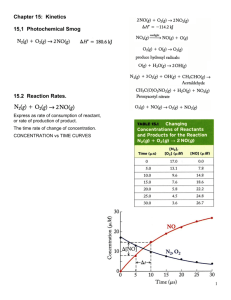
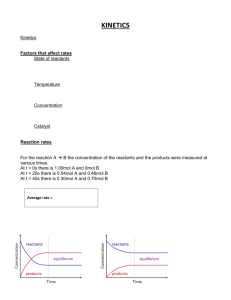
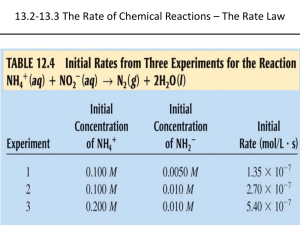
Rate
advertisement

Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Unit 7 (Chp 14): Chemical Kinetics (rates) John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall, Inc. Chemical Kinetics Chemical Kinetics is the study of: 1) Rates at which reactants are consumed or products are produced in a chemical rxn. 2) Factors that affect the rate of a reaction according to Collision Theory (temperature, concentration, surface area, & catalyst). 3) Mechanisms, or sequences of steps, for how a reaction actually occurs. 4) Rate Laws (equations) used to calculate rates, rate constants, concentrations, & time. Reaction Rates Reaction rates are described by the change in concentration (DM) of reactants (consumed) or of products (produced) per change in time. Reaction Rates C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) [C4H9Cl] measured at various times recall: [ ] brackets represent concentration in molarity (M) Reaction Rates C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) D[C4H9Cl] Dt “change in” Reaction Rates C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) average rate slows WHY? fewer reactant collisions Reaction Rates C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) D[C4H9Cl] (rise) (run) Dt • The slope of a line tangent to the curve at any point is the instantaneous rate at that time. Reaction Rates C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) • All reactions slow down over time. • The initial rate of reaction is commonly chosen for analysis and comparison. Reaction Rates and Stoichiometry C4H9Cl(aq) + H2O(l) C4H9OH(aq) + HCl(aq) rate of consumption of reactant = rate of production of product. (IF 1:1 mol ratio) Rate: –D[C4H9Cl] D[C4H9OH] = Dt Dt ↓ reactant = ↑ product Summary Reaction Rates: change in conc. per Reaction: 2 NO2 2 NO + O2 change in time consumption of –D[NO2] reactants per time Dt D[NO2] production of Dt D[NO] products per time Dt 2 NO Are proportional stoichiometrically Rate : 1O 2NO2(g) 2NO(g) + O2(g) 2 D[NO] D[O2] –D[NO2] = = 2 Dt Dt 2 Dt change during rxn Rate Ratios aA + bB HW p. 619 #20 cC + dD 1 D[A] 1 D[B] Rate = − = = − a Dt b Dt 1 D[C] 1 D[D] = c Dt d Dt 2 HI(g) H2(g) + I2(g) Initial rate of production of H2 is 0.050 M∙s–1. What is the rate of consumption of HI? = 0.050 M∙s–1 H2 x 2 mol HI = 0.10 M∙s–1 HI ? mol L∙s 1 mol H2 mol ratio –0.10 M∙s–1 HI Rate Laws Rate equations (or rate laws) have the form: rate = k[A]x[B]y[C]z rate constant order with respect to reactants A, B, & C …or… number of each particle involved in collision that affects the rate overall order of reaction = x + y +… Example: overall order (4 particles – – rate = k[BrO3 ][Br ][H+]2 = ___ 4th order in collision) The Collision Model • reactant bonds break, then product bonds form. • Reaction rates depend on collisions between reactant particles with: activation energy: 1) greater frequency minimum E required 2) enough energy(Eact ) to start reaction 3) proper orientation unsuccessful successful reactants Collision products Reaction Coordinate Diagram transition state activated complex Potential Energy demo Eact …aka… Energy Profile ∆Hrxn Reaction progress Factors That Affect Reaction Rates 4 Factors that Affect Reaction Rates: 1) 2) 3) 4) Concentration Temperature (exposed) Surface Area (particle size) Catalyst Factors That Affect Reaction Rates 1) Concentration of Reactants ↑ concentration, ↑ collision frequency increase pressure of gases Fe(s) + O2(g) Fe2O3(s) 20% of air is O2(g) 100% O2(g) Factors That Affect Reaction Rates 2) Temperature ↑ Temp, ↑ rate collisions of… greater