Scientific Measurement
advertisement

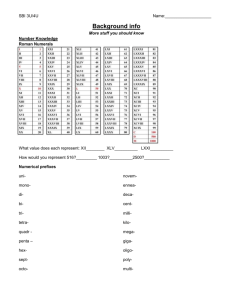
Scientific Measurement Add Corporate Logo Here David Chan Northwestern University Today (Day Eight) • • • • • • • • • (0-15) Discuss Test (0-15) (15-20) Discuss Test Resubmittal Process (15-20) Chapters 2+3 Syllabus (20-35) Qualitative vs. Quantitative (20-35) Accuracy vs. Precision (35-43) Class Jobs (0-15) Website intro, online grades (15-30) Scientific Notation (35-43) (30-43) Popsicle sticks match game Scientific Measurement • Questions to Ponder: – Why is measurement important? – What are the different types of measurement? – What is the history of measurement? – What do you know already? – What do you think we will focus on when studying measurement? Types of Measurements Types of Measurements 3 140 6 350 868 Types of Measurements Types of Measurement Types of Measurements • Qualitative Measurements – Descriptive, nonnumerical form • Quantitative Measurements – Definite form, numbers and units • Examples: – Basketball is brown – Diameter of basketball is 31 cm – Air pressure in the basketball is 12 psi. – Surface of basketball has indented seams. Accuracy, Precision, and Error • Accuracy – how close to actual or true value • Precision – how close measurements are to one another • Dartboard analogy • Examples – Precision or accuracy? • Multiple measurements • Correct • Repeatable • Reproducible Converting Measurements to Scientific Notation • Useful for large and small numbers • 2 numbers: Coefficient + 10 raised to power • Addition and Subtraction – Exponents must be same to add coefficients • Multiplication – Multiply coefficients and add the exponents • Division – Divide coefficients and subtract exponents • EE button on calculator • Practice Problems #4 Examples: – – – – – 91.4m 0.000000000154 m 6378000 m 0.000008 m 149600000000 m Today (Day Nine) • (0-15) Cat’s Meow – qualitative observations • (15-25) Significant Figures – Paper rulers • (25-35) SI Units – Length, Volume, Mass • (35-43) Discuss HW – clock buddies Significant Figures • Important when making measurements in experiments • All digits known with certainty plus one estimated or uncertain digit Rules/Examples • • • • • 1) 24.7m (3) 2) 7003 m (4) 3) 0.0071 m (2) 4) 25.0 (3) 5) 27,210 m (4) Rules Explained • • • • • 1) Nonzero digits are always significant (24.7m) 2) Zeroes in between are significant (7003m) 3) Left zeroes are not significant (0.0071 m) 4) Right zeroes are significant (25.0 m) 5) Right zeroes left of understood decimal are not sig.(27,210m) • 6) Unlimited sigfigs – Counting, exact quantities (60min=1hr) • Practice Problems #5,6 More Rules • Rounding: answers cannot be more precise than least precise measurement from which it was calculated • Rounding: greater than 5, less than 5 • Add/Subtract: round to least significant place • Mult/Divide: round to same # as least number of sigfigs • Practice Problems #11,12 SI units and Prefixes • International System of Units (SI) • SI Base Units (table 2.1) – Length, Mass, Temp, Time • SI Unit Prefixes – Table 3.3 • Derived Units – Density, volume Length • Basic SI unit = meter • Common: millimeters, centimeters, kilometers • Running races: 5K, 10K Volume • • • • • Derived unit: cubic _____meters CC’s = cubic centimeters 1 CC = 1ml 1 cubic decimeter = 1L Volumetric flasks, graduated cylinders Mass vs. Weight • Mass – amount of matter (1 kg) • Weight – force on mass by gravity (Physics) Today (Day 10) • • • • (0-5) Opener – sci notation (5-15) Density (15-20) Density Demo: How Sweet It is! (20-25) Temperature – Making thermometers? • (25-30) Percent Error – Activity • (30-35) Discuss HW – clock buddy Calculating Density • • • • Density = Mass/Volume Characteristic of substance Water displacement technique Temperature increases, V increases, Density decreases • Density of Common substances (table 