Electron Dot Diagrams / Lewis Structures
advertisement

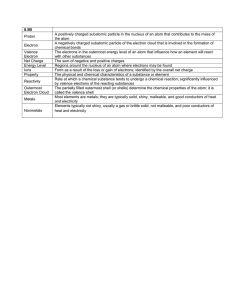
Electron Dot Diagrams / Lewis Structures Atom and Covalent Compound diagramming Electron Dot Diagrams • Shows valence electrons around an atom • Valence electrons – electrons in the outermost energy level of an atom • Valence electrons are only located in the outer level “s” and “p” orbitals • Valence electrons are shown in the outer energy level of a “shell diagram” Electron Shell Diagrams • A shell diagram shows all electrons, but we are most interested in the electrons in the outermost energy level called the valence level or valence shell. Electron Dot Diagrams • To draw: 1. Write chemical symbol 2. Place one dot around chemical symbol for each valence electron (up to eight dots) 3. All inner level electrons and nucleus are symbolized by the chemical symbol Electron Dot Diagrams • Draw electron dot diagrams of the following elements: 1. Carbon 6. Hydrogen 2. Nitrogen 7. Chlorine 3. Phosphorus 8. Argon 4. Boron 9. Iodine 5. Oxygen 10. Helium Electron Dot Diagrams • Line up single dots to form compounds and to make either 2 or 8 electrons around each element. The elements share their single unpaired electrons. The Basics: Drawing Lewis Structures Step 1: Calculate the total number of valence electrons in the molecule or ion Step 2: Determine the central atom(s) of the molecule or ion – usually it’s the least electronegative atom. Step 3: Draw a tentative diagram for the molecule or ion. Rules a) A hydrogen atom always forms one bond. Hydrogen is always a terminal atom in a Lewis diagram – an atom that is bonded to only one other atom. b) A carbon atom normally forms four bonds c) When several carbon atoms appear in the same molecule, the are often bonded to each other. (No cyclic compounds in Chem 60/68) Electron Dot Diagrams • Some electrons are already paired up within a single atom and are not shared. These are called non-bonding / unshared pairs. Electron Dot Diagrams • Some elements are able to share more than one pair of electrons to form double and triple bonds Draw the Lewis Structure for the following molecules. 1. H2O Oxygen has 6 valence electrons & Hydrogen has 1 valence electron for a total of 8 electrons. .. H:O:H .. 2. CO Oxygen has 6 valence electrons & Carbon has 4 valence electrons for a total of 10 electrons. :C:::O: Draw the Lewis Structure for the following molecules. 3. BH3 Boron has 3 valence electrons & Hydrogen has 1 valence electron for a total of 6 electrons H:B:H .. H 4. NH3 Nitrogen has 5 valence electrons & Hydrogen has 1 valence electron for a total of 8 electrons .. H:N:H .. H p. 362 Cations p. 363 Anions p. 366 Resonance p. 366 Homework • Draw electron dot diagrams of the following compounds: 1. CCl4 2. N2O2 3. PBr3 4. BI3 5. C3H6O 6. H2O 7. CH4 8. SO2 9. NH3 10. SiO4