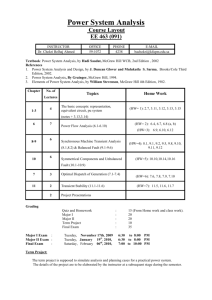
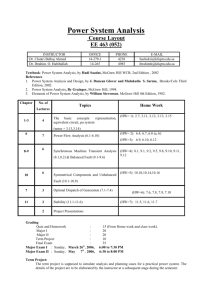
1.1 Safety in the Science Classroom
advertisement

Section 2.1 Introduction to elements Why are elements studied in chemistry? 1) Chemistry is the study of matter and its properties. 2) An element is a pure substance that cannot be broken down or separated into simpler substances. Each element is one kind of atom. 3) By studying elements, we can learn more about the structure of matter. 2.1 Elements Element names and symbols 1) Because elements have different names in different languages, chemists use international symbols for them 2) Chemical symbols consist of one or two letters. 3) Ancient names are used as the source of many of the symbols. Example: Hydrogen (H) Hydros genes = water forming See page 44 All elements are represented by symbols. Here are just a few element symbol examples: Common Elements See page 45 Hydrogen (H) 1) 2) 3) 4) 5) Colorless, odorless, tasteless, and highly flammable gas. Makes up over 90% of the atoms in the universe Used in producing fertilizers Lighter than air Automobiles made to be run on hydrogen rather than gasoline. Why? No pollution like producing CO2, the only by-product is H20 ! Yay water!! Oxygen (O) - gaseous element we breathe See page 45 21 % of the atmosphere Reacts with most other elements Major component of water which covers ¾ of our planet Our cells use oxygen to convert sugar into energy we can use Plants produce Oxygen as a by-product of photosynthesis. For your info, about 65% of your body’s mass is made of Oxygen!!! Iron (Fe) See page 45 - mixed with carbon to make steel - very strong metal - good structural material, but can rust when mixed with water and oxygen Iron in a river turns water and rocks red Sodium (Na) 1) Very soft metal that can even be cut with a knife !!! 2) It also reacts so fast and so violently with water 3) Cannot be used to even hold waterwould react and could cause an explosion (c) McGraw Hill Ryerson 2007 Chlorine Cl 1) Chlorine (Cl) - yellow-green gas that is highly toxic 2) Safe enough in pools (used to kill bacteria) but in high concentrations it can be deadly 3) It combines with sodium to form table salt ! Cl See pages 46 - 47 Other Common Elements 1) Mercury (Hg) - liquid at room temperature metal. 2) Silver (Ag) - precious metal mined in British Columbia 3) Silicon (Si) - brittle, grey, semiconductor that is second most common element in Earth’s crust. Hg Ag Si Homework Worksheet 2.1 (c) McGraw Hill Ryerson 2007 (c) McGraw Hill Ryerson 2007