values agreed with experimentally observed values for lines in each series • Scientists tried to apply this model to other element’s atoms 75 Energy Calculations: 1. What is the energy of a mole of photons with a wavelength of 1.60 10-3 m? 2. What is the energy of a photon with a wavelength of 2.65 10-4 m? 3. What is the frequency of a photon with a wavelength of 3.70 10-6 m? 4. What is the frequency of a photon with a wavelength of 8.60 103 nm? 5. What is the energy of a photon with a wavelength of 6.55 102 nm? 6. What is the wavelength of a photon with an energy of 3.40 10-18 J? 7. What is the wavelength of a photon with a frequency of 2.35 1012 s-1? 8. What is the energy of a photon emitted by the 3 to 1 transition in a hydrogen atom? a. What is the wavelength of the photon emitted, in meters and nanometers? 76 GENERAL PROPERTIES OF THE PERIODIC TABLE Mendeleev's Periodic Table (1869) A. Organization 1. Vertical columns in atomic weight order a. Mendeleev placed elements in rows with similar properties 2. Horizontal rows have similar chemical properties B. Missing Elements 1. Gaps existed in Mendeleev‘s table a. Mendeleev predicted the properties of the ―yet to be discovered‖ elements (1) Scandium, germanium and gallium agreed with his predictions C. Unanswered Questions 1. Why didn't some elements fit in order of increasing atomic mass? 2. Why did elements exhibit periodic behavior? Moseley and the Modern Periodic Table (1911) A. Protons and Atomic Number 1. The periodic table was found to be in atomic number order, not atomic mass order B. The Periodic Law 1. The physical and chemical properties of the elements are periodic functions of their atomic numbers 2. Elements with similar properties are found at regular intervals within the periodic table * Moseley was killed in battle in 1915, during WWI. He was only 28 years old Organization of the Table 1. Groups or Families a. Vertical columns containing elements with similar chemical properties 2. Periods (series) a. Horizontal rows of elements 3. Metals and Nonmetals a. A stair-step line on the table separates the metals from the nonmetals b. Metalloids (Semimetals) straddle the line and have properties of both metals and nonmetals 4. Lanthanide and Actinide Series (Inner Transition Metals) a. Metals and man-made metal elements 5. Group 1 – Alkali metals (the most reactive metal elements) (except hydrogen (H) also in this group) 6. Group 2 – Alkaline earth metals (very reactive metal elements) 7. Group 17 – Halogens (the most reactive nonmetal elements) 8. Group 18 – Noble gases (the least reactive elements – inert and very stable) Types of Elements A. Metals 1. Luster 2. Good conductors of heat and electricity 3. Malleable 4. Ductile 5. High tensile strength B. Nonmetals 1. Many nonmetals are gases at room temperature 2. Solid nonmetals tend to be brittle and non-lustrous 3. Poor conductors of heat and electricity C. Metalloids 1. Some properties of metals and some properties of nonmetals 2. Solids at room temperature 3. Semiconductors of electricity D. Noble Gases 1. All are gaseous members of group 18 2. Generally unreactive and stable 77 Trends in the Periodic Table Periodic Table • Prior to 1860 no agreement/method to accurately determine masses of atoms. • First International Congress of Chemists – 1860 – Stanislao Cannizzaro presented method for accurately measuring atomic masses – Looked for relationships between atomic masses and other properties of elements • First tables arranged elements by atomic weight – Could not agree on atomic weights therefore tables were different John Newlands • Noticed elements properties repeated every 8th element when arranged by atomic mass • Named this phenomenon ―the Law of Octaves‖ • Did not work for all elements 78 Julius Lothar Meyer • Developed first modern table – Consisted of 28 elements divided into 6 families – Families (groups) had similar chemical and physical properties – Discovered all elements in same family had same number of valence e- -outermost electrons in highest energy level – Why? Dmitri Mendeleev • Noticed that properties repeat themselves at certain intervals • Arranged all known elements into one table based on properties– 1869 • 1871 - Proposed the ―Periodic Law‖ • Based on the properties spaces were left for unknown elements (Sc, Ga, Ge) 79 • Upon discovery of other elements inconsistencies were found with Mendeleev’s table • Atomic masses improved and they no longer arranged the elements by increasing atomic mass • Why can most elements be arranged by atomic mass? • What was the reason for chemical periodicity? Henry Mosely • Discovered elements contain unique number of protons (atomic number) - 1911 • Arranged elements by atomic number 1913 • Fully explained the Periodic Law Periodic Law • The physical and chemical properties of the elements are periodic functions of their atomic numbers. • Aka – when elements are arranged by increasing atomic number, elements with similar properties appear at regular intervals. 