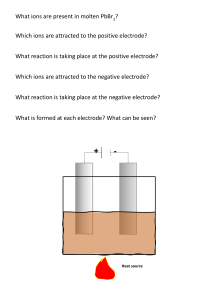
14. CHEMICAL EFFECTS OF ELECTRIC CURRENT 1. Fill in the
advertisement

14. CHEMICAL EFFECTS OF ELECTRIC CURRENT 1. Fill in the blanks: (a) acids, bases, salts (b) chemical (c) negative (d) electroplating 2. When the free ends of a tester are dipped in to a solution containing ion, conduction of current takes place which changes magnetic field around the magnetic compass. Therefore, the magnetic needle shows deflection. 3. Brine, Lemon juice and vinegar. 4. Possible reasons for the bulb not glowing in the given setup: (a) Loose connection in the circuit. (b) Discharged battery. (c) Fused bulb. (d) Absence of ion in the solution for the conduction of electric current. 5. (i) liquid A is a better conductor than liquid B. 6. No, pure water does not conduct electricity as it has no free ions for the conduction of electric current. It can be made conducting by adding few drops of an acid or base or any salt. 7. Tap water or the water used by the foremen contains impurities in the form of dissolved salts which conduct electricity very well. Therefore, to avoid electric shock, they shut off the main electrical supply for the area. 8. Sea water contains more number and different types of ions than tap water which is mainly contains fresh water. Therefore, sea water conducts electricity very well. 9. It is not safe for an electrician to carry out electrical repairs outdoors during heavy downpour because moisture can soak into his insulations and this may cause electrical shock to him. 10. Thought rainwater should be as good as distilled water but due to air pollution the oxides of sulphur, nitrogen and carbon would mix with it and this will cause increase in electrical conductivity of rain water. 11. Cycle rim and other parts, gold plated ornaments, watches, steel buckets and chrome plated parts of motorcycles and cars. 12. Impure electrode of copper metal should be used as a positive terminal as metal from this electrode would erode and pass into the solution. Whereas, the copper metal ions when come in contact with the negative terminal would form a layer after accepting electrons from the electrode.