Name: Hour: ______ Naming Compounds and Writing Formulas
advertisement
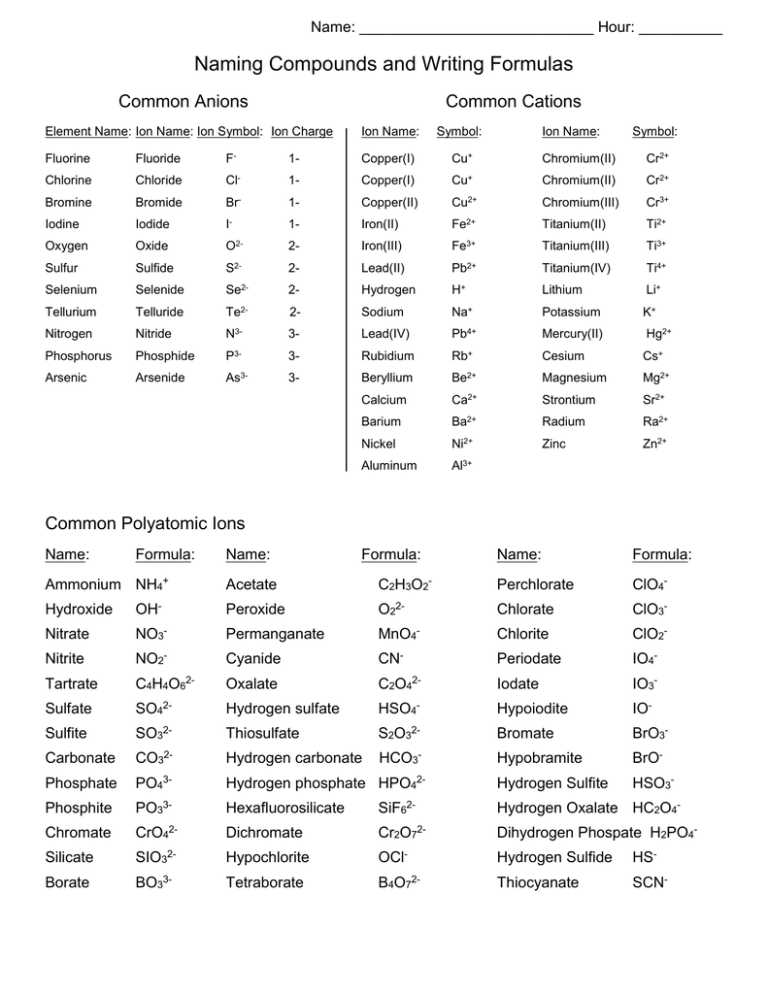
Name: ____________________________ Hour: __________ Naming Compounds and Writing Formulas Common Anions Common Cations Element Name: Ion Name: Ion Symbol: Ion Charge Ion Name: Symbol: Ion Name: Symbol: Fluorine Fluoride F- 1- Copper(I) Cu+ Chromium(II) Cr2+ Chlorine Chloride Cl- 1- Copper(I) Cu+ Chromium(II) Cr2+ Bromine Bromide Br- 1- Copper(II) Cu2+ Chromium(III) Cr3+ Iodine Iodide I- 1- Iron(II) Fe2+ Titanium(II) Ti2+ Oxygen Oxide O2- 2- Iron(III) Fe3+ Titanium(III) Ti3+ Sulfur Sulfide S2- 2- Lead(II) Pb2+ Titanium(IV) Ti4+ Selenium Selenide Se2- 2- Hydrogen H+ Lithium Li+ Tellurium Telluride Te2- 2- Sodium Na+ Potassium K+ Nitrogen Nitride N3- 3- Lead(IV) Pb4+ Mercury(II) Hg2+ Phosphorus Phosphide P3- 3- Rubidium Rb+ Cesium Cs+ Arsenic Arsenide As3- 3- Beryllium Be2+ Magnesium Mg2+ Calcium Ca2+ Strontium Sr2+ Barium Ba2+ Radium Ra2+ Nickel Ni2+ Zinc Zn2+ Aluminum Al3+ Common Polyatomic Ions Name: Formula: Ammonium NH4+ Name: Formula: Name: Formula: Acetate C2H3O2- Perchlorate ClO4- Hydroxide OH- Peroxide O22- Chlorate ClO3- Nitrate NO3- Permanganate MnO4- Chlorite ClO2- Nitrite NO2- Cyanide CN- Periodate IO4- Tartrate C4H4O62- Oxalate C2O42- Iodate IO3- Sulfate SO42- Hydrogen sulfate HSO4- Hypoiodite IO- Sulfite SO32- Thiosulfate S2O32- Bromate BrO3- Carbonate CO32- Hydrogen carbonate HCO3- Hypobramite BrO- Phosphate PO43- Hydrogen phosphate HPO42- Hydrogen Sulfite HSO3- Phosphite PO33- Hexafluorosilicate SiF62- Hydrogen Oxalate HC2O4- Chromate CrO42- Dichromate Cr2O72- Dihydrogen Phospate H2PO4- Silicate SIO32- Hypochlorite OCl- Hydrogen Sulfide HS- Borate BO33- Tetraborate B4O72- Thiocyanate SCN- Writing Formulas for Ionic Compounds 1. Place the symbol for the cation first, followed by the anion. List the symbols and charges for the cation and anion. 2. Use subscripts to show the ratio of the ions in the compound. The total charge on the cations and anions must add up to zero. Use the cross over method: Fe3+ O2+ 3. Write the formula Fe2O3 Naming Molecular Compounds 1. The name of the most metallic element appears first. The most metallic elements are further to the left and nearest to the bottom on the periodic table. 2. Use prefixes to describe the number of atoms of each element. Writing Molecular Formulas 1. Write the symbols for the elements in the order they appear in the name. 2. The prefixes indicate the number of atoms of each element appear in the molecule. The prefixes appear as subscripts in the formula. 3. If there is no prefix for an element, there is only one atom of that element in the molecule. OF2 = oxygen difluoride N2O5 = dinitrogen pentoxide Prefixes for Naming Compounds Prefix: Number of Atoms: mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10