80 Parts…. • • • • • Alkali metals – group 1 Alkaline earth metals – group 2 Halogens – group 17 Transition metals – d block elements Inner Transition metals – Lanthanides (elements 58-71) added in early 1900’s • Have very similar properties – Actinides (elements 90-103) s-Block Elements • Groups 1 & 2 • All elements in group 1 & 2 will have an electron configuration of – ns1 or ns2 where n = highest energy level occupied Alkali Metals • Group 1 elements • In the elemental state – Soft – Silvery metal – High melting points – Extremely reactive therefore are not found in elemental state in nature • React violently with water to produce hydrogen gas Alkaline – Earth Metals • Group 2 elements • Outer most s orbital is full – Do not exhibit stability (outer p orbital is empty) • Properties – Harder, denser than group 1 – Higher melting points than group 1 – Not as reactive but too reactive to be found in nature in elemental form 81 Burning Mg Hydrogen & Helium • H has same valence electrons as group 1 but does not share any other properties • He share same electron configuration (valence e-) as group 2 but does not share same properties – Placed with group 18 because it is very stable d-block elements • Transition elements – Beginning filling the 3d orbitals – Good conductors of electricity – High luster – Less reactive than s-block elements • Can be found in elemental form Exceptions in the d-block • The following elements have odd configurations – Cr: [Ar]4s13d5 – Cu: [Ar]4s13d10 – Ag: [Kr]5s14d10 • More stable with half filled s & d orbitals or full d orbital • Exceptions follow throughout the d element similar to Chromium and Copper 82 p-block elements • All elements in p block have a full s orbital • Properties – Contain all non metals except H & He – Contain all metalloids (exhibit properties of both metals and non metals) • Have semi conducting properties – Contains 6 metals • Elements in s & p block make up the representative elements Halogens • Group 7A/17 – Most reactive non metals (Fluorine is most reactive) – Will bond with a metal to form a salt – F & Cl are gases at room temp – Br is a liquid at room temp – I & At are solids at room temp Atomic Radius • Atomic radius is simply the radius of the atom, an indication of the atom's volume. • Atomic radius is one-half the distance between the two nuclei in a molecule consisting of two identical atoms. Trends in Atomic Size • - Period - atomic radius decreases as you go from left to right across a period. • Why? Stronger attractive forces in atoms (as you go from left to right) between the opposite charges in the nucleus and electron cloud cause the atom to be 'sucked' together a little tighter. 83 Trends in Atomic Size cont. • Group - atomic radius increases as you go down a group. Why? • There is a significant jump in the size of the nucleus (protons + neutrons) each time you move from period to period down a group. • Additionally, new energy levels of elections clouds are added to the atom as you move from period to period down a group, making the each atom significantly more massive, both is mass and volume. Electronegativity • Electronegativity is an atom's 'desire' to grab another atom's electrons. Electronegativity Trends • Period - electronegativity increases as you go from left to right across a period. • Why? Elements on the left of the period table have 1 -2 valence electrons and would rather give those few valence electrons away (to achieve the octet in a lower energy level) than grab another atom's electrons. As a result, they have low electronegativity. Elements on the right side of the period table only need a few electrons to complete the octet, so they have strong desire to grab another atom's electrons. 