presentation_of_graduation_project - An
advertisement

An-Najah National University
Industrial Engineering Department
Graduation project (SIX SIGMA)
Prepared by:
Alaa Alsadi
Ameera Dawoud
Mays Hijjawi
Ons Shawahni
Supervisor:
Dr.Husam Arman
Birziet Pharmaceutical company
BPC was established in 1974 in Birzeit village as a private
shareholding company with a total capital Investment of USD
150,000.
BPC combines many factors in order to maintain its success:
Obtaining the latest quality standards certificates such as GMP
(Current Good Manufacturing Practices) and ISO quality systems.
Highly educated and well trained staff members distributed among
the different departments.
BPC Mission
— BPC realizes that the significance of the Palestinian
Pharmaceutical Industry extends far beyond the
size of the revenues.
BPC Vision
— Its vision is to be the backbone of the health
care security system in Palestine and the
region with superior quality product
Motivation
Pharmaceutical industry is a very sensitive and dangerous
industry because it deals with people's life . so we choose
to apply six sigma as a disciplined, data driven problem
solving approach supported by powerful statistical order
to reduce variation and improve quality of products and
tools in business processes in the way to achieve
perfection.
Research objectives
1. Evaluate the current practices in the
pharmaceutical companies.
2. Test and apply the Six Sigma concept in a
pharmaceutical company.
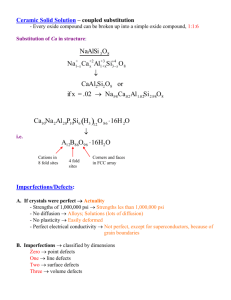
Definition
Six Sigma (in statistics) : the definition of outcomes as close
as possible to perfection. With six standard deviations, we
arrive at 3.4 defects per million opportunities, or 99.9997
percent, so “reaching Six Sigma” means that your process or
product will perform with almost no defects.
Six Sigma (as a philosophy) : is a total management
commitment and philosophy of excellence, customer focus,
process improvement, and the rule of measurement.
Levels of sigma performance per million
opportunities
Defect Per Million Opportunities
800000
690,000
700000
600000
500000
400000
DPMO
308,537
300000
200000
100000
0
3.4
233
6,210
6
5
4
66,807
3
2
1
Methodology
Structured
Interviews
With
Companies.
Evaluation of
Current
Situation
Results
Analysis of
Results.
Methodology
DMAIC
Methodology
Case Study.
Data
Collection
Plan
Solve The
Case Study
Using DMAIC
Methodology
1. Evaluation of current situation in pharmaceutical
companies.
Pharmaceutical companies were evaluated by a
questionnaire which contains many sections:
Introduction
Management style
Customer satisfaction
Defects
Education of employers
Muda(wastes)
Quality
2. Structured interviews with pharmaceutical companies.
Then structured interviews were held with the
pharmaceutical companies
quality managers to answer the questionnaire questions
( companies were given symbols from A to D for
confidentially purposes .
3. Analysis
Analysis Criteria was to give questions rates from four
to one , four for best answer and one for worst answer .
Finally rates of every company were summed so that
the company with highest rate is the best company.
Company
Rate
A
107
B
96
C
83
D
67
From Rating it's obvious that company A is the best
according to our evaluation so we choose company A
to implement our project which is Berzeit
Pharmaceutical company.
Evaluation of blistering
process in Birzeit
Pharmaceutical Company
DEFINE
This phase includes several steps:
1. Determine project charter.
Item
Business case
Description
Our project based on evaluation of blistering Process in Birzeit pharmaceutical company, we
chose this topic because blistering Process is the most critical Process in the manufacturing
process in the company.
Goal statements
Scope
We expect to achieve many things such as:
•
Reducing defects in blistering from 4% to3%.
•
Reducing customer complaints from 85.7% to 20%.
The scope of the project is the reducing customer complaints and defects in the blistering
Process.
Players
Preliminary plan
Mays Hijjawi .
Alaa Al-sa'adi.
Ameera Dawood.
Ons Shawahneh.
Define Phase: (7-11)/3
Measure phase: (21/2)-(20/3)
Analyze phase: (21-25)/3
Improve phase: (26-28)/3
Control phase: (29-30)/3
2. Validate
problem and goal statements.