frequency greater energy Eact at higher Temp unsuccessful collisions (bounce off) more particles over Eact successful collisions (react) Temperature and k (rate constant) ↑ Temp, ↑ rate rate = k [A]x k is temp. dependent (k changes with temp) Factors That Affect Reaction Rates 3) Surface Area (particle size) smaller pieces, more exposed surface area for collision. Factors That Affect Reaction Rates potential energy 4) Catalyst ↑ rate by changing the reaction mechanism by… Uncatalyzed Catalyzed 2 H2O2 + 2 Br– + 2 H+ – …lowering the Eact . consumed, then produced (not used up) Br2 (intermediate) H+ 2 H2O2 Br , 2 H2O + O2 reaction progress 2 H2O + O2 + 2 Br– + 2 H+ Surface Catalysts H2 + H2C=CH2 Catalysts can orient reactants to help bonds break and form. H2 + H2C=CH2 H3C–CH3 CH3CH3 Enzymes • biological catalysts in living systems. • A substrate fits into the active site of the enzyme much like a key fits into a lock. (IMAFs work here) HW p. 621 #50,51,64 recall… Rate Laws Rate equations (or rate laws) have the form: rate = k[A]x[B]y[C]z rate constant order with respect to reactants A, B, & C …or… number of each particle involved in collision that affects the rate overall order of reaction = x + y +… Example: overall order (4 particles – – rate = k[BrO3 ][Br ][H+]2 = ___ 4th order in collision) Reaction Mechanisms The sequence of molecular collisions and changes by which reactants become products is called the reaction mechanism. • Rxns may occur in separate elementary steps. • The overall reaction occurs only as fast as the slowest, rate-determining step. (RDS) Slow First Step NO2 (g) + CO (g) NO (g) + CO2 (g) • A proposed mechanism for this reaction is: Step 1: NO2 + NO2 NO3 + NO (slow) Step 2: NO3 + CO NO2 + CO2 (fast) • NO3 intermediate is produced then consumed. • Rate law depends on the slow 1st step: rate = k [NO2]2 • CO is not involved in the slow RDS, so it does not appear in the rate law. (OR…the order w.r.t. CO is ___) 0th Slow Second Step 2 NO(g) + Br2(g) 2 NOBr(g) • A proposed mechanism is: Step 1: NO + Br2 NOBr2 (fast) Step 2: NOBr2 + NO 2 NOBr (slow) • NOBr2 intermediate is produced then consumed. • Rate law depends on the slow 2nd step: rate = k2 [NOBr2] [NO] • But we cannot include intermediate [NOBr2] (b/c it’s difficult to control conc.’s of intermediates). Slow Second Step Step 1: NO + Br2 NOBr2 Step 2: NOBr2 + NO 2 NOBr (fast) (slow) From RDS Step 2: rate = k2 [NOBr2] [NO] b/c step 1 is in equilibrium From Step 1: rateforward = ratereverse k1 [NO] [Br2] = k−1 [NOBr2] solve for [NOBr2] substitute for k1 [NO] [Br ] = [NOBr ] 2 2 [NOBr2] k−1 Slow Second Step Step 1: NO + Br2 NOBr2 Step 2: NOBr2 + NO 2 NOBr HW p. 625 #92, 94 WS Reaction Mechanisms (fast) (slow) rate = k2 [NOBr2] [NO] k k 2 1 [NO] [Br ] [NO] rate = 2 k−1 rate = k [NO]2 [Br2] substitute for k1 [NO] [Br ] = [NOBr ] 2 2 k−1 [NOBr2] recall… Rate Laws Rate equations (or rate laws) have the form: rate = k[A]x[B]y[C]z rate constant order with respect to reactants A, B, & C …or… number of each particle involved in collision that affects the rate overall order of reaction = x + y +… Example: overall order (4 particles – – rate = k[BrO3 ][Br ][H+]2 = ___ 4th order in collision) Orders from Experimental Data: Rate varies with Concentration Initial Concentrations 1 0.0050 0.25 0.30 Rate in M per unit time 10 2 0.010 0.25 0.30 20 3 0.010 0.50 0.30 40 4 0.010 0.50 0.60 160 Experiment [BrO3–] , M [Br–] , M [H+] , M Orders from Experimental Data: Rate varies with Concentration Initial Concentrations 1 0.0050 0.25 0.30 Rate in M per unit time 10 2 0.010 0.25 0.30 20 3 0.010 0.50 0.30 40 4 0.010 0.50 0.60 160 Experiment [BrO3–] , M [Br–] , M [H+] , M By comparing the results from