Lecture 2 - Philadelphia University
advertisement

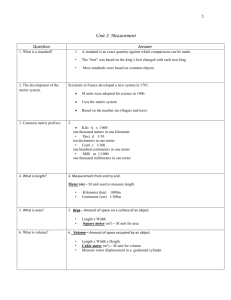
Philadelphia University Faculty of Engineering Department of Civil Engineering First Semester, 2013/2014 ENVIRONMENTAL ENGINEERING 441 Lecture Time: Class 1: 11:15 to 12:45 Monday & Wednesday INTERNATIONAL SYSTEM OF UNITS The International System of Units (abbreviated SI) is the modern form of the metric system. It is the world's most widely used system of units, both in everyday commerce and in science. The older metric system included several groupings of units. The SI was developed in 1960 from the metre-kilogram-second (mks) system, rather than the centimetre-gram-second (cgs) system which, in turn, had many variants. The SI introduced several newly named units. The SI is not static; it is a living set of standards where units are created and definitions are modified with international agreement as measurement technology progresses. With few exceptions, the system is used in every country in the world, and many countries do not maintain official definitions of other units. In the United States, industrial use of SI is increasing, but popular use is still limited. In the United Kingdom, conversion to metric units is official policy but not yet complete. Those countries that still recognize non-SI units (e.g. the U.S. and UK) have redefined their traditional non-SI units in terms of SI units. SI BASE UNITS Type Name Symbol length metre m mass kilogram kg time second s electric current ampere A temperature kelvin K amount of substance mole luminous intensity candela mol cd SI DERIVED UNITS Type Name Symbol inductance henry H mass flow rate kilogram/second kg/s rad mole flow rate mole/second mol/s m2 power watt W pressure pascal Pa speed meter/second m/s surface tension newton/meter N/m torque newton meter N-m voltage volt V Type Name Symbol acceleration meter/square second m/s2 angle radian area square meter capacitance farad density kilogram/cubic meter F kg/m3 dynamic viscosity pascal second Pa-s electric charge coulomb C electric conductance siemens S volume cubic meter m3 electric resistance ohm Ω volume flow rate cubic meter/second m3/s energy joule J force newton N mole/cubic meter frequency hertz Hz amount-ofsubstance concentration mol/m3 SI PREFIXES Factor 1024 1021 1018 1015 1012 109 106 103 102 101 101 10-1 10-2 10-3 10-6 10-9 10-12 10-15 10-18 10-21 10-24 1E24 1E21 1E18 1E15 1E12 1E9 1E6 1E3 1E2 1E1 1E1 1E-1 1E-2 1E-3 1E-6 1E-9 1E-12 1E-15 1E-18 1E-21 1E-24 Prefix yotta zetta exa peta tera giga mega kilo hecto deca deka deci centi milli micro nano pico femto atto zepto yocto Symbol Y Z E P T G M k h da da d c m µ n p f a z y USCS - UNITED STATES CUSTOMARY SYSTEM UNITS USCS - United States Customary System Units: are the measuring units used in the U.S. consisting of the Mile (eq 1609.344 m) Foot (equal 0.3048 m) Inch (eq 0.0254 m) Gallon ( US eq 3.785 L) (UK eq 4.546 L) second Pound (eq 0.45359 Kg) Commonly used USCS and SI-units USCS unit SI unit SI symbol Conversion factor (mutiply USCS unit with factor ) Square foot Cubic foot Pound per square inch Pound force Square meter Cubic meter m2 m3 0.0929 0.2831 Kilopascal kPa 6.894 Newton N 4.448 Foot pound torque Newton meter N·m 1.356 Kip foot Kilonewton meter kN·m 1.356 Gallon per minute Liter per second L/s 0.06309 Kip per square inch Megapascal MPa 6.89 FOR ONLINE CONVERSION YOU CAN USE THE FOLLOWING HTTP://WWW.ONLINECONVERSION.COM/ EXAMPLE #1: CONCENTRATIONS AND CONVERSIONS Some employees at GE wash the PCB tainted floor with organic solvent (TCE) and the discharge enters a holding tank that is 25 m x 25 m x 5 ft and is full with water. The volume of solvent is 3 L and the concentration of PCBs in the solvent is 10 ppm. What is the final concentration of PCB in mg/l in the holding tank? MATERIAL BALANCES MB is a key tools in achieving a quantitative understanding of the behavior of environmental systems. Mass Balances provide us with a tool for modeling the production, transport, and fate of pollutants in the environment. MATERIAL BALANCES / MASS BALNCE Conservation of mass Mass is neither created nor destroyed” Mass that comes in either stays, reacts or goes out. “the sum of weights (masses) of substances entering into a reaction is equal to the sum of weights (masses) of the products of the reaction Feed In Products out Inputs - Output = Accumulation STEADY STATE FLOW CONSERVATIVE SYSTEM: Steady State Flow, conservative system: Σ CinQin = ΣCoutQout Assumption #1: Steady‐state: no change in conc. throughout control volume Assumption #2: Conservative system: No reaction EXAMPLE #1 The Hudson river flows with a flow rate of 300,000 cfs. GE discharges to the Hudson with a flow rate of 10 cfs. The concentration of PCBs in the discharge is 5 g/L. What is the final concentration in the river? downstream from the discharge? Assume perfect mixing. Also assume concentration of PCBs upstream of the discharge is 0 ppm. STRATEGY OF SOLVING MB Sketch a flow chart or figure defining the boundary of the process Label the flow of each stream & their composition with symbols Show all known flows and compositions on the figure. Calculate additional compositions from the data where possible Select the basis for calculations e.g. 1h, 1 day, 1 kg, etc Write the MBs which includes the total balance and component balances. There must be x independent equations if there are x unknowns Solve the equations and check the solutions EXAMPLE #2 A wastewater treatment plant with an output of 38400m3/day discharges the liquid effluent with a BOD of 20mg/L into a river. If the BOD of the river upstream of the discharge point is 0.2mg/l, at a minimum flow of 20m3/s, compare the BOD of the river downstream of the discharge, assuming complete mixing. Answer = 0.63mg/L EXAMPLE #3 A slurry containing 20 percent by weight of limestone (CaCO3) is processes to separate pure dry limestone from water. If feed rate is 2000kg/h, how much CaCO3 is produced per hour? HOME WORK 1. 2. Each day 3780 m3 of wastewater is treated at a municipal wastewater treatment plant. The influent contains 220 mg/L of suspended solids. The clarified water has a suspended solids concentration of 5mg/L. Determine the mass of sludge produced daily from the clarifier and write down the mass balance of the clarifier. As a fuel source 20kg of ethylene (C2H4) is burned with 400 kg of air. Determine the composition of the resulting mixture. What is the percentage of excess air, assuming complete conversation? ANALYSIS PERFORMANCE OF REACTOR TYPE Reactor influent Qin CAin Effluent V (m3) Q out C Aout Input – output+ generation= accumulation GENERAL MATERIAL BALANCE EQUATION FOR FIRST ORDER REACTION RATES: Reactor influent Qin CAin Effluent V (m3) Q out C Aout VdC/dt =ΣCinQin − ΣCoutQout ± kCV C = concentration in the control volume (river/stream/reactor) [=] mg/L V = volume of control volume [=] L, m3, ft3 Qin = flowrate of inlet streams [=]m3/s, L/s, cfs, MGD Qout = summation of all outlet streams [=]m3/s, L/s, cfs, MGD Cin = concentration in each inlet stream [= ] mg/L Cout = concentration in each inlet stream [= ] mg/L k = 1st order reaction rate constant (will be given) [=] 1/s t = time [=] sec, mi