Chapter 1 Notes
advertisement

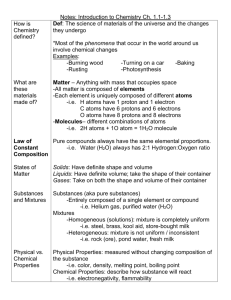
Chapter 1 How is chemistry applied to the matter that makes up the world around us? What does the science of chemistry study? How does matter change? Chemistry is the study of matter- its properties and changes it goes through Atoms are the most basic component of matter. A chemical is any substance that has definite composition ◦ Natural– water (H2O), carbon dioxide (CO2) ◦ Synthetic– plastics, teflon (tetrafluoroethylene) They’re everywhere! Some good, some bad Solid, liquid, gas, plasma Macroscopic – can see with your eye (macro = large) Microscopic – cannot be seen with the unaided eye (micro = small) solid liquid gas Figure 2 pg.6 – atoms in each state Solid Liquid Gas Fixed volume Fixed volume Indefinite volume Fixed shape Indefinite shape Indefinite shape Rigid structure, Loosely held organized, together, slide vibrate in place past each other Independent, weak attractions Physical Changes ◦ Identity of matter stays the same. Arrangement, shape, or location might change ◦ Examples: Cutting, dissolving, boiling, crushing Identity of matter changes. ◦ New substances are formed = chemical reaction ◦ Reactants – starting materials ◦ Products – ending materials ◦ Example: mercury(II) oxide → mercury + oxygen 1. 2. 3. 4. Evolution of a gas (bubbles, odor) Formation of a precipitate (solid formed from 2 solutions/liquids – looks cloudy) Release or absorption of energy (change in temperature or light given off) Color change How can we measure and describe matter? Matter – anything that has mass and takes up space (has volume) Volume – a measure of the size of an object ◦ Uses mL, L, cm3 as units (1mL = 1cm3) ◦ Measured using ruler or graduated cylinder Mass- measure of the quantity of matter in an object ◦ Uses: kg, g, mg Weight – measure of the gravitational force exerted on an object ◦ Can change with location Gravity changes ◦ Units: Newtons (N) Earth’s gravity is 9.8m/s2 The moon’s gravity is 1.6m/s2 Jupiter’s gravity is 25m/s2 A person weighs 150lb on Earth The same person weighs 25lb on the moon And he/she weighs 360lb on Jupiter These are NOT masses! Unit – quantity adopted as a standard of measurement Quantity – something that has size, magnitude or amount SI – Système International (international system) Not the same as the metric system! 1960 decided to unify measurement worldwide ◦ 7 base units All others are a combination of these Use prefixes to change magnitude Quantity Symbol Unit Abbreviation Length l Meter m Mass m Kilogram kg Time t Second s Temperature T Kelvin K Amount of a substance n Mole mol King Henry Died Belly-Up Drinking Chocolate Milk Under (a) Nice Picture. Prefix Abbreviation Power of Ten Meaning kilo- k 103 1000 hecto- h 102 100 deka- da 101 10 100 1 Base Unit deci- d 10-1 0.1 centi- c 10-2 0.01 milli- m 10-3 0.001 micro- 10-6 0.000 001 nano- n 10-9 0.000 000 001 pico- p 10-12 0.000 000 000 001 We can form other necessary units from the seven SI base units. ◦ Examples: Speed = distance/time = m/s Area = length ∙ width = m2 Volume = l ∙ w ∙ h = m3 Use mL in chemistry 1L = 1000 mL = 1000 cm3 (1mL = 1cm3) Why identify properties? ◦ They help us to learn more about substances. ◦ The more we know, the better we understand the substance. How are properties identified? ◦ By making observations about substances. Physical vs. Chemical ◦ Physical Property – a property that can be measured or observed without changing the chemical identity of the substance Examples: density, color, hardness, transparency (how well light is able to pass through), melting point ◦ Chemical Property – property that describes a substance’s ability to chemically interact with another substance Determined by trying to cause a chemical change Examples: flammability, reactivity with water, ability to be oxidized Density = mass divided by volume = m/V ◦ Units = g/mL or g/cm3 ◦ Found by measuring mass and volume Volume – use a ruler (l∙w∙h) or water displacement (graduated cylinder) ◦ Graph results show density by slope of line: rise = ∆y = mass run ∆x volume Can be used to identify substances. How do we classify matter? Atom – smallest unit of an element that maintains the properties of that element. ◦ Ex: copper, nickel, boron, etc. ◦ Almost all elements are made up of many individual atoms; but some are made of atoms that bond together: Br, I, N, Cl, H, O, F Pure substance – an element or compound that has definite chemical and physical properties (in other words has a chemical formula) ◦ Element – cannot be broken down into simpler substances; contain only 1 kind of atom ◦ Molecule – 2 or more atoms bonded together that retain the chemical & physical properties of that element or compound Elements: H2, O2, N2, Cl2, Br2, I2, F2 (same type of atoms bond together) Compounds: H2O, CO2, HCl (different types of atoms bond together) Allotrope: different form of an element *Examples: oxygen = O2, ozone = O3, diamond, graphite diamond graphite carbon Compound – substance made up of atoms of two or more different elements joined by chemical bonds. ◦ Can be further classified as ionic or covalent to be discussed in later chapters) ◦ Examples: salt (NaCl), caffeine (C8H10N4O2) Mixture – combination of two or more substances that are NOT chemically combined ◦ Can vary in composition and properties ◦ Homogeneous – uniform throughout, often called a solution Examples: completely dissolved salt water, tea, stainless steel, maple syrup ◦ Heterogeneous –not evenly mixed Examples: not completely dissolved salt water, orange juice with pulp, chocolate chip cookie, granite, salad Matter Mixture Homogeneous separate physically Pure Substance Heterogeneous Compound Element separate chemically Helpful hints for classification: ◦ Homogeneous (solutions): cannot see the individual substances that make it up (same uniform appearance & composition throughout). ◦ Heterogeneous: consist of visibly different substances or states. Difference #1: Properties of compounds do not reflect properties of elements making it up. ◦ Properties are different from the elements. Properties of mixtures do reflect properties of substances making them up. Difference #2: Compounds always have definite compositions of elements. Mixtures can have varying amounts of substances making them up.