exp.’s A & B: w.r.t. • doubling [BrO3–] doubles the rate. (with respect to) (other conc.’s kept constant) • rate is said to be 1st order w.r.t. [BrO3–] Orders from Experimental Data: Rate varies with Concentration Initial Concentrations 1 0.0050 0.25 0.30 Rate in M per unit time 10 2 0.010 0.25 0.30 20 3 0.010 0.50 0.30 40 4 0.010 0.50 0.60 160 Experiment [BrO3–] , M [Br–] , M [H+] , M By comparing the results from exp.’s B & C: • doubling [Br–] doubles the rate (other conc.’s kept constant) • rate is said to be 1st order w.r.t. [Br–] Orders from Experimental Data: Rate varies with Concentration Initial Concentrations 1 0.0050 0.25 0.30 Rate in M per unit time 10 2 0.010 0.25 0.30 20 3 0.010 0.50 0.30 40 4 0.010 0.50 0.60 160 Experiment [BrO3–] , M [Br–] , M [H+] , M By comparing the results from exp.’s C & D: • doubling [H+] quadruples the rate (other conc.’s kept constant) • rate is said to be 2nd order w.r.t. [H+] Orders from Experimental Data: Rate varies with Concentration Initial Concentrations 1 0.0050 0.25 0.30 Rate in M per unit time 10 2 0.010 0.25 0.30 20 3 0.010 0.50 0.30 40 4 0.010 0.50 0.60 160 Experiment [BrO3–] , M [Br–] , M [H+] , M Rate law is: – – rate = k [BrO3 ] [Br ] [H+]2 Orders in Rate Laws 1)…must be found experimentally (from data). 2)…do NOT come from the coefficients of reactants of an overall reaction. 3)…represent the number of reactant particles (coefficients) in the RDS of the mechanism. 4)…zero order reactants have no effect on rate b/c they do not appear in the RDS of the mechanism (coefficient of 0 in RDS). 5)…are typically 0, 1, 2, but be any # or fraction. Units of k (rate constant) HW p. 618 Units of the rate constant (k) matter. #22,24,28 Rate is usually (M∙s–1), or (M∙min–1), etc. 1st 2nd 3rd Order 0th rate = rate = rate = Rate rate = k[A] k[A]0 k[A]2 k[A]2[B] Law M=? M = ?∙M M = ?∙M2 M = ?∙M3 s s s s k Units M s 1 s M∙s–1 s–1 1 M∙s M–1∙s–1 1 M2∙s M–2∙s–1 HW p. 619 #28 Determine the rate law for the reaction (from experimental data). (a) rate = k [ClO2]x [OH–]y rate1 = (0.0248) = k (0.060)x(0.030)y rate2 = (0.00276) = k (0.020)x(0.030)y x 0.030 y 0.060 (0.0248) = (0.00276) = 0.020 0.030 9= (3)x (1)y 9 = (3)x x = 2 HW p. 619 #28 Determine the rate law for the reaction (from experimental data). (a) rate = k [ClO2]2 [OH–]y rate3 = (0.00828) = k (0.020)2(0.090)y rate2 = (0.00276) = k (0.020)2(0.030)y 3 = (3)y y=1 rate = k [ClO2]2 [OH–]1 OR rate = k [ClO2]2 [OH–] HW p. 619 #28 Calculate the rate constant (with units). (b) rate = k [ClO2]2 [OH–] Exp 1: (0.0248) = k (0.060)2(0.030) (0.0248) k= (0.060)2(0.030) k = 230 M–2∙s–1 M = ?∙M2∙M s HW p. 619 #28 Calculate the rate when [ClO2] = 0.010 M and [OH–] = 0.025 M. (c) rate = k [ClO2]2 [OH–] rate = (230)(0.010)2(0.025) rate = 0.00058 M∙s–1 Rate Laws Differential rate laws express (reveal) the relationship between the concentration of reactants and the rate of the reaction. The differential rate law is usually just called “the rate law.” Integrated rate laws express (reveal) the relationship between concentration of reactants and time. Integrated Rate Laws Using calculus to integrate a first-order rate law gives us: [A]t ln = −kt [A]0 [A]0 = initial conc. of A at t = 0 . [A]t = conc. of A at any time, t . given on exam ln [A]t – ln [A]0 = –kt ln [A]t = –kt + ln [A]0 y = mx + b First-Order Processes ln [A]t = –kt + ln [A]0 y first-order m = –k ln [A] t = mx + b If a reaction is first-order, a plot of ln [A] vs. t is a straight line, and the slope of the line will be –k. First-Order Processes CH3NC CH3CN at 198.9 oC First-Order Processes • When ln P is plotted as a function of time, a straight line results. • Therefore, The process is first-order. k is the negative