84 Electronegativity Trends cont. • Group - electronegativity decreases as you go down a group. • Why? Elements near the top of the period table have few electrons to begin with; every electron is a big deal. They have a stronger desire to acquire more electrons. Elements near the bottom of the chart have so many electrons that loosing or acquiring an electron is not as big a deal. This is due to the shielding effect where electrons in lower energy levels shield the positive charge of the nucleus from outer electrons resulting in those outer electrons not being as tightly bound to the atom. Ionization Energy • Ionization energy is the amount of energy required to remove the outermost electron/s. • Ionization energy is closely related to electronegativity. Ionization Energy Trends • Period - ionization energy increases as you go from left to right across a period. • Why? Elements on the right of the chart want to take others atom's electron (not given them up) because they are close to achieving the octet. The means it will require more energy to remove the outer most electron. Elements on the left of the chart would prefer to give up their electrons so it is easy to remove them, requiring less energy (low ionization energy). 85 Ionization Energy Trends cont. • Group - ionization energy decreases as you go down a group. • Why? The shielding effect makes it easier to remove the outer most electrons from those atoms that have many electrons (those near the bottom of the chart). Reactivity Reactivity refers to how likely or vigorously an atom is to react with other substances. This is usually determined by two things: 1) How easily electrons can be removed (ionization energy) from an atom 86 2) or how badly an atom wants to take other atom's electrons (electronegativity) The transfer/interaction of electrons is the basis of chemical reactions. Reactivity of Metals Period - reactivity decreases as you go from left to right across a period. Group - reactivity increases as you go down a group Why? The farther to the left and down the periodic chart you go, the easier it is for electrons to be given or taken away, resulting in higher reactivity. Reactivity of Non-Metals Period - reactivity increases as you go from the left to the right across a period. Group - reactivity decreases as you go down the group. Why? The farther right and up you go on the periodic table, the higher the electronegativity, resulting in a more vigorous exchange of electron. 87 Ionic Radius vs. Atomic Radius • Metals - the atomic radius of a metal is generally larger than the ionic radius of the same element. • Why? Generally, metals loose electrons to achieve the octet. This creates a larger positive charge in the nucleus than the negative charge in the electron cloud, causing the electron cloud to be drawn a little closer to the nucleus as an ion. Ionic Radius vs. Atomic Radius cont. Non-metals - the atomic radius of a nonmetal is generally smaller than the ionic radius of the same element. Why? Generally, non-metals loose electrons to achieve the octet. This creates a larger negative charge in the electron cloud than positive charge in the nucleus, causing the electron cloud to 'puff out' a little bit as an ion. Ionic Radius vs. Atomic Radius Summary of Periodic Trends 88 Periodic Table Trend War Purpose: To teach students periodic trends regarding the properties of elements. Materials: 1 die, index cards with main group element symbols written on them (one element per card), a list of trends written on the board numbered from 1 - 6. Trends should include such things as ionization energy, electronegativity, atomic radius, ionic radius, etc… A "wild" category can be included which allows the roller of the dice to choose the trend. Playing the game: • Students are placed in groups of 4 - 5. • Cards are dealt until each student has the same number of cards. Each student should have about 7 cards per hand. • The dealer begins play by throwing the die. The number of the die determines the trend being played. • After the trend for the hand is determined the dealer plays the first card. Play continues to the left of the dealer. The card with the highest value for the current trend wins. The player who takes the hand rolls the die and makes the next lead. Multiple hands may be played. 