File
advertisement

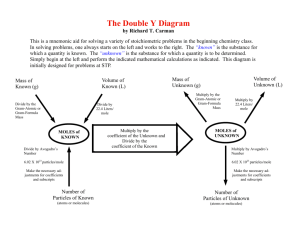
Unit 3: Chemical Reactions Lesson 4 • C11-3-07: Describe the concept of the mole and its importance to measurement in chemistry. • C11-3-08: Calculate the molar mass of various substances. • • • • • • • • • Became a lawyer at the age of 16 Had quite a successful legal career, but showed a lot of interest in science Studied science and became a professor at the age of 23 Avogadro studied the works of scientists like Dalton and Gay-Lussac Determined that gaseous particles of hydrogen, oxygen, nitrogen, etc. were not atoms but "molecules." 1811 – equal volumes of gases at the same temperature and pressure contain the same number of particles: Avogadro's Principle Avogadro's ideas were not immediately accepted because his support came from analyzing other scientists' data and his ideas did not coincide with those of the prominent scientists of his day. Avogadro's work was left forgotten until 1860, when it was presented at a conference as a way to calculate the atomic masses of some elements 1865 Avogadro was vindicated and his work was established as correct. Unfortunately, Avogadro had died several years earlier. • Formula and molecular mass deal with individual atoms and molecules • Chemists do not work with amounts of individual atoms or molecules. • These particles are far too small to see or mass. • Balances or scales do not measure mass in terms of atomic mass units. • Chemists need a practical unit that relates mass, in grams, with the number of particles. • Particle = generic term meaning atoms, molecules ions, or any other entity • The mole (abbreviated mol) is the unit that relates number of particles and mass • Defined as the number of particles in 12.0000 g of carbon containing 6 protons and 6 neutrons. • In one mole of carbon, there are 6.022136736 x 1023 atoms; we will round this to 6.02 x 1023. • To honor Avogadro the scientific community named: 6.02 x 1023 Avogadro's Number and has the symbol NA. • 6.02 x 1023 is a very large number. It is 602 followed by 21 zeros or 602 000 000 000 000 000 000 000 • One mole contains 6.02 x 1023 of anything! • 6.02 x 1023 donuts is one mole of donuts • 6.02 x 1023 pencils is one mole of pencils • 6.02 x 1023 carbon atoms is one mole of carbon atoms • 6.02 x 1023 water molecules is one mole of water molecules • The mole is just a name that describes a number. It’s like a dozen is twelve of anything, a few is three of anything, a mole is 6.02 x 1023 of anything etc. • Avogadro’s Number relates the number of particles to mass • One mole of carbon atoms has a mass of 12.0000 g • If the mass of one mole of any atom is its atomic mass in grams, then: • • • • 1 mole of aluminum atoms = 27.0 g 1 mole of silver atoms = 107.9 g 1 mole of sodium atoms = 23.0 g 1mole of iron atoms = 55.8 g • The mass of 1 mole of any compound = formula mass • EXAMPLE: the formula mass of water is 18.0 amu. The mass of one mole of water molecules is 18.0 g/mol • Molar Mass (M) - the mass of one mole of a substance • The units for molar mass are grams per mole (g/mol) Example 1: What is the molar mass of lead (II) chloride, PbCl2? • Calculate the formula mass. PbCl2 = 1 Pb + 2 Cl PbCl2 = 207.2µ + 2(35.5µ) PbCl2 = 278.2µ • The molar mass is the formula mass in g/mol, so the molar mass of lead (II) chloride is 278.2 g/mol. Example 2: What is the molar mass of ammonium dichromate? • Write the formula. (NH4)2Cr2O7 • Determine the formula mass. (NH4)2Cr2O7 = 2 N + 8 H + 2 Cr + 7 O (NH4)2Cr2O7 = 2(14.0µ) + 8(1.0µ) + 2(52.0µ) + 7(16.0µ) (NH4)2Cr2O7 = 28.0µ + 8.0µ + 104.0µ + 112.0µ (NH4)2Cr2O7 = 252.0µ • (NH4)2Cr2O7 = 252.0 g/mol Example 3: How many moles of carbon atoms in 24.0 g of carbon? Example 4: How many moles of water molecules in 81.0 g of water? Example 5: What is the mass of 2.50 moles of gold? Example 6: What is the mass of 1.20 x 10–5 moles of carbon tetrachloride, CCl4? 1 mole of particles = 6.02 x 1023 particles Example 7: How many atoms in 25.0 moles of copper atoms? The type of atom, molecule, ion, etc. is not significant in this calculation. Example 8: How many molecules of water in 1.50 x 10–5 moles of water? Example 9: How many moles are there in 5.00 x 1025 particles? Example 10: How many moles of atoms is 5 atoms of zinc? Example 11: How many molecules of water in a 10.0 g sample of water? • Example 12: How many atoms in 25.0 g of sodium chloride, NaCl? • Notice the question asks for atoms, not molecules • Since there are 2 atoms, 1 Na and 1 Cl, in each molecule of NaCl, we multiply the number of NaCl molecules by 2. Example 13: What is the mass of 3.01 x 1023 molecules of water? Example 14: What is the mass of a single atom of carbon? • Step 1: Determine the number of moles using Avogadro’s Number. • Step 2: Determine the mass using the molar mass ratio..