Alkynes
advertisement

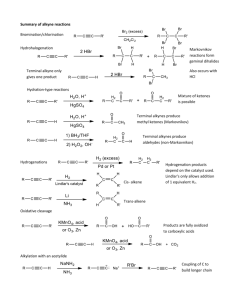
211 LSW ALKYNES F04 Alkynes Doubly unsaturated C≡C functional group Terminal alkynes, R-C≡C-H 1) 2) 3) 4) Nomenclature: Functional group suffix = Disubstituted alkynes, R-C≡C-R' Monosubstituted alkynes, R-C≡C-H Stability: Substituted alkynes The "extra" π bond in an alkyne than the alkene π bond by kJ/mol or kcal/mol kJ/mol or kcal/mol ∆Hh 1-hexyne = ∆Hh 1-hexene = kJ/mol or kcal/mol C≡C - C=C = kJ/mol or kcal/mol C=C - C-C = kJ/mol or kcal/mol 171 211 LSW ALKYNES F04 Rank 1-hexyne (terminal) and 3-hexyne(internal) for each of the following properties: Heat of Heat of hydrogenation formation Stability Heat of combustion Structure: Hybridization: Shape: Y C C X Y C Physical Properties: Polarity: Volatililty: Melting points: Boiling points: Aqueous solubility: Organic solubility: Acidity: pKa = 172 C X 211 LSW ALKYNES F04 Reactivity: Alkynes act as nucleophiles. Addition reactions: Terminal alkynes: R-C≡C-H R C C H B: R C Acetylide ion: 173 C + H-B 211 LSW ALKYNES F04 Reactions: X X NaNH2 C C Alkyne H H Preparation H X NaNH2 C C C C + 2 HX C C + 2 HX H X Hydrogenation H2 / Catalyst C C Metal Reduction Na / NH3(l) C C H + 1 HX H H C C X H X xs HX C C C C C C H C C Hydrogen Halides H C C H X Hydration C C Halogenation C C Ozonolysis Deprotonation Alkylation C C R R C C + H 2O H+ H Hg2+ C C X2 X + O3 C C Then H2O H B: R 1. NaNH2 H 2. R'X 174 H C C X C C HO H O OH C O C R C C + O C + H-B C R' OH 211 LSW ALKYNES F04 Preparation of Alkynes X X C C NaNH2 C C + 2 HX C C + 2 HX H H H X C C NaNH2 H X Reaction Type: Overall reaction: Leaving groups: Requires: Regioselectivity: Stereospecificity: Mechanism: H B: + LG LG Leaving Group Base: Selectivity: 175 211 LSW ALKYNES F04 What is the alkyne product from the reactions of the following with NaNH2 : 1,2-dibromopropane 2,3-dibromooctane 1,2-dibromohexane 2,2-dibromohexane Synthesis: Try it: 2-methyl-4-octene Æ 2-methyl-4-octyne E2 mechanism (For both alkenes and alkynes) Leaving Groups: -LG Base: B 176 211 LSW ALKYNES F04 Stereochemistry E2 reactions 180º 0º Synperiplanar Regioselectivity: Zaitsev's rule: Zaitsev product: Hoffman product: Stereoselectivity: 177 211 LSW ALKYNES F04 Addition Reactions of Alkynes Two factors influence the relative reactivity of alkynes compared to alkenes: Hydrogenation of Alkynes H2 / Catalyst C C H C C H Reaction type: Overall reaction: Regioselectivity: Stereoselectivity: Requirements: Alkanes: Alkenes: Lindlar's catalyst: Pd / CaCO3 / quinoline Mechanism: CATALYTIC HYDROGENATION Step 1: Step 2: Step 3: Step 4: H H H HH HH 178 H HH H H H H 211 LSW ALKYNES F04 Dissolving Metal Reduction of Alkynes Na / NH3(l) C C C C H Reaction type: Overall reaction: H Reactivity order: Regioselectivity: Stereoselectivity: Requirements: Mechanism: Step 1: Step 2: Step 3: Step 4: CH3 CH3 C C CH3 Na C C + Na NH2 H CH3 H C C CH3 + Na CH3 H C CH3 C CH3 NH2 H CH3 H H C + + Na NaNH2 Synthesis: How would you prepare trans 2-pentene from cis 2-pentene? 