CPES Study/Note Guide 1 text pages: 99-100, 125
advertisement

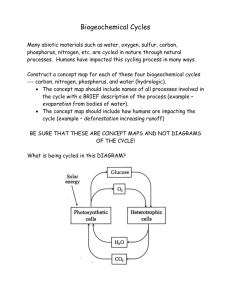
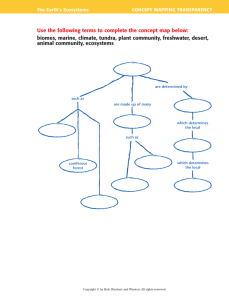
CPES Study/Note Guide 1 text pages: 99-100, 125-133 Ecosystems & the Carbon Cycle; physical science review Unit 1 Standard: Students will investigate the flow of energy and cycling of matter within an ecosystem and relate these phenomena to human society. SEV1a. Interpret biogeochemical cycles including hydrologic, nitrogen, phosphorus, oxygen, and carbon cycles. Recognize that energy is not recycled in ecosystems. Main Idea: Matter (water, nitrogen, phosphorus, oxygen, carbon) is cycled (reused by plants & animals) through ecosystems by chemical reactions and physical changes. Energy is not recycled; it flows and is transferred in ecosystems according to the Laws of Conservation of Matter and Energy through food webs and food chains. 1. Define ecosystem: 2. What 2 things make up ecosystems? 3. Define abiotic factors: 4. Give examples of abiotic factors: 5. Define biotic factors: 6. Give examples of biotic factors: 7. Define matter: 8. Define biogeochemical cycle: 9. Give examples of biogeochemical cycles: 10. Describe carbon: 11. Quick physical science review: What are the parts of an atom? 12. Draw an atom here and label the parts: 13. What is an isotope? Give examples of carbon isotopes. 14. Define half-life: 13. Show the chemical notation for carbon dioxide and describe the 2 elements that make up carbon dioxide. 14. Define reservoir: 15. Write the chemical reaction for photosynthesis: 16. Write the reaction for photosynthesis in English: 17. Write the chemical formula for cellular respiration: 18. Write the reaction for cellular respiration in English: 19. Define fossil fuels: 20. Give examples of fossil fuels: 21. Define combustion: 22. Describe the relationship between combustion, fossil fuels and carbon. 23. Define decomposition: 24. Define limestone: 25. What are some major reservoirs of carbon on the earth? 26. What are some carbon sources?