What Element am I?
advertisement

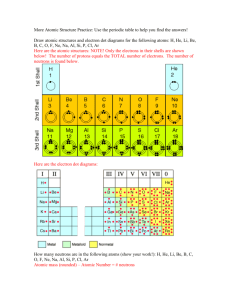
Particle Relative Mass (atomic mass units) Proton 1 +1 Nucleus Neutron 1 0 Nucleus -1 In space outside the nucleus Electron 1 2000 Relative Charge Position within Atoms Learning Objectives 1. Know where protons, neutrons and electrons are in an atom and their mass and charge. 2. Know how electron shells are filled. 3. Know what mass number and atomic number (proton number) are. 4. Know what an ion is and how to work out the ions charge. 5. Show respect for others by listening to their opinions and answers. Learning Outcomes (How you will demonstrate your learning!) All Pupils will: • Be able to label protons, neutrons and electrons on a picture of an atom. • Be able to work out atomic number for an atom. Most pupils will: • Be able to calculate the mass number for an atom. • Be able to draw an ion and work its charge. Some pupils will: • Be able to link the group number to the charge of an ion. Learning Outcomes (How you will demonstrate your learning!) • Fill out a table of information • Group activity to draw the atomic structure • Group activity – What element am I? • 3 Exam questions Atomic Structure Memory Game • Groups of 4. • 10 seconds to look at picture and then you have to draw what you remember! • Stick on the board. • Assess – Which is the best? Why? Build an Atom Activity What Element am I? Success Criteria 1. Draw and write in numbers the electron configuration. 2. Draw on the diagram the number of protons and neutrons. 3. Draw me the elements symbol, atomic number and mass number (as it would appear on the periodic Table) 4. Draw me the electron configuration for the ion it forms and tell me how many electrons it gains or loses. Information Card - A I have 12 electrons Information Card - B I have a mass number of 24. Information Card - C I have 3 electron shells. Information Card - D When I become an ion, I lose 2 electrons. What Element am I? Success Criteria 1. Draw and write in numbers the electron configuration. 2. Draw on the diagram the number of protons and neutrons. 3. Draw me the elements symbol, atomic number and mass number (as it would appear on the periodic Table) 4. Draw me the electron configuration for the ion it forms and tell me how many electrons it gains or loses. 3 Exam Questions Gimme 5! - 4 things you have learnt - 1 question or target for improvement