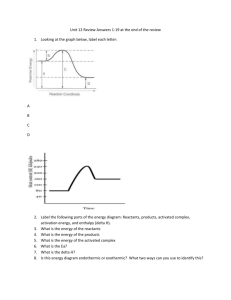
Two metals of equal mass with different heat capacities are
advertisement

A 97 g sample of gold at 785oC is dropped into 323 g of water, which has an initial temperature of 15oC. If gold has a specific heat of 0.129 J/goC, what is the final temperature of the mixture? A 97 g sample of gold at 785oC is dropped into 323 g of water, which has an initial temperature of 15oC. If gold has a specific heat of 0.129 J/goC, what is the final temperature of the mixture? Assume that the gold experiences no change in state of matter. -LOST heat (metal) = GAINED heat (water) - [(s ) (mass) (DT)] = (s ) (mass) (DT) Au H2O -[(0.129 J/goC)(97 g)(Tf - 785oC)] = (4.184 J/goC)(323 g)(Tf -15oC) -[(12.5J/oC)(Tf - 785oC)]= (1.35x 103J/oC)(Tf -15oC) (1.35x 103J/oC) (1.35x 103J/oC) -[(0.009259 )(Tf - 785oC)]= Tf -15oC - 0.009259 Tf + 7.268 oC = Tf -15oC 22.268 oC = 1.009259Tf Tf = 22.0637oC → 22oC Two metals of equal mass with different heat capacities are subjected to the same amount of heat. Which undergoes the smallest change in temperature? 1) The metal with the higher heat capacity. 2) The metal with the lower heat capacity. 3) Both undergo the same change in temperature. 4) You need to know the initial temp of the metals. Convert 5.721 kcal to Joules The reaction pictured in the graph below is ______________. Energy Products Reactants Reaction progress Exothermic Reaction Reactants Products + Energy Energy of reactants Energy Energy of products Reactants -DH Products Reaction Progress When heat flows into a system from the surroundings, this process is ______ Cooking an egg is __________ 1) Exothermic 2) Endothermic Formation of snow in clouds is ______ 1) Exothermic 2) Endothermic Water condensing on the outside of a cold glass on a hot day: 1. Exothermic 2. Endothermic 4Fe + 3O2 2Fe2O3 ΔH= -1652 kJ • How much heat is released when 2.00g of O2 is reacted? Consider the following standard heats of formation: P4O10(s) = –3110 kJ/mol H2O(l) = –286 kJ/mol H3PO4(s) = –1279 kJ/mol Calculate the change in enthalpy for the following: P4O10(s) + 6H2O(l) 4H3PO4(s) Consider the following numbered processes: 1. A 2B 2. B C + D 3. E 2D DH for the process A 2C + E is 1) DH1 + DH2 + DH3 2) DH1 + DH2 3)DH1 + DH2 – DH3 4) DH1 + 2DH2 – DH3 5) DH1 + 2DH2 + DH3 A 0.0459kg sample of a metal is heated to 95.5°C and then placed in a calorimeter containing 120.0 g of water (s = 4.18 J/g°C) at 21.6°C. The final temperature of the water is 24.5°C. Which metal was used? 1) 2) 3) 4) Aluminum (s = 0.89 J/g°C) Iron (s = 0.45 J/g°C) Copper (s = 0.20 J/g°C) Lead (s = 0.14 J/g°C) The heat combustion of acetylene, C2H2(g), at 25°C is –1299 kJ/mol. At this temperature, ΔHf° values for CO2(g) and H2O(l) are –393 and –286 kJ/mol, respectively. Calculate ΔHf° for acetylene. 1) 2) 3) 4) 5) 2376 kJ/mol 625 kJ/mol 227 kJ/mol –625 kJ/mol –227 kJ/mol