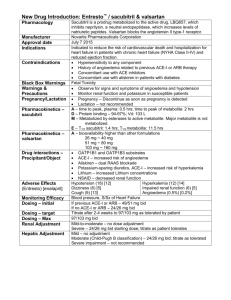
®
Entresto
(sacubitril &
valsartan)
Manufacturer: Novartis
Pharmaceuticals Corporation
FDA Approval Date: July 7 2015
Entresto® - sacubitril/valsartan
Clinical Application
• Indications:
• Neprilysin inhibitor and angiotensin II
receptor blocker combination to reduce
the risk of cardiovascular death and
hospitalization for heart failure in
patients with chronic heart failure (NYHA
Class II-IV) and reduced ejection fraction
• Place in therapy:
• Patient who have progressed in severity
of their heart failure on optimum ACE
inhibitor therapy
Entresto® - sacubitril/valsartan
Clinical Application
• Contraindications
• Hypersensitivity to any component
• History of angioedema related to
previous ACE inhibitor or ARB therapy
• Concomitant use with ACE inhibitors
• Concomitant use with aliskiren in
patients with diabetes
Entresto® - sacubitril/valsartan
Clinical Application
• Warnings & precautions
• Observe for signs and symptoms of
angioedema and hypotension
• Monitor renal function and potassium in
susceptible patients
Entresto® - sacubitril/valsartan
Clinical Application
• Pregnancy – contraindicated
• Lactation – not recommended
Entresto® - sacubitril/valsartan
Drug Facts
• Pharmacology:
• Sacubitril – prodrug metabolized to
active metabolite (LBQ657), which
inhibits neprilysin
• Neprilisyn – neutral endopeptidase
• Leads to increase in level of peptides,
including natriuretic peptides
• Valsartan – blocks the angiotensin II
type-1 (AT1) receptor
Entresto® - sacubitril/valsartan
Drug Facts
• Pharmacokinetics:
A
D
M
E
Time to peak: 0.5 hrs
Time to peak of metabolite: 2 hrs
Protein binding – 94-97%
Vd: 103 L
Metabolized by esterases to active
metabolite
Major metabolite is not metabolized
T1/2 sacubitril: 1.4 hrs
T1/2 metabolite: 11.5 hrs
Entresto® - sacubitril/valsartan
Drug Interactions
Precipitant
Entresto*
• Drug
Object
Nature of interaction
Decrease
AUC and Cmax
Furosemide,
Interactions
– Object
Drugs:
levonorgestrel,
• Object
drugs are affected by “the
HCTZ, metformin
drug”
Entresto*reviewed
Atorvastatin
Increase AUC and Cmax
• List (
##%) if available
Entresto
ACE-I
Increased risk of
angioedema
• Ex: ASA (100%)
Entresto
Aliskiren
Dual RAAS blockade
Entresto
Potassium-sparing
Increased risk of
diuretics, ACE-I
hyperkalemia
NSAID
Entresto
Decreased renal function
Entresto
Lithium
Increased concentrations
Entresto® - sacubitril/valsartan
Adverse Effects
Side effect
Angioedema
Hypotension
Impaired renal function
Hyperkalemia
Cough
Entresto
0.5%
18%
6%
Enalapril
0.2%
12%
5%
12%
9%
14%
13%
Entresto® - sacubitril/valsartan
Monitoring Parameters
• Efficacy Monitoring:
• Blood pressure at each visit and dose
titration
• Toxicity Monitoring:
• Serum electrolytes (K+)
• SCr
Entresto® - sacubitril/valsartan
Prescription Information
• Dosing: initial
• Previous ACE-I or ARB – 49/51 mg bid
• No ACE-I or ARB or low doses – 24/26 mg
bid
• Dosing: target
• Titrate after 2-4 weeks to 97/103 mg bid
as tolerated by the patient
Entresto® - sacubitril/valsartan
Prescription Information
• Renal impairment
• Mild-moderate – no dose adjustment
• Severe – 24/26 mg bid (initial)
• Hepatic impairment
• Mild – no adjustment
• Moderate (Child-Pugh B) – 24/26 mg bid
(initial)
• Severe impairment – not recommended
Entresto® - sacubitril/valsartan
Prescription Information
• If switching from ACE-I to Entresto, 36
hour washout period is recommended
• Cost – Source: NY Times; Accessed
8/21/15
• $4,500/year
• Novartis offers free 30-day supply and
$10 co-pay cards
Entresto® - sacubitril/valsartan
Literature Review
PARADIGM-HF
• Purpose: To compare the combination
of sacubitril/valsartan with enalapril in
patients who have HFrEF
• Design: randomized, double-blind,
phase 3 trial
• 1043 sites in 47 countries
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
Inclusion Criteria
• Inclusion
Criteria
Exclusion Criteria
• Age >18 years
• Symptomatic hypotension
•• NYHA
Exclusion
class II-IVCriteria
• SBP <100 mg
• Ejection fraction <40%
(amended to <35%)
• eGFR <30 ml/min/1.73 m2
or eGFR >25%
• BNP >150 pg/mL or proBNP >600 pg/mL
• Serum K+ >5.2 mEq/L
• Treatment with ACE-I or
ARB
• Hx of angioedema or
unacceptable side effects
during receipt of ACE-I or
ARB
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
• Intervention: LCZ696 200 mg bid vs.