89 Common Ions H+ Li+ Na+ K+ NH4+ Ag+ 1+ ions Hydrogen Lithium Sodium Potassium Ammonium Silver Mg Ca2+ Ba2+ Zn2+ 2+ ions Magnesium Calcium Barium Zinc Al3+ 3+ ions Aluminum 2+ - H FClBrINO2NO3BrO3ClOClO2ClO3ClO4IO3OHCNHCO3HSO3HSO4C2H3O2MnO4- 1- ions Hydride Fluoride Chloride Bromide Iodide Nitrite Nitrate Bromated Hypochlorite Chlorite Chlorate Perchlorate Iodate Hydroxide Cyanide Bicarbonate Bisulfite Bisulfate Acetate Permanganate O2S2CO32SO32SO42CrO42S2O32C2O42O22- 2 - ions Oxide Sulfide Carbonate Sulfite Sulfate Chromate Thiosulfate Oxalate Peroxide 3- N P3PO33PO43BO33- 3- ions Nitride Phosphide Phosphite Phosphate Borate Variable Charge Metals Fe2+ Fe3+ Latin Names Ferrous Ferric Cu+ Cu2+ Cuprous Cupric Pb2+ Pb4+ Plumbous Plumbic Sn2+ Sn4+ Stannous Stannic Hg2 2+ (1+) Hg2+ Mercurous Mercuric 90 Compounds and Molecules A molecule is made up of two or more atoms chemically combined. A molecule is the smallest part of an element or compound that can exist on its own Compounds are substances made from two or more different elements chemically combined. So what does this phrase ‗chemically combined‘ mean? First we need to note that there are two ways for atoms to combine chemically, and that another word for the process of combining chemically is ‗bonding‘. Bonding occurs because all atoms try to have Bonding a full outer shell, and will lose, gain or share electrons in order to do so There are two separate methods by which atoms join up: 1. Ionic bonding occurs when an atom loses or gains electrons 2. Covalent bonding occurs when an atom shares electrons. Ionic bonding An ion is a charged atom – i.e. an atom which has lost or gained electrons If an atom loses an electron then it becomes a positive ion. If an atom gains an electron then it becomes a negative ion. In ionic bonding positive ions are attracted to negative ions. An ionic bond is a bond formed by the force of attraction between two oppositely charged ions An example of an ionic bond is sodium chloride The bonds in sodium chloride are formed by sodium atoms losing electrons and chlorine atoms gaining electrons. Examples of ionic bonding: (i) Sodium Chloride (NaCl) The atomic number of sodium is 11. It has an electronic configuration of 2, 8, 1. This means that it needs to lose the one electron which it has in its outer shell in order to have a complete outer shell. The atomic number of chlorine is 17. Chlorine has an electronic configuration of 2, 8, 7. This means it needs to gain one electron in order to have a complete outer shell. When a sodium atom bonds with a chlorine atom the sodium atom loses its outer electron to form a positive ion while the chlorine atom gains an electron to form a negative ion. 91 The two atoms now have opposite charges and because opposite charges attract both atoms move off together as a sodium-chloride (NaCl) molecule. Examples of ionic bonding: (ii) Magnesium Oxide (MgO) The atomic number of magnesium is 12. It has an electronic configuration of 2, 8, 2. This means that it needs to lose the two electrons which it has in its outer shell in order to have a complete outer shell. The atomic number of oxygen is 8. Oxygen has an electronic configuration of 2, 6. This means it needs to gain two electrons in order to have a complete outer shell. When a magnesium atom bonds with an oxygen atom the sodium atom loses its two outer electrons to form a positive ion while the oxygen atom gains two electrons to form a negative ion. The two atoms now have opposite charges and because opposite charges attract both atoms move off together as a magnesium-oxide (MgO) molecule. Covalent bonding A covalent bond is a bond formed by different atoms sharing electrons in order to have a complete outer shell Examples of covalent bonds are H2, O2, H2O and CH4 (methane) molecules. The bonds in these molecules are formed by the atoms in the molecules sharing electrons with each other.. Examples of covalent bonding: (i) A hydrogen molecule (H2) The atomic number of hydrogen is 1. It needs to have two electrons in its outer shell, and so if it ‗bonds‘ with another hydrogen atom and they both ‗share‘ their electron with each other, it has the effect of allowing both atoms to have a complete outer shell. Examples of covalent bonding: (ii) An oxygen molecule (O2) The atomic number of oxygen is 8. It has an electronic configuration of 2, 6. It needs to have 8 electrons in its outer shell, and so if it ‗bonds‘ with another oxygen atom and they both ‗share‘ two of their electrons with each other, it has the effect of allowing both atoms to have a complete outer shell. 