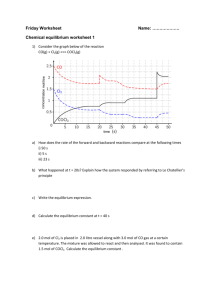
Name Chemistry: Kinetics & Equilibrium Review Write the
advertisement

Name Chemistry: Kinetics & Equilibrium Review Write the equilibrium expressions (for K) for the following reactions. 1. CO (g) + 2H2 (g) CH3OH (g) 2. H2 (g) + I2 (g) 2HI (g) 3. CaSO4 (s) Ca+2(aq) + SO4 -2 (aq) 4. 2HBr (g) H2 (g) + Br2 (l) 5. CO (g) + 2H2 (g) CH3OH (g) For each of the following, write the equilibrium expression and solve for K. Show all work!! 6. PCl5(g) → PCl3(g) + Cl2(g). What is the equilibrium constant if the equilibrium concentrations are as follows: PCl5 is 0.0096 mol/L, PCl3 is 0.0247 mol/L and Cl2 is 0.0247 mol/L? 7. At a given temperature, the Keq for the reaction 2HI(g) → H2(g) + I2(g) is 1.40 x 10-2. If the concentration of both H2 and I2 at equilibrium are 2.00 x 10-4M, find the concentration of HI. In each problem, calculate the missing concentration or constant at equilibrium. 8. 9. 10. [HI] 1.78 X 0.78 [H2] 0.172 0.242 0.112 [I2] 0.172 0.242 X Keq X 0.217 2.06 x 10_2 Be sure to review what influences he rate of a reaction and Le Chatelier’s Principle!!!