2.8) • Practice Problems #23,24 • Examples: – Volume = 245 cm3 and mass of 612 g. Is it aluminum? – density of gold = 19.3 g/cm3. What is volume Specific Gravity • Comparison of density of substance with density of reference, usually at same temp. • Specific Gravity = density of substance/density of water Converting Celsius and Kelvin • Temperature – Determines direction of heat transfer • Thermal expansion – Generally expand as temp increases – Generally contracts as temp decreases • Celsius – history • Kelvin = -273.15 C = 0 K – Accounts for absolute zero (-273.15C) • Examples – 170 Celsius to Kelvin Percent Error, Experimental Error • Accepted value – correct value established by references • Experimental Value – measured in lab • Error: accepted – experimental • Percent Error: error/accepted x 100% • Example – Estimated volume = 200 mL – Accepted volume = 208 mL Calc Percent error Today (Day 11) • (0-10) Discuss HW – jigsaw (0-10) • (10-15) Class Jobs (10-15) • (15-20) How to Write Lab Reports, Sample Reports (15-20) • (20-30) PreLab: Observing Chemical Changes – 4 more safety rules • (30-43, 0-20) Lab: Observing Chemical Changes • (20-43) Prep Debate (20-43) Today (Day 12) • • • • • (0-5) discuss HW answers (5-10) Review Debate Reading Volumes Demo (10-40) Debate (40-43) Review for Quiz Today (Day 13) • • • • • • (0-10) Opener – sigfig/equipment activity (10-15) Review Debate/Labs – Peer review (15-20) Demo – Can You Pour a Gas? (20-35) Quiz (35-43) PreLab (0-43) Lab – Density POURING CARBON DIOXIDE. • Construct a series of three 5-cm steps that will fit into a wide mouth jar. Set a lighted candle on each step. Slowly pour carbon dioxide gas from an open container into the jar. Carbon dioxide is heavier than air. As it settles it extinguishes the candles one by one starting with the candle at the lowest level. There are many ways to generate carbon dioxide. Try mixing some vinegar with bicarbonate of soda. [HG] Today (Day 14) • • • • • • (0-10) discuss quiz (10-20) Discuss lab (20-35) Problem Solving Conversion Factors - list (35-43) Powers of Ten Website/Video Converting between units Dimensional Analysis – Converting Units • Write starting quantity • Write conversion factors – Numerator: What are you trying to convert into? – Denominator: What are you converting from? – Which one is bigger? = 1 – How many does it equal? • 1 m = 100 cm • 1000 m = 1 km List of Unit Prefixes • Powers of ten Java – http://micro.magnet.fsu.edu/primer/java/sci enceopticsu/powersof10/ • List of 20: – http://physics.nist.gov/cuu/Units/prefixes.ht ml • Common ones: – http://lamar.colostate.edu/~hillger/common. htm • Prefixes revisited – http://www.wordwizz.com/pages/10exp0.ht Practice – 0.044 km to meters – 860 mg to grams – 6.7 s to milliseconds Today (Day 15) • • • • (0-5) Opener (5-10) Discuss HW (10-15) Multistep, Complex Units (15-43) Stations Activity – Converting Units Multistep Problems • Use more than one conversion factor • Examples: – 261 nm to millimeters – 642 cg to kilograms Complex Units • Areas, Volumes, Speeds • Examples: – 55 mph to m/s – 1.54 kg/L to grams per cubic centimeter Today (Day 16) • Peer Review – Density Labs (20 min) • Review for Test (30-40 min) Today (Day 17) • Test (40-60 min) Intro to Labs • • • • Lab Report Format Lab Safety Lab Notebooks Lab Preps Lab #1: Chemical and Physical Changes • Objectives: – Chemical and physical properties – Chemical and physical changes – Become acquainted with lab facilities • Lab Techniques: – Filtration/Evaporation – Bunsen Burners – General Lab Procedures: • Supplies/Cleaning • Contamination of materials • Dissolving small quantities Exit Slips • On a separate sheet of paper labeled “Exit Slip”, please include the following: • 1 thing you learned today • 1 thing that still confuses you • Any other comments about the pace, methods, etc. • Thank you!