Naming Compounds and Writing Formulas

Varying the Number of Oxygens in a Polyatomic Ion
Professional Hillbilly Engineer Exam
Calculate the smallest limb diameter on a persimmon tree that will support a 10-pound possum.
Naming Various Polyatomic Ions:
At the conclusion of our time together, you should be able to:
1. Name 3 different polyatomic ions by adding or subtracting oxygens
2. Use these various ions in a compound and name them or write them
6 Types Of Chemical Compounds
•Binary ionic - metal ion – nonmetal ion
•Binary molecular - two nonmetals
•Ternary ionic - at least one ion is a polyatomic ion
•Binary acid
•Ternary acid
•Base
- H – nonmetal
- H – Polyatomic ion
- metal ion – OH
Things You Can Learn
From Your Dog!!
When your loved one comes home, run to greet him/her.
Eat with gusto to show your appreciation for the meal.
New Element On The Periodic Table
Element
: Woman
Symbol
:
Atomic Mass
:
Xx
120 (More Or Less)
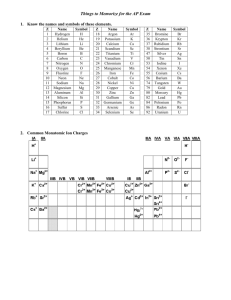
Let’s learn some variations on the polyatomic ions…
•The standard ions that you have on your handout that end in –ate can be changed by adding or subtracting oxygens to form 3 more ions.
Let’s see what happens when we add or subtract oxygens…
Name Ion
BrO
4
1-
BrO
3
1-
BrO
2
1-
BrO 1-
Perbromate
Bromate
Bromite
Hypobromite
Let’s try it again…
Ion
SO
5
2-
SO
4
2-
SO
3
2-
SO
2
2-
Name
Persulfate
Sulfate
Sulfite
Hyposulfite
New Element On The Periodic Table
(cont.)
Physical Properties of Xx
:
Generally round in form. Boils at nothing and may freeze anytime. Melts whenever treated properly. Very bitter if not used well.
Chemical Properties
:
Very active. Possesses strong affinity to gold, silver, platinum, and precious stones. Violent when left alone.
Able to absorb great amount of exotic food. Turns slightly green when placed beside a better specimen.
Ages rapidly.
New Element On The Periodic Table
(cont.)
Usage of Xx
:
Highly ornamental. An extremely good catalyst for disintegration of wealth. Probably the single most powerful income reducing agent known.
Caution
:
Highly explosive in inexperienced hands.
Naming Compounds with Polyatomic Ions as
Cations and Anions
( NH
4
)
3
PO
3
Ammonium phosphite
Name the first ion.
Name the second ion.
Cu
2
SO
5
Copper (I) persulfate
Name the second ion.
Name the first ion.
You should realize that you need a Roman Numeral since copper is a transition metal.
The Roman Numeral is the same as the charge.
What is the charge?
The charge of persulfate is -2.
Since there are two coppers, the charge of the copper must be +1.
Euphemisms in Science
We all know that some politicians and government spokesmen use certain euphemistic phrases to give an aura of respectability to descriptions of events or actions which would be offensive when expressed in plain English. The following is a list of
Euphemisms in Science and their translations into plain English.
“It has long been known...”
I haven't bothered to look up any references!
Aluminum nitrate First, you can tell from the name
Al 3+
Al(NO
NO
2
)
3
2
that there is a polyatomic ion present (nitrite). All binary ionic compounds have suffixes of
–ide
.
Write the formula/symbol and charge of each ion.
Balance the charges by supplying subscripts.
Since Al is +3 and NO
2 to balance one Al 3+ is -1, you need a total of 3NO
2
-
Since you will need a subscript of 3 for NO to put this in parentheses with the 3 outside.
2
, you need
Lead (IV) peracetate Write the formula/symbol and charge of each ion.
Pb 4+ C
2
H
3
O
3
-
Balance the charges by supplying
Pb(C
2
H
3
O
3
)
4 subscripts. Since Pb is +4 and C
2
H
3
O
3
1, you will need 4 C
2
H
3
O
3
-1 to balance is -
1Pb +4
Calcium phosphite
Ca 2+ PO
3
3-
Ca
3
(PO
3
)
2
Write the formula/symbol and charge of each ion.
Balance the charges by supplying subscripts. Since Ca is +2 and PO
3 is -3, you will need 3 Ca+2 to balance 2 PO
3
-3
Strategy For Naming Ternary Ionic
Compounds.
Naming Various Polyatomic Ions:
Let’s see if you can:
1. Name 3 different polyatomic ions by adding or subtracting oxygens
2. Use these various ions in a compound and name them or write them
Exit Quiz
Al 3+ combines with sulfate (SO
3 sulfite.
) 2– to make aluminum
Write the chemical formula for aluminum sulfate.
Exit Quiz Answer
Al
3+
2
(SO
3
)
2-
3
Let’s Try Another One:
Manganese (IV) hypophosphite
Mn
4+
3
3-
(PO
2
)
4
Let’s Try One More:
Manganese (IV) carbonite
Mn
4+
2
(CO
2-
2
)
4
Mn(CO
2
)
2
Chemical Compound Quiz
Hypochlorous acid helps get the laundry white and bright. What is the common name?
Bleach
Exit Quiz
Formula
LiOH
Mn(OH)
3
Ca(OH)
2
Cu(OH)
2
KOH
Name
Lithium hydroxide
Manganese (III) hydroxide
Calcium hydroxide
Copper (II) hydroxide
Potassium hydroxide
Exit Quiz
Formula
Na
2
SO
5
Na
2
SO
4
Na
2
SO
3
Na
2
SO
2
Name
Sodium persulfate
Sodium sulfate
Sodium persulfite
Sodium hyposulfite
Exit Quiz
Formula
FeCO
4
FeCO
3
FeCO
2
FeC
Name
Iron (II) percarbonate
Iron (II) carbonate
Iron (II) carbonite
Iron (IV) carbide
Redneck News:
Down home in Hicksville, folks now go to some movies in groups of 18 or more.
They were told 17 and under are not admitted.