92 Examples of covalent bonding: (iii) A water molecule (H2O) The atomic number of hydrogen is 1 so it has 1 electron in its outer shell. The atomic number of oxygen is 8 so it has 6 electrons in its outer shell. Oxygen can share 1 electron with one hydrogen atom and a second electron with a second hydrogen atom, which has the effect of allowing all atoms to have a complete outer shell. Examples of covalent bonding: (iv) A methane molecule (CH4) The atomic number of hydrogen is 1 so it has 1 electron in its outer shell. The atomic number of carbon is 6 so it has 4 electrons in its outer shell. Carbon can share 1 electron with each of 4 different hydrogen atoms, which has the effect of allowing all atoms to have a complete outer shell. Properties of Ionic and Covalent compounds Ionic Compounds Usually solid at room temperature High melting and boiling points Soluble in water Conducts electricity Undergo fast reactions Covalent Compounds Usually liquid or gas at room temperature Low melting points Insoluble in water Do not conduct electricity Undergo slow reactions WHAT ARE METALS? A metal is an element that readily forms positive ions (cations) and has metallic bonds. On the periodic table, a diagonal or stair step line drawn from boron (B) to polonium (Po) separates the metals from the nonmetals. Elements on this line are metalloids, sometimes called semi-metals; elements to the lower left are metals; elements to the upper right are nonmetals. Almost 80% of the elements on the periodic table are metals. METALLIC BONDS The valence electrons of metal atoms can drift freely from one part of the metal to another- this is sometimes called a ―sea of electrons‖ Metallic bonds consist of the attraction between these free floating electrons and the positively charged metal ions (cations). This attraction is the “bond” that holds metals together. PHYSICAL PROPERTIES OF METALS Lusterous- they are shiny! High density- atoms are tightly packed. Good conductors of electricity o Reason- electrons can flow freely. Ductile- they can be drawn into wires AND Malleable- they can be hammered into shapes o Reason- cations can slide easily past each other because the sea of electrons insulates them and prevents strong repulsions. 93 CHEMICAL PROPERTIES OF METALS Most metals are chemically unstable and will react will oxygen in the air- that is they form oxides- over varying timescales (for example iron rusts over years and potassium burns in seconds). o The alkali metals react quickest followed by the alkaline earth metals. o The transition metals take much longer to oxidize (such as iron, copper, zinc, nickel). Others, like palladium, platinum and gold, do not react with the atmosphere at all. o Some metals form a barrier layer of oxide on their surface which cannot be penetrated by further oxygen molecules and thus retain their shiny appearance and good conductivity for many decades (like aluminium, some steels, and titanium). ALLOYS Very few metals that you encounter daily are pure metals. Most metals are alloys, a mixture of two or more elements of which at least one is a metal. o Examples: Brass is an alloy of copper and zinc Sterling silver is an alloy of silver (92%) and copper (8%) Stainless steel is an alloy of iron (81%), chromium (18%), nickel (1%), and trace amounts of carbon. Alloys are important because their properties are often superior to those of their component elements. o Examples: Sterling silver is harder and more durable than pure silver, but still soft enough to make jewelry and tableware. Brass is harder and easier to shape than either copper or zinc Practice Questions 1. Name one property of ionic and one property of covalent compounds: 2. Draw a diagram (using suitable circuit symbols) of the circuit needed to investigate the ability of ionic and covalent compounds to conduct electricity. 3. How could you tell whether a compound was ionic or covalent? 4. Draw a diagram showing the ionic bonding in the compound magnesium oxide. 5. Draw a diagram showing the bonding in a water molecule. 6. Draw a labelled diagram to show the type of bonding in an oxygen molecule. 