slope: 5.1 10–5 s−1. Second-Order Processes Similarly, integrating the rate law for a process that is second-order in reactant A, we get… given 1 1 on – = kt [A]t [A]0 exam 1 1 = kt + [A]t [A]0 y = mx + b Second-Order Processes 1 1 = kt + [A]t [A]0 y = mx + b second-order If a reaction is m=k second-order, a plot of 1/[A] vs. t 1 is a straight line, [A] and the slope of the line is k. t Second-Order Processes The decomposition of NO2 at 300°C is described by the equation NO2 (g) NO (g) + 1/2 O2 (g) and yields the following data: Time (s) 0.0 50.0 100.0 200.0 300.0 [NO2], M 0.01000 0.00787 0.00649 0.00481 0.00380 Second-Order Processes • Graphing ln [NO2] vs. t yields: • The plot is NOT a straight line, so the process is NOT first-order in [A]. Time (s) 0.0 50.0 100.0 200.0 300.0 [NO2], M 0.01000 0.00787 ln [NO2] −4.610 −4.845 0.00649 0.00481 0.00380 −5.038 −5.337 −5.573 Second-Order Processes • Graphing 1/[NO2] vs. t, however, gives this plot. • Because this IS a straight line, the process is second-order in [A]. Time (s) 0.0 50.0 100.0 200.0 300.0 [NO2], M 0.01000 0.00787 1/[NO2] 100 127 0.00649 0.00481 0.00380 154 208 263 Zero-Order Processes zero-order [A] t If a reaction is zero-order, a plot of [A] vs. t is a straight line, with a slope = rate. Summary of Integrated Rate Laws and Straight-Line Graphs zero-order [A] HW p. 620 #33, 38, 43 ln [A] first-order m = –k t ln [A]t = –kt + ln [A]0 1 [A] t second-order m=k t 1 1 = kt + [A]t [A]0 Half-life (t1/2): • time at which half of initial amount remains. [A]t = 0.5 [A]0 initial concentration at time, t concentration at time, t = 0 Half-life (t1/2) is constant for 1st order only. Half-life (t1/2) depends on k: For a 1st order process: [A]t1/2 = 0.5 [A]0 ln [A]t – ln [A]0 = –kt ln 0.5 [A]0 – ln [A]0 = –kt1/2 0.5 [A]0 ln = −kt1/2 [A]0 ln 0.5 = −kt1/2 given on exam −0.693 = −kt1/2 0.693 = t1/2 k Half-life & Radiometric Dating A wooden object from an archeological site is subjected to radiometric dating by carbon-14. The activity of the sample due to 14C is measured to be 11.6 disintegrations per second (current amount of C-14). The activity of a carbon sample of equal mass from fresh wood is 15.2 disintegrations per second (assumed as original amount of C-14). The half-life of 14C is 5715 yr. What is the age of the archeological sample? Half-life & Radiometric Dating A wooden object from an archeological site is subjected to radiometric dating by carbon-14. The activity of the sample due to 14C is [A]t = 11.6 current dis/s measured to be 11.6 disintegrations per second at time, t (current amount of C-14). The activity of a carbon sample of equal mass from fresh wood is 15.2 disintegrations perdis/s [A]0 = 15.2 initial second (assumed as originalat amount time, t0of=C-14). 0s The half-life of 14C is 5715 yr. t1/2 = 5715 yr What is the age of the archeological sample? t = ? yr Half-life & Radiometric Dating First, determine the rate constant, k. ln Nt − ln N0 = –kt easy way (given: t1/2 = 5715 yr) ln 0.5N0 − ln N0 = –k(5715) 0.5 N0 ln = −k(5715) N0 ln 0.5 = −k(5715) –0.693 = −k(5715) –0.693 =k –5715 0.693 = t1/2 k 0.693 = 5715 yr k 0.693 =k 5715 yr k = 1.21 10−4 yr−1 Half-life & Radiometric Dating Now we can determine t: ln Nt − ln N0 = –kt ln(11.6) – ln(15.2) = −(1.21 10−4) t –0.270 = −(1.21 10−4) t 2230 yr = t Summary 0th Order 1st Order 2nd Order Rate Law Rate = k Rate = k[A] Rate = k[A]2 ln[A] = –kt + ln[A]0 1 1 kt [ A] [ A]0 Integrated Rate Law [A] = –kt + [A]0 y Linear plot [A] k & slope of line Units of k Half-Life = mx + b ln[A] t t y = mx + b 1 [A] t Slope = –k Slope = –k Slope = k M=? s M = ?∙M s M = ?∙M2 s M∙s–1 s–1 0.693 t1/ 2 k M–1∙s–1 depends on [A]0 HW p. 620 #34,36,40,32 depends on [A]0