fs_ecoli
advertisement
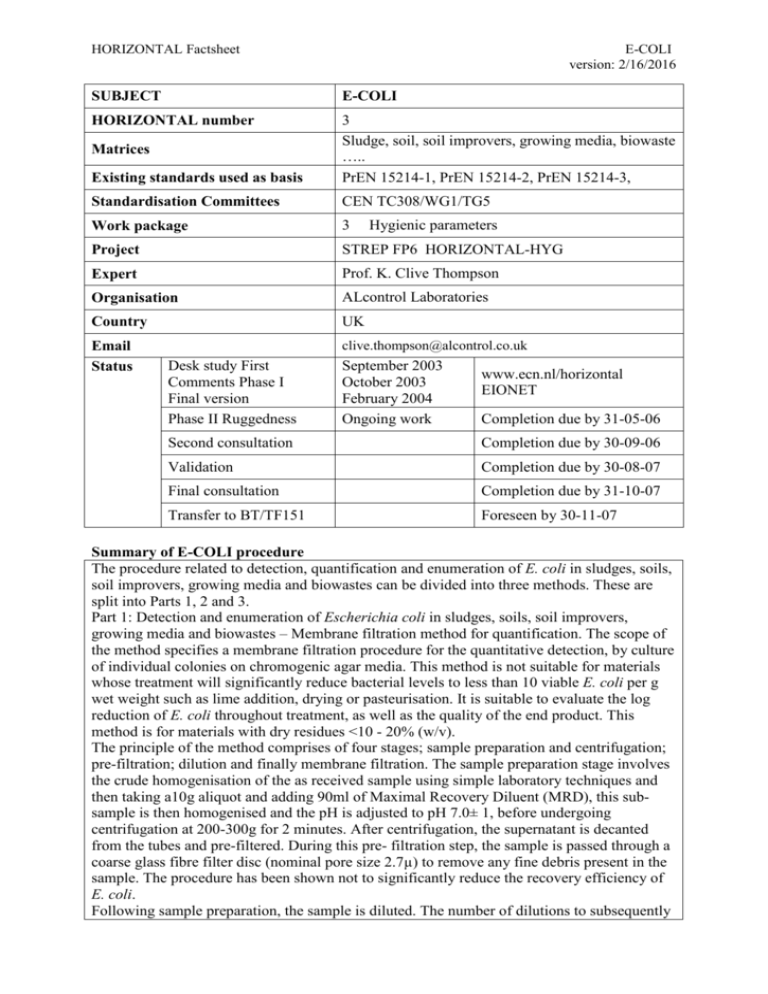
HORIZONTAL Factsheet E-COLI version: 2/16/2016 SUBJECT E-COLI HORIZONTAL number Existing standards used as basis 3 Sludge, soil, soil improvers, growing media, biowaste ….. PrEN 15214-1, PrEN 15214-2, PrEN 15214-3, Standardisation Committees CEN TC308/WG1/TG5 Work package 3 Project STREP FP6 HORIZONTAL-HYG Expert Prof. K. Clive Thompson Organisation ALcontrol Laboratories Country UK Email Status clive.thompson@alcontrol.co.uk Matrices Desk study First Comments Phase I Final version Phase II Ruggedness Hygienic parameters September 2003 October 2003 February 2004 Ongoing work www.ecn.nl/horizontal EIONET Completion due by 31-05-06 Second consultation Completion due by 30-09-06 Validation Completion due by 30-08-07 Final consultation Completion due by 31-10-07 Transfer to BT/TF151 Foreseen by 30-11-07 Summary of E-COLI procedure The procedure related to detection, quantification and enumeration of E. coli in sludges, soils, soil improvers, growing media and biowastes can be divided into three methods. These are split into Parts 1, 2 and 3. Part 1: Detection and enumeration of Escherichia coli in sludges, soils, soil improvers, growing media and biowastes – Membrane filtration method for quantification. The scope of the method specifies a membrane filtration procedure for the quantitative detection, by culture of individual colonies on chromogenic agar media. This method is not suitable for materials whose treatment will significantly reduce bacterial levels to less than 10 viable E. coli per g wet weight such as lime addition, drying or pasteurisation. It is suitable to evaluate the log reduction of E. coli throughout treatment, as well as the quality of the end product. This method is for materials with dry residues <10 - 20% (w/v). The principle of the method comprises of four stages; sample preparation and centrifugation; pre-filtration; dilution and finally membrane filtration. The sample preparation stage involves the crude homogenisation of the as received sample using simple laboratory techniques and then taking a10g aliquot and adding 90ml of Maximal Recovery Diluent (MRD), this subsample is then homogenised and the pH is adjusted to pH 7.0± 1, before undergoing centrifugation at 200-300g for 2 minutes. After centrifugation, the supernatant is decanted from the tubes and pre-filtered. During this pre- filtration step, the sample is passed through a coarse glass fibre filter disc (nominal pore size 2.7µ) to remove any fine debris present in the sample. The procedure has been shown not to significantly reduce the recovery efficiency of E. coli. Following sample preparation, the sample is diluted. The number of dilutions to subsequently HORIZONTAL Factsheet E-COLI version: 2/16/2016 filter will vary according to the presumed level of contamination of the material to be tested. Typically, the sample solution should be serially diluted 10-1 – 10-3 using MRD, this will permit enumeration of up to 104 E. coli per g wet weight sludge. Higher bacteria levels (untreated sludge) will require additional dilutions of the filtrate to 10-7. The diluted sludge sample is subsequently filtered, the membrane is recovered aseptically and incubated on membrane lactose glucuronide agar, initially at 30 ± 1°C for 4 ± 0.5h to aid resuscitation of stressed E. coli and then the temperature is increased to 44 ± 1°C for 14 ± 2h. The presence of E. coli is indicated by green colonies resulting from the hydrolysis of BCIG. The method has a limit of detection of approximately 10 cfu g-1 wet weight sludge, dependant on the solids content which at high concentrations (>20% (w/v)) will restrict filtration of the sample volume through the membrane, if not first diluted sufficiently. Samples can be confirmed through a biochemical test (API20E) if required. Part 2: Detection and enumeration of Escherichia coli from sludges, soils, soil improvers, growing media and biowastes- Miniaturised method (MPN) in liquid medium. The scope of the method is to detect E. coli in accordance with the requirements of the European Sewage Sludge Draft Regulation. The principle of the miniaturised method can be split into four stages; sample preparation; inoculation; reading the microtitre plate and finally calculation of the result. The as received sample is crudely homogenised using simple laboratory techniques and is then prepared by suspending a sample aliquot equivalent to 10g (dry matter) with tryptone diluent in a final volume of 100ml. This is