7. Name the type of bonding present in a crystal of sodium chloride. Give one property of this type of bonding: 8. In a negatively charged ion the number of . is greater than the number of 9. In a positively charged ion the number of . is greater than the number of 10. Underline the two elements in the list below that normally exist as molecules: 94 Hydrogen I. II. Chlorine Helium Aluminium Gold. Draw Lewis dot structures for the following elements and ions A. Mg B. Ga C. S D. Sn E. P F. I G. Rb H. Xe I. Cr J. Y K. S1- L. Ga 2+ Draw Lewis structures for the following molecules [single bonds] A. CH4 B. BH3 C. SCl2 D. ICl E. PF3 F. CH3OH G. NH2OH H. PBr3 95 Greek Prefixes Nomenclature (inorganic)Greek prefixes: -Nonmetal + Nonmetal 1. First element retains its name. 3-8 2. Second element gets -ide ending. 4-9 3. Use Greek prefixes to identify the # of atoms 5-10 1– 2345- 678910½- Examples N2O = ______________________ SO3 = ______________________ Now Try These: carbon dioxide ___________________ BrF3 ________________________ diphosphorus pentasulfide __________ CS2 ________________________ 1. Metal retains its name. • make sure ions join to form a neutral compound • 2. Non-metal retains its ionic name. + Examples: Na + Cl ---> ___________ name = __________________________ +2 Ca - + Cl ---> ___________ name = __________________________ + ---> ___________ name = ___________________________ + ---> ___________ name = ___________________________ + ---> ___________ name = ___________________________ Now Try These: lithium bromide ____________ Al2S3___________________________ magnesium hydroxide____________ (NH4)2SO3 _______________________ aluminum acetate ____________ Ba(NO3)2 _________________________ -variable charge metal + nonmetal 1a. Metal gets its charge written w/ Roman # in ( ) after name (w/ new system), -or1b. Metal gets Latin name (w/ old system). 2. Non-metal retains its ionic name. +2 Examples: Fe + Cl ---> ________ name = __________________ ( ) +3 Fe - + Cl ---> ________ name = __________________ ( + ---> ________ name = ____________________ + ---> ________ name = ____________________ ) 96 Now Try These: manganese (IV) chloride __________ Cr(OH)3 ______________________ nickel (III) carbonate __________ CuCO3 ______________________ - Acids (positive ion = ―H+‖ 1. Drop the ending on the negative ion. The -ate ending changes to -ic acid. The -ite ending changes to -ous acid. The -ide ending uses the prefix hydro- and the suffix -ic acid Examples: H2CrO4 = ______________________ HNO2 = ______________________ HCl = ______________________ Now Try These: hydrobromic acid __________ H3PO3 _____________________ perchloric acid ____________ HI __________________________ Example: MgSO4 • 3 H2O = ___________________ Now Try These: calcium phosphate tetrahydrate = _________________ NaC2H3O2 • 2 H20 = ________________ 97 Nomenclature 1. carbon dioxide _________ 2. potassium cyanide _________ 3. selenium disulfide __________________ 4. potassium chlorate _________ 5. nitrous acid _________ 6. zinc sulfate _________ 7. aluminum acetate _________ 8. copper(II) phosphate _________ 9. disilicon trioxide _________ 10. chloric acid _________ 11. sodium chloride _________ 12. aluminum iodide _________ 13. barium cyanide _________ 14. carbon disulfide _________ 15. strontium nitrate _________ 16. cuprous phosphate Cu3PO4 17. phosphorous acid _________ 18. potassium hydroxide _________ 19. bromine heptafluoride _________ 20. lead (II) sulfide _________ 21. carbon monoxide _________ 22. ammonium acetate _________ 23. mercuric borate _________ 24. calcium hydride _________ 25. boron trichloride _________ 26. oxalic acid _________ 27. BrF3 __________________ 28. Li2C2O4 __________________ 29. Fe3(PO4)2 30. SCl4 __________________ 31. KHCO3 __________________ 32. SnI2 __________________ 33. HF hydrofluoric acid 34. PO3 __________________ 35. PO3-3 __________________ 36. CaCO3 __________________ 37. Fe(IO3)2 __________________ 38. CuCO3 copper (II) carbonate 39. CaF2 __________________ 40. HNO3 __________________ 41. (NH4)2S __________________ 42. SO3 __________________ 43. KNO3 __________________ 44. Sn3(PO4)2 __________________ 45. MgS2O3 __________________ 46. Ca2C __________________ 47. H2S __________________ 48. CCl4 __________________ 49. NaHSO3 __________________ 50. NH4OH __________________ 51. H3BO3 __________________ 52. V(BrO3)5 __________________ 53. MgSO4 • 7 H2O ______________________________ 54. sodium acetate pentahydrate __________________ 55. CuCl26 H2O __________________________________________ 98 Chemistry Unit #2 Review: 1. Define a family. _______________________________________________________ 2. What is a period? ________________________________________________________ 