179 C CH3 211 LSW ALKYNES F04 Reaction of Alkynes with Hydrogen Halides C C C H + 1 HX H X xs HX C X C C C C H X Reaction type: Overall reaction: Reactivity order: Regioselectivity: H CH3 C C H + 1 HX C C CH3 X X H + C C CH3 H H Stereoselectivity: Requirements: CH3 C C H HX CH3 C C H X H H X CH3 C C H + CH3 C C H H X X H + 1 HX H C C hυ CH 3 X H + X C C CH3 H H Mechanism: - + CH3 C C H H CH3 H 180 + Br Br H H CH3 H CH3 Br H H H Br CH3 211 LSW ALKYNES F04 Hydration of Alkynes C C H + H2O + H Hg 2+ H O OH C C H C C Reaction type: Overall reaction: Regioselectivity: Stereoselectivity: Requirements: Mechanism: + CH3 H + C C H CH3 H + O H2O H H CH3 H H H2O O H H CH3 H + H + O H2O H CH3 H H Tautomerization: enol and keto: O HN N H O T H NH O H N N N N NH HN O A T 181 O N NH H N NH H O G O H CH3 H 211 LSW ALKYNES F04 Halogenation of Alkynes C X2 C X Reaction type: Overall reaction: C C X Reactivity order: Regioselectivity: Stereoselectivity: Requirements: Mechanism: Br + Br Br Br _ + Br 182 Br 211 LSW ALKYNES F04 Ozonolysis of Alkynes C C + O3 HO Then H2O C O + O Reaction type: Overall reaction: Requirements: Mechanism: What would be the products of the ozonolysis: ethyne ? 1-butyne ? 2-butyne ? cyclooctyne ? 183 C OH 211 LSW ALKYNES F04 Acidity of Terminal Alkynes R C C Terminal alkynes: C C H B: H R C H C C C + H-B C C H Stability of the conjugate base (i.e. the carbanion). C s character: (sp = %, sp2 = Increased s = = Conjugate base of alkyne = = Mechanism: R C C C C C C H B: C % and sp3 = R C C %) + H-B Could you use a NaOH or NaOEt for this reaction ? pKa H2O = pKa CH3CH2OH = pKa NH3 = 184 211 LSW ALKYNES F04 Alkylation of Alkynes: Acetylide ion: R C C Reaction type: Overall reaction: H 1. NaNH2 2. R'X R C C R' Reactivity order: Alkyne: Alkyl halide: Halide: Regioselectivity: Stereoselectivity: Requirements: Mechanism: CH3 H+ _ RCH2Br NH2 CH3 + NH3 185 CH3 CH2R 211 LSW ALKYNES F04 Examples: Ethylene (acetylene): Æ monosubstituted (R-C≡C-H): Æ symmetrical (R = R'): R-C≡C-R': Æ unsymmetrical (R ≠ R') R-C≡C-R': Problems to try: 1. What is the product of the reactions of CH3-C≡C- with: (a) 2-bromopropane (b) ethanol (c) 1-iodooctane (d) ethyl tosylate (e) (R)-2-bromohexane (f) bromobenzene 2. What would be the product from the reaction of propene with the following reagent sequence: (i) Br2 (ii) NaNH2 , heat (iii) NaNH2 then MeI Final product: 186 211 LSW ALKYNES F04 3. For the reaction 2-butyne with HBr: (i) reaction with 1 equivalent gives 2-bromo-2-butene (ii) reaction with excess gives 2,2-dibromobutane (a) What conclusions can you reach about the reactivity of 2-bromo-2-butene compared to 2-butyne? (b) Consider the two possible carbocations that could be formed in the conversion of 2-bromo-2-butene to 2,2-dibromobutane. Suggest a reason for the observed regioselectivity. 5. Show the major products, with stereochemistry where applicable, for the reactions of: With: 1-pentyne 2-pentyne (i) excess H2 / Pd (ii) H2 / Lindlar Pd (iii) Na / NH3 (iv) 1 equiv. HCl (v) excess HCl (vi) aq. H2SO4, HgSO4 (vii) excess Br2 (viii) O3 then H2O 187