then homogenised for a period of 1 minute in a mixer and then subsequently analysed without delay. The microtitre plate is inoculated with 2ml of primary suspension mixed with 18ml of special diluent. The dilutions are prepared from this solution from a starting concentration of 10-1 down to a final dilution of 10-6. Once the microtitre plate is inoculated it is incubated for between 36-72 h at 44 ±0.5 °C The presence of E. coli under Ultra Violet light at 366nm is indicated by a blue florescence resulting from hydrolysis of MUG. The results are expressed as Most Probable Number per g dry weight sludge. Part 3: Detection and enumeration of Escherichia coli from sludges, soils, soil improvers, growing media and biowastes – macromethod MPN in liquid medium. The scope of the method described in this standard to detect Escherichia coli in sewage sludge in accordance with the requirements of the European Sewage Sludge Draft Regulation. The principle of the macromethod MPN in liquid medium can be split into four parts; sample preparation; dilution; inoculation and finally interpretation and reading. The preparation of the primary suspension is achieved by placing 20 g (wet weight) of the crudely homogenised as received sample (sludge/compost etc.) obtained by using simple laboratory techniques, into 180 ml 0.9% m/V sterile NaCl solution. This is then shaken at a minimum of 150 rpm for up to 20 h at 6 ± 2°C. For relatively homogeneous samples (e.g. mesophilic digested sludges), only a 30 min shaking is necessary for the homogenisation step. A 20 hour shaking is only necessary for dry hydrophobic and/or inhomogeneous samples. For disinfectant (e.g. lime, peracetic acid) treated sludges, a suitable pre-treatment for neutralising the disinfecting agent and suitable pH adjustment is required. The samples are prepared for analysis using a serial tenfold dilution (1 ml of primary suspension + 9 ml 0.9% m/V sterile NaCl solution) up to 10-7. From each dilution step is transferred 1 ml per tube; into 3 tubes each containing 5 ml MUG Fluorocult lauryl sulphate broth. The samples are then incubated for 40 ± 4 h at 44 ± 1°C. The confirmation of E. coli is HORIZONTAL Factsheet E-COLI version: 2/16/2016 achieved through the adding 0.5 ml NaOH 1N to each tube, and observing for fluorescence with 366 nm UV-light irradiation. The result per gram of sludge is then calculated according to E. coli / g (wet weight) = 10 x E. / ml of Primary Suspension. Placement of E-COLI method in overall structure HORIZONTAL SLUDGE, SOIL, TREATED BIOWASTE AND RELATED WASTES SAMPLING IN-SITU ,TRANSPORT, STORAGE SAMPLE PRETREATMENT - INORG INORGANIC CONSTITUENTS PHYSICAL PROPERTIES SAMPLE PRETREATMENT - ORG & BIOL LEACHING ORGANIC PARAMETERS BIOLOGICAL PARAMETERS SAMPLE PRETREATMENT - HYG HYGIENIC PARAMETERS PAH pH Bulk density Characterisation leaching tests Impurities (e.g glass, plastic) E. Coli Stability (bio) Salmonella Germinating weed seeds Clostridium Phytotoxicity Enterococci AOX Electrical conductivity Dry matter Compliance leaching tests PCB Organic matter* Solidity LAS, NP,NPE Nutrients N, P, K* Thixotropic behaviour DEPH, DBP Trace elements solid * Trace elements determination* Piling behaviour Helminth ova BFR Flowability Rapid methods PCCD/PCDF Soluble P Pharmaceuticals Virusses and bacteriophages Plant Pathogens Position of E-COLI method in the sequence of steps from sampling to reporting Sampling Taking of plan sample Transport & Preparation storage of test portion Extraction/ Leaching Pre-treatment Detection & Summary Quantification, reports Enumeration 1)Sample conservation, storage and transport are issues that generally are covered in as the final step in sampling standards 2) Preparation of the test portion from a laboratory sample is sometimes part of the next step (digestion or analysis preparation) or can be covered in a separate standard 3) Summary of test results is a concise summary of the relevant data for the end-user, which can be merged in an overall test report covering all aspects relevant for the given situation. 4) After defining sampling strategy based on the question to be answered either one of the sampling standards will generally apply. All the following steps will be the same for either route. 5) It is at present not clear where extraction of organic parameters begins and pre-treatment ends. It seems that pre-treatment now includes the extraction step. Major issues of discussion HORIZONTAL Factsheet .................................... E-COLI version: 2/16/2016








