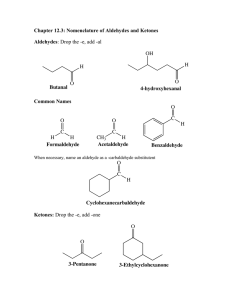
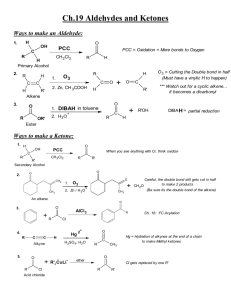
Chapter 13. Aldehydes and Ketones Learning objectives:
advertisement

Chapter 13. Aldehydes and Ketones Learning objectives: 1. Provide both IUPAC and common names for aldehydes and ketones. 2. Recognize the physical properties of for aldehydes and ketones. 3. Write the general electron-pushing (arrow-pushing) mechanisms for the nucleophilic addition to a carbonyl group with the following nucleophiles: Grignard reagent, alcohol, water, and amine. 4. Write the general electron-pushing (arrow-pushing) mechanism for the tautomerization between keto and enol, and explain the observed racemization at the α-carbon. 5. Recognize the reagents for the transformation from aldehyde to carboxylic acid. 6. Recognize the reagents and conditions used for the reduction of aldehyde and ketone to alcohol. 7. Utilize reductive amination for the synthesis of amines. 8. Recognize the reagents and conditions used for α-halogenation. Sections to be covered (in the order of delivery): 13.1 13.2 13.3 13.4 13.5 13.6 13.7 13.8 13.9 13.10 13.11 Introduction# Structure and Bonding Nomenclature Physical Properties Reactions* Addition of Grignard Reagents* Addition of Alcohols* Addition of Ammonia and Amines* Keto-Enol Tautomerism* Oxidation Reduction * Sections that will be focused # Sections that will be skipped Recommended additional problems 13.12 – 13.30, 13.33, 13.35 – 13.39, 13.43 1 13.2 Structure and Bonding O O R H H 13.3 Nomenclature O R H O O R' H H O O H H O H H HO O H O H O Cl H OCH2CH3 O O OCH2CH3 CHO 2 O H H O O O O O O O O O O CH3 13.4 O Physical Properties 3 13.5 Reactions A. In basic condition O O R H (R) Nu O H R H(R) Nu R H(R) tetrahedral intermediate Nu ( Z) B. In acidic condition H B B + H O R O H (R) R H O H H (R) 4 R B H(R) 13.6 Addition of Grignard Reagents A. Formation and Structure of Organomagnesium Compounds B. Reaction with Protic Acids C. Addition of Grignard Reagents to Aldehydes and Ketones 5 13.7 Addition of Alcohols A. Formation of Acetals and Ketals with Water O HO R OH H (R) R H(R) H2O (i) Consideration of pKa (ii) Stability consideration 6 B. Addition of Alcohol: Formation of Hemiacetal, Acetal, Hemiketal, and Ketal) O R'O R'O R OR' OH H R H H R HOR' (2 equivalents) H2O O R'O R'O R OR' OH R R R R R HOR' (2 equivalents) H2O C. Mechanism 7 D. Acetals or Ketals as the Carbonyl-Protecting Groups (i) Stability of Acetals and Ketals (ii) Example O O + O + HOCH2CH2OH H2O (i) OH OH O O H + O O ? H Br H 8 13.8 Addition of Ammonia and Amines A. Formation of Imines O NR' R H H+ R H + H2NR' + H2O (i) Mechanism (ii) Why optimal pH is necessary for the formation of imine? 9 B. Reductive Amination of Aldehydes and Ketones 13.9 Keto-Enol Tautomerism A. Keto and Enol Forms OH O R CH2R' R 10 CHR' B. Racemization at an α-Carbon O C H3C H C CH3 (R)-3-Phenyl-2-butanone Mechanism and Consequence of tautomerization: 11 C. α-Halogenation O O X2 (Cl2, Br2, I2) H3O+ CH3 CH2 X 13.10 Oxidation A. Oxidation of Aldehydes to Carboxylix Acids (i) Examples H2CrO4 O O H2CrO4 (ii) Tollens’ Reagent (Silver-mirror Test) AgNO3 + Ag(NH3)2+ NO3- 2 NH3 O O H + O- 2 Ag(NH3)2+ 12 + 2 Ag + 4 NH3 B. Oxidation of Ketones to Carboxylic Acids (Skipped) 13.11 Reduction A. Catalytic Reduction H2 Raney Ni O H H2 Raney Ni H H2 Pt O O (i) Commonly Used Metal Catalysts: Pd, Pt [Pt(0), Pt(II)], Ni (Raney Nickel), Rh B. Metal Hydride Reduction 1) NaBH4 2) H3O+ O H 1) NaBH4 2) H3O+ O 1) LiAlH4 2) H3O+ O H 1) LiAlH4 2) H3O+ O 13