Unit 4: Gases
advertisement

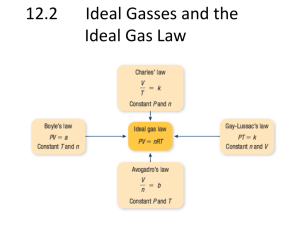
Unit 4: Gases Kinetic Molecular Theory & Pressure States of Matter Solids: Are they compressible? No. Liquids: Are they compressible? Essentially not. Are Gases compressible? Yes. Look at particle distribution in each physical state. In which state are particles furthest apart? In gas phase. Kinetic Molecular Theory of Gases Assumptions: 1. Volume of individual gas particles (molecules or atoms) is negligible with respect to the volume that they occupy. i.e. Most of a gas is empty space; particles of gas are very far apart. 2. No attractive or repulsive forces between gas particles. 3. Gas particles are in rapid, random, straight line motion in all 3-D. 4. Collisions of gas particles with other gas particles or with container walls are perfectly elastic. i.e. No loss of kinetic energy on collision 5. Average KE of a gas α Temperature. As T ↑, so does average KE (i.e. speed) of gas particles. Gases at same T have same KEaverage. Maxwell-Boltzmann Distribution of Molecular Speeds: KE = (½)mv2 At the same temperature, which of the following gases molecules will have the greatest average speed? CO2, N2, H2, O2. H2 — why? It is the lightest molecule. Pressure P = Force/Area . . . units are N·m-2 or Pascal (Pa) The pressure of a gas arises from collisions of gas molecules with the sides of the container. As T ↑, pressure of gas also increases. Measuring Atmospheric Pressure Evangelista Torricelli built the first barometer ca. 1644 He inverted a long tube of Hg in a bowl of Hg. Column of Hg supported by Patm. Variations in column height due to variations in air pressure. Can make a Torricelli Barometer Using Water Instead of Hg. density of Hg(l) = 13.5 g/mL density of H2O(l) = 1.00 g/mL How much taller would an equivalent column of water have to be in order to be used as a Torricelli barometer? 13.5 X taller. Crescent’s Water Barometer Built in atrium of Science Wing using plastic tube, garbage can full of water, measuring tape. height of water column = 10.16 m equivalent ht of column of Hg = 10.16 m/13.5 = 0.753 m = 753 mmHg Classroom barometer read 754 mmHg. Different Pressure Units Standard atmospheric pressure (sea level) can be expressed as: 1.00 atm 101.3 kPa (Pa = N·m-2) 760 mmHg (or torr) 14.7 psi (pounds per square inch) Converting Pressure Units 1. Convert 102.3 kPa to mmHg. Ans: 768 mmHg 2. Convert 0.978 atm to kPa. Ans: 99.1 kPa 3. Convert 50 kPa to psi. No calculator Ans: 100 kPa ≈ 15 psi and so 50 kPa ≈ 7 psi How does the Patm at the top of Mount Kilimanjaro compare to Patm at sea level? Much lower—ca. 350 mmHg atop Kilimanjaro. HW Text sections 11.2, 11.2 to end p 509 P 506 RQ # 1 – 15. Read these questions before you read the chapter. Then answer the questions. P 510 LC # 7 - 12