Chapter 7 - Review
advertisement

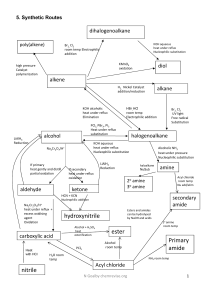
Chapter 7 – Review “Functional Groups and Organic Chemistry” Chapter 7 - Review What is the name of the functional group in the following? O CH3 – O – C – CH2 – CH3 Chapter 7 - Review What is the name of the following compound? CH3 CH3 – C – Br CH3 Chapter 7 - Review What type of compound is: CH3 – O – CH2 – CH2 – CH3 and, O CH3 – C – CH2 – CH3 Chapter 7 - Review Which of the following halocarbons has the higher boiling point: a) 1-chloropropane, or b) 1,2,3-trichloropropane? Chapter 7 - Review Name this compound: Cl Cl – C – Cl H Chapter 7 - Review Give the name for: OH CH3-CH2-CH2-CH2-CH-CH3 Chapter 7 - Review Name the following compound: CH3-CH2-CH2-CH2-O-C6H5 and, CH3-CH2-O-CH2-CH2CH3 Chapter 7 - Review What type of alcohol is: OH CH3-CH2-CH-CH3 Chapter 7 - Review Which of the following would you expect to be more soluble in water: a) CH3CH2Cl or, b) CH3CH2CH2OH Phenols are characterized by having a(n) _______. Chapter 7 - Review What type of alcohol is: OH CH3-CH2-C-CH3 H Chapter 7 - Review Which is true of isopropyl alcohol: a) it is insoluble in water, or b) it has a relatively high boiling point? Based on your knowledge of intermolecular forces, which of these would you expect to have the higher boiling point: a) acetone, or b) 1propanol? Chapter 7 - Review In an addition reaction, which bond of the reactant is broken? What type of compound is the following: C-C-C-O-C-C-C Which has the lower boiling point: a) 4-octanol, or b) diethyl ether? Chapter 7 - Review What is the name of the following compound: O C6H5 – C – H Chapter 7 - Review What is the name of the following compound: O CH3 – C – CH2 – CH2 – CH3 Chapter 7 - Review Name this compound: O CH3CCH2CH3 Chapter 7 - Review Which of the following has the higher boiling point: a) pentane, or b) 2-pentanone? What is a carboxyl group? Name the following: O CH3 – C – OH Chapter 7 - Review Carboxylic acids with long hydrocarbon chains are called _______. Which of the following compounds is more soluble in water: a) propanoic acid, or b) propane A carboxylic acid with 6 straight carbons would be named ____. Chapter 7 - Review What type of compound is: O C–C–C–C–C–O–C–C Which of the following has the higher boiling point: a) butane, or b) butanoic acid Chapter 7 - Review Esters contribute which property to fruits? Which compound reacts with a carboxylic acid to form an ester?