3. What is the symbol for the following elements. a. Magnesium _____________ b. Potassium ______________ What are the names of the following elements. a. C __________________ b. Cl _________________ What period are the following elements in? a. He _______________ b. Ge _________________ What group are the following elements? a. Sulfur _______________ b. Ca _________________\ Give me an atom with the following characteristics. a. Halogen _________________ b. Nonmetal ________________ c. Alkali metal ______________ d. metalloid ________________ e. Lanthanide series __________ f. Alkaline Earth metal ________________ g. Transition metal ___________ h. Nobel gas ________________ Write the electron longhand and shorthand configuration for a. Li _______________________________________________________________ 4. 5. 6. 7. 8. 9. b. Na ______________________________________________________________ c. K _______________________________________________________________ What are valence electrons? __________________________________________________ _________________________________________________________________________ 10. How many valence electrons are in the following element? a. F ________ e. O ________ 11. 12. 13. 14. 15. 16. b. Cl ___________ c. Br ____________ d. I _____________ f. S ___________ g. Se ____________ h. Te ____________ On the blank periodic table below Label the s, p, d, and f block elements Create a circle that fills the whole box where the largest atom exists in the periodic table. Put a dot where the smallest atom is in the periodic table. Put a triangle on the box with the atom with the highest electronegativity Put a square in the box with the lowest ionization energy Label with arrows the trends for: atomic radius, ionic radius (metals and nonmetals), ionization energy, and electronegativity Name the following ionic compounds: 17. NaBr __________________________________________ 18. CaO __________________________________________ 99 19. Li2S __________________________________________ 20. MgBr2 __________________________________________ 21. Be(OH)2 __________________________________________ Write the formulas for the following ionic compounds: 22. potassium iodide __________________________________________ 23. magnesium oxide __________________________________________ 24. aluminum chloride __________________________________________ 25. sodium nitrate __________________________________________ Write the names of the following covalent compounds: 26. BF3 __________________________________________ 27. P2Br4 __________________________________________ 28. SF6 __________________________________________ 29. NH3 __________________________________________ 30. NO2 __________________________________________ Write the formulas of the following covalent compounds: 31. nitrogen trichloride __________________________________________ 32. boron carbide __________________________________________ 33. dinitrogen trioxide __________________________________________ 34. phosphorus pentafluoride ______________________________________ 35. Make a chart that compares and contrasts the different characteristics of ionic and covalent bonds. 36 What is the different between a molecular and empirical formula? Give an example. From Electron Configuration Notes: 37. What is the shape of the s orbital? ________________ p orbital?______________ d orbital? _________________ 38. Which of the following orbitals is closest to the nucleus? a. 2s b. 3p c. 1s d. 4d 39. In the wave-mechanical (quantum) model of the atom, orbitals are regions of the most probable locations of: a. protons b. positrons c. neutrons d. electrons 40. (CHALLENGE) Heiseinberg’s Uncertainty Principle states: ____________________________________________________________________________ ____________________________________________________________________________/ 100 41. Identify the following atom, 1s22s22p63s23p64s23d6 _________________________ 42. Write out the orbital notation for F, N, Zn: 43. Write the electron configuration for the above elements: 44.Write the shorthand (noble gas) notation for the above elements: F:_________________________________________________ F:__________________________________________ Zn:________________________________________________ Zn:_________________________________________ Sr:________________________________________________ Sr:_________________________________________ 101