enalapril 10 mg bid
• Primary endpoint: composite of death
from cardiovascular causes or a first
hospitalization for HF
• Secondary endpoint:
• Time to death from any cause
• Change from baseline to 8 months in clinical
summary score (KCCQ)
• Time to new onset atrial fibrillation
• Time to first occurrence of a decline in renal
function
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
• Baseline characteristics
Entresto (N=4187)
Enalapril (N=4212)
63.8
63.8
Female
879 (21.0%)
953 (22.6%)
White
2763 (66.0%)
2781 (66.0%)
Medical History:
• HTN
• Afib
• Hospitalization for HF
• MI
• Pretrial use of ACE-I
• Pretrial use of ARB
2969 (70.9%)
1517 (36.2%)
2607 (62.3%)
1818 (43.4%)
3266 (78.0%)
929 (22.2%)
2971 (70.5)
1574 (37.4%)
2667 (63.3%)
1816 (43.1%)
3266 (77.5%)
963 (22.9%)
Treatment at randomization:
• Diuretic
• Beta-blocker
• Mineralocorticoid antagonist
3363 (80.3%)
3899 (93.1%)
2271 (54.2%)
3375 (80.1%)
3912 (92.9%)
2400 (57.0%)
NYHA class II
2998 (71.6%)
2921 (69.3%)
Age
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
• Results
Entresto
(N=4187)
Enalapril
(N=4212)
HR or Difference
(95% CI)
P-value
Composite outcome
914 (21.8)
1117 (26.5) 0.80 (0.73-0.87)
<0.001
Death from cardiovascular causes
558 (13.3)
693 (16.5)
0.80 (0.71-0.89)
<0.001
1st hospitalization for
worsening HF
537 (12.8)
658 (15.6)
0.79 (0.71-0.89)
<0.001
Death from any cause
711 (17.0)
835 (19.8)
0.84 (0.76-0.93)
<0.001
Change in KCCQ
clinical summary
score at 8 mo
-2.99
-4.63
1.64 (0.63-2.65)
0.001
New-onset afib
84 (3.1)
83 (3.1)
0.97 (0.72-1.31)
0.83
Decline in renal fxn
94 (2.2)
108 (2.6)
0.86 (0.65-1.13)
0.28
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
• Safety endpoints
Entresto
(N=4187)
Enalapril
(N=4212)
P-value
Symptomatic
588 (14.0)
388 (9.2)
<0.001
Symptomatic w/ SBP <90
mmHg
112 (2.7)
59 (1.4)
<0.001
SCr > 2.5 mg/dl
139 (3.3)
188 (4.5)
0.007
Serum K >6.0 mmol/L
181 (4.3)
236 (5.6)
0.007
Cough
474 (11.3)
601 (14.3)
<0.001
Hypotension
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto® - sacubitril/valsartan
Literature Review
• Conclusions
• Entresto’s dual inhibition was more
effective in reducing the risk of death
from cardiovascular causes or
hospitalization for HF than ACE inhibition
with enalapril
• The only significant side effect was
symptomatic hypotension, though this
did not increase the rate of
discontinuation
McMurray JJV, et al. N Engl J Med. 2014;371(11): 993-1004
Entresto™ - sacubitril/valsartan
Summary
• Entresto™ inhibits neprilysin and angiotensin
receptors
• Indicated to reduce the risk of cardiovascular
death and hospitalization for heart failure in
patients with chronic heart failure (NYHA Class
II-IV) and reduced ejection fraction
• Initial dose is based on receipt of ACE-I or ARB
therapy prior to initiation
• Avoid use in combination with an ACE-I or in
patients with a history of angioedema
• Most common side effect is hypotension
Entresto® - sacubitril/valsartan
References
1.
Entresto [sacubitril and valsartan] package
insert. Novartis Pharmaceutical Corporation. July
2015.
2.
McMurray, J, et al. PARADIGM-HF Study. New
England Journal of Medicine. 2014;371;11:9931004.
3.
Pollack, A. The New York Times Website. F.D.A.
Approves Heart Drug Entresto Said to Cut Death
Risk by 20%.
http://www.nytimes.com/2015/07/08/business/inte
rnational/fda-approves-heart-drug-entresto-afterpromising-trial-results.html. Published July 7,
2015. Accessed August 21, 2015.
 0
0