SUMMARY VALIDATION of the COMPLEMENT COMPONENT C1q
advertisement

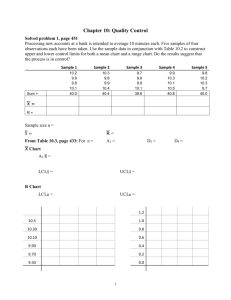
VALIDATION of the COMPLEMENT COMPONENT C1q RID ASSAY Igor Y. Pavlov, Julio C. Delgado ARUP Institute for Clinical and Experimental Pathology, Department of Pathology, University of Utah School of Medicine, Salt Lake City, Utah SUMMARY RESULTS Figure 2. Normalized data: ARUP vs National Jewish Health Center comparison EP Evaluator C1q Research and Development -- ARUP Alternate (Quantitative) Method Comparison X Method: ARUP Y Method: NJH Scatter Plot Bias 0.2 Deming Regr 1:1 Line Percent Bias 15 Mean Bias 10 0.1 1.2 1 Percent Bias 5 Bias (%) NJH (%) C1q is a subunit of the C1 complex that activates the classical pathway of the complement system. The Binding Site radial immunodiffusion assay was validated for the human EDTA plasma samples. It was found that C1q RID assay was accurate and linear in the range between 30 and 230 µg/mL. Recovery of the spiked samples varied between 99% and 110%. Reproducibility from run to run and within runs was below 10%. Reference interval was estimated to be from 109 µg/mL to 242 µg/mL. Comparing results of C1q determination by ARUP, Quest and National Jewish Health Center (NJHC), we found no correlations. Obviously, all 3 kits utilize different proteins as a standard. When results were normalized by the average value of each laboratory set, normalized data sets correlated to each other. Correlation Coefficient R between ARUP and Quest results was very poor, only 0.4; NJHC vs ARUP: R=0.87. Bias for ARUP – Quest comparison was twice as high (30% vs 15%) and confidence intervals for slope and intercept were much wider than for the NJHC – ARUP comparison results. Direct results comparison between different laboratories should not be considered because of the differences in utilized standards and, accordingly, reference intervals. 0 0 -5 -0.1 -10 0.8 -0.2 0.8 INTRODUCTION 1 ARUP (%) -15 1.2 0.8 1 ARUP (%) 1.2 0.8 1 ARUP (%) 1.2 Regression Analysis C1q is a 400kDa hexameric gamma-2 protein that is a subunit of C1 – the first component of complement system. The binding of two or more of C1q’s six globular domains initiates the classical pathway of complement activation. C1q binds readily to the CH2 domains of aggregated IgG molecules in an immune complex or the CH3 domain of a single IgM molecule whose conformation has been altered following antigen binding. It can also bind directly to certain microorganisms and mycoplasmas. The multivalent binding of C1q is believed to lead to a conformational change in C1q complex, activating C1r, then C1s and thereby initiating the classical complement pathway. The collagen-like tail domain of C1q (which is only exposed once C1 inactivator dissociates C1r2 and C1s2 from the C1q-activator complex) increases the phagocytosis of particles by monocytes and macrophages. Serum levels of C1q are reduced in immune complex disease, SLE and meningitis. Hereditary deficiency is also known Slope: Intercept: Std Err Est: Deming Regular 1.237 (0.864 to 1.611) -0.239 (-0.617 to 0.139) 0.100 1.048 (0.692 to 1.403) -0.049 (-0.409 to 0.311) 0.095 95% Confidence Intervals are shown in parentheses Supporting Statistics Corr Coef (R): Bias: X Mean ± SD: Y Mean ± SD: Std Dev Diffs: 0.8701 -0.001 1.002 ± 0.155 1.001 ± 0.187 0.092 SubRange Bounds: Points (Plotted/Total): Outliers: Scatter Plot Bounds: CONCLUSIONS None 15/15 Not Tested 95% CI Experimentvs Description Figure 3. Normalized data: ARUP QUEST comparison Expt Date: Rep SD: Result Ranges: Units: Analyst: Research and Development -- ARUP Comment: EP Evaluator Figure 1. Accuracy and linearity of the C1q RID assay Accepted by: C1q C1q 1.3 QUEST (%) Scatter Plot Percent Recovery 120 1:1 Line Fitted Overall 110 % Recovery 300 200 Y Method: QUEST Scatter Plot Instrument: Plasma* Accuracy and Linearity Measured (µg/mL) 08 Mar 2010 1 0.71 to 1.35 % IP Bias Deming Regr 1:1 Line 1.2 0.2 20 1.1 0.1 10 0 -0.1 -10 0.8 -0.2 -20 -0.3 0.7 100 100 0 0.9 0.7 0.8 0.9 1 1.1 ARUP (%) 1.2 1.3 -30 0.7 0.8 0.9 1 1.1 ARUP (%) 1.2 1.3 0.7 0.8 0.9 90 Regression Analysis 0 80 0 100 200 300 400 0 100 Assigned (µg/mL) 200 300 Linearity Summary N Slope Intercept Error 5 1.026 1.7 4.2% LINEAR within Allowable Systematic Error of 10.0% Overall Deming Regular 1.058 (0.375 to 1.740) -0.059 (-0.751 to 0.632) 0.183 0.410 (-0.152 to 0.971) 0.590 (0.021 to 1.159) 0.151 400 Assigned (µg/mL) Slope: Intercept: Std Err Est: 95% Confidence Intervals are shown in parentheses Supporting Statistics Statistical Analysis and Experimental Results Smpl 5 Smpl 4 Smpl 3 Smpl 2 Smpl 1 Assigned Pct 18.7 6.25% 37.4 12.5% 74.8 25% 149.5 50% 299.0 100% Mean 20.0 41.0 81.0 162.0 296.0 % Rec. 107.0 109.7 108.4 108.4 99.0 Est 20.8 40.0 78.4 155.1 308.6 Resid -0.8 1.0 2.6 6.9 -12.6 Linear? Pass Pass Pass Pass Pass Measured Concentrations 18 22 43 39 81 81 162 162 296 296 See User's Specifications for Pass/Fail criteria 0.4007 -0.001 1.002 ± 0.155 1.001 ± 0.158 0.172 20.0% 50% 10.0% SubRange Bounds: Points (Plotted/Total): Outliers: Scatter Plot Bounds: X: Excluded from calculations Experiment Description Supporting Data User's Specifications Allowable Total Error: Systematic Error Budget: Allowable Systematic Error: Corr Coef (R): Bias: X Mean ± SD: Y Mean ± SD: Std Dev Diffs: Analyst: Date: Value Mode: Units: Lot Number: IP 02 Apr 2010 Pct-Assigned µg/mL Expt Date: Rep SD: Result Ranges: Units: REFERENCES Percent Bias Mean Bias 1 Complement component C1q radial immunodiffusion assay was validated and ARUP reference interval defined. Direct comparison to other laboratories results should not be considered because of differences in utilized standards and, accordingly, reference intervals. 30 Percent Bias Research and Development -- ARUP 400 08 Mar 2010 1 0.73 to 1.30 % IP X Method: ARUP EP Evaluator Radial immunodiffusion assay from (The Binding Site Limited, Birmingham, U.K) was applied to 81 healthy donor plasmas for reference interval determination, 29 serum/plasma pairs of the healthy donor specimens, and to samples with low, medium and high C1q concentrations to access accuracy , linearity and reproducibility of the assay. EP Evaluator software was applied to the result interpretation. Eighteen EDTA plasma samples were sent to the Quest Diagnostics, National Jewish Health Center (NJHC), and run at ARUP for C1q assays. Both Quest and NJHC are utilizing home-brewed radial immunodiffusion kits. Y Method Alternate (Quantitative) Method Comparison Signature Date Bias (%) MATERIALS and METHODS X Method During the validation study we found that C1q RID assay was accurate and linear in the range between 30 and 230 µg/mL with allowable error of 20% with 50% systematic error. Recovery of the spiked samples varied between 99% and 110%. Reproducibility from run to run and within runs was within 10%. By the nature of radial immunodiffusion assay, detection of C1q concentration below 50µg/mL (corresponding to the ring diameter below 5.0 mm) is not reliable: potential reading error of 0.1 – 0.2 mm generates 15% – 30% error in C1q concentration. Reference interval calculated with transformed parametric was estimated to be from 109 µg/mL (90% confidence limits from104 to117 µg/mL) to 242 µg/mL (90% confidence limits from 227 to 258 µg/mL). Comparing results of C1q determination by ARUP, Quest and NJHC, we found no correlations. Obviously, all 3 kits utilize different proteins as a standard. When results were normalized by the average value of each laboratory set, normalized data sets correlated to each other: confidence intervals for slopes included 1, and confidence intervals for the intercept included 0. Correlation Coefficient R between ARUP and Quest results was very poor, only 0.4; NJHC vs ARUP: R=0.87. Bias for ARUP – Quest comparison was twice as high (30% vs 15%) and confidence intervals for slope and intercept were much wider than for the NJHC – ARUP comparison results. X Method Y Method 08 Mar 2010 1 0.73 to 1.30 % 08 Mar 2010 1 0.76 to 1.25 % None 15/15 Not Tested 95% CI 1 1.1 ARUP (%) 1.2 1.3 1. Cooper NR (1985). The classical complement pathway: Activation and regulation of the first complement component. Adv. Immunol. 37, 151-216 2. Arlaud, GJ et al (1987). A functional model of the human C1 complex. Immunol. Today, 8, 106-111. 3. Ross, SC & Densen, P. (1984). Complement deficiency states and infection: Epidemiology, pathogenesis and consequences of neisserial and other infections in an immune deficiency. Medicine, 63, 243-273 4. Schifferli, GA et al (1986). The role of complement and its receptor in the elimination of immune complexes. New Eng. J. 315, 488-495