Entresto Drug Information: Sacubitril/Valsartan Details
advertisement

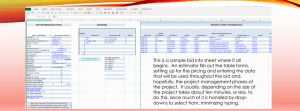
New Drug Introduction: Entresto™ / sacubitril & valsartan Pharmacology Manufacturer Approval date Indications Contraindications Black Box Warnings Warnings & Precautions Pregnancy/Lactation Pharmacokinetics – sacubitril Pharmacokinetics – valsartan Drug interactions – Precipitant/Object Adverse Effects (Entresto) [enalapril] Monitoring Efficacy Dosing – initial Dosing – target Dosing – Max Renal Adjustment Hepatic Adjustment Sacubitril is a prodrug metabolized to the active drug, LBQ657, which inhibits neprilysin, a neutral endopeptidase, which increases levels of natriuretic peptides. Valsartan blocks the angiotensin II type-1 receptor. Novartis Pharmaceuticals Corporation July 7 2015 Indicated to reduce the risk of cardiovascular death and hospitalization for heart failure in patients with chronic heart failure (NYHA Class II-IV) and reduced ejection fraction Hypersensitivity to any component History of angioedema related to previous ACE-I or ARB therapy Concomitant use with ACE inhibitors Concomitant use with aliskiren in patients with diabetes Fetal Toxicity Observe for signs and symptoms of angioedema and hypotension Monitor renal function and potassium in susceptible patients Pregnancy – Discontinue as soon as pregnancy is detected Lactation – not recommended A – time to peak, plasma: 0.5 hrs; time to peak of metabolite: 2 hrs D – Protein binding – 94-97%; Vd: 103 L M – Metabolized by esterases to active metabolite. Major metabolite is not metabolized. E – T1/2 sacubitril: 1.4 hrs; T1/2 metabolite: 11.5 hrs A – bioavailability higher than other formulations 26 mg ~ 40 mg 51 mg ~ 80 mg 103 mg ~ 160 mg OATP1B1 and OATP1B3 substrates ACE-I – increased risk of angioedema Aliskiren – dual RAAS blockade Potassium-sparing diuretics, ACE-I – increased risk of hyperkalemia Lithium – increased Lithium concentrations NSAID – decreased renal function Hypotension (18) [12] Hyperkalemia (12) [14] Dizziness (6) [5] Impaired renal function (6) [5] Cough (9) [13] Angioedema (0.5%) [0.2%] Blood pressure, S/Sx of Heart Failure If previous ACE-I or ARB – 49/51 mg bid If no ACE-I or ARB – 24/26 mg bid Titrate after 2-4 weeks to 97/103 mg as tolerated by patient 97/103 mg bid Mild-to-moderate – no dose adjustment Severe – 24/26 mg bid starting dose; titrate as patient tolerates Mild – no adjustment Moderate (Child-Pugh B classification) – 24/26 mg bid; titrate as tolerated Severe impairment – not recommended Cost: Source: NY Times – Accessed 08/21/15 Dose(s) Brand – Generic 49/51 mg Entresto™ – sacubitril & valsartan $ (year) ~$4,500 Summary Entresto™, sacubitril/valsartan, is a novel combination agent that lowers blood pressure and reduces the risk of cardiovascular death and hospitalization by inhibiting neprilysin and angiotensin II type-1 receptor, indicated for patients with chronic heart failure (NYHA Class II-IV) and reduced ejection fraction. Black box warning for fetal toxicity. Avoid use in combination with an ACE-I or in patients with a history of angioedema. Interactions include OATP substrates (lithium), RAAS agents, NSAIDs, and ACE-I. Blood pressure should be monitored, as hypotension is the most common side effect of sacubitril/valsartan. References: 1. Entresto™ [sacubitril and valsartan] package insert. Novartis Pharmaceutical Corporation. July 2015. 2. McMurray, J, et al. PARADIGM-HF Study. New England Journal of Medicine. 2014;371;11:993-1004. 3. Pollack, A. The New York Times Website. F.D.A. Approves Heart Drug Entresto Said to Cut Death Risk by 20%. http://www.nytimes.com/2015/07/08/business/international/fda-approvesheart-drug-entresto-after-promising-trial-results.html. Published July 7, 2015. Accessed August 21, 2015. Date Prepared: August 24, 2015 Editor: Peter G. Koval, Pharm.D., BCPS Author: Leslie Perry, Pharm.D. Candidate, UNC Eshelman School of Pharmacy