Problem Statement
In the last year we found that 85.7% of customer complaints
were because of final packaging (Blistering ) , this is due to:
defects in this Process( about 4%) which leading to losing
519,832,891 NIS in the last year(Losses From Time and
defects) , and because of blistering is the bottleneck of the
processes in the company . so , we need to solve this
problem using DMAIC methodology.
Objective Statement
Our Objectives are: first "reduce the defects in blistering
Process from 4% to 3% during 3 months (30/4/2011),
"second reduce customer complaints to 20%". In order to
restore the company image and to save 519,832,891 NIS
in a year.
3. Create process map and scope.
UHLMANN UPS 1680
MEASURE
Monitoring and measuring the performance
indicators using data collection plan.
Three blistering machines were monitored:
1. UHLMANN UPS 1680
2. UHLMANN UPS 300
3. UHLMANN B 1240
But, here we will talk about the first machine
UHLMANN UPS 1680.
Machine breakdowns
Machine Breakdowns.
Total time of machine breakdowns.
(min)
Squashing
150
Sealing
55
Sealing retest
75
Unclear number
60
AL shiftiness
115
Specifications of process conditions
Batch #
Product
Type of
Mold size
Tablet type
blistering
Calculated
Tab. in
velocity
sachet
110007
NAPREX 500 MG
PVC-Al
Round
coated
20
10
100155
ERYTHROTAB
PVC-Al
oblong
coated
20
12
110034
ORACAL CHEWABLE
PVC-Al
Round
uncoated
20
10
100682
MOBICOL 200 MG
PVC-Al
Round
coated
20
10
110059
SEREPAM 5 MG
PVC-Al
Round
uncoated
20
20
100853
LARICID 500 MG
PVC-Al
oblong
coated
20
7
100721
ORALUTE
PVC-Al
Round
uncoated
20
20
100517
SPIRONE
PVC-Al
Round
coated
20
10
100851
PHENOTAB 15 MG
PVC-Al
Round
uncoated
20
20
Quantities (in, out, Defects) of process inputs
Batch #
Tablet In
Tablet
Defects of
PVC
PV C
Defects of
Al in
Al
Defects of
Out
Product
in
out
PVC(Kg)
(Kg)
out
Al (Kg)
(Kg)
(Kg)
(Kg)
110007
91988
91840
349
49.7
16.5
14.10
9.2
3.4
2.06
100155
142960
139836
1000
89.5
20.9
26.6
30.9
4.7
12.4
110034
155214
154360
520
105.2
27.8
8.9
24.6
5.8
1.08
100682
86041
83230
2100
51.5
21.5
13.7
9
3.7
2.25
110059
294424
290380
3060
51.4
29.1
21.9
12.8
5.5
3.7
100853
37230
36001
400
27.8
11.8
11
8.6
2
1.67
100721
190240
186260
3756
52.5
16.8
20.5
8.1
3.7
2.38
100517
86692
85050
500
26.4
15.3
8.7
9.2
3.2
.95
100873
7851
7469
100
26.7
1.9
11.7
4.2
.4
1.85
100851
388022
383560
5352
80.1
34.5
20
13
6.9
3.05
Operations time study
Batch #
Machine Cleaning
Machine Preparation Machine decomposing
Machine production
time (min).
time (min).
time (min).
time (min).
110007
15
240
15
330
100155
30
60
15
250
110034
30
45
15
390
100682
45
120
15
780
110059
15
80
15
790
100853
15
180
15
435
100721
15
40
15
755
100517
30
100
15
280
100873
15
50
15
105
100851
25
120
15
745
Analyze
1. Cause and effect diagram.
People
Materials
Methods
Experience
Motivation
Concentration
Reproducibility
level
Training
Mechanical failures
Responsibility
Out of
specification
Defects from
supplier
Incorrect
procedure
Planning
Damaged from
material handling
Worn tool
Inappropriate
Devices
Preventive
Maintenance
Lack of
spare parts
Machinery
Wrong Work
sequence
Bad Lighting
System/
Excessive noise
Excessive
temperature
Measurement
Environment
Defected
Sachets
Poor
Humidity
Not clean
2. Pareto charts
MACHINE BREKDOWNS
Total time
400
300
100
80
60
200
40
100
20
0
r
g
g
st
ss
ed
e
in
e
be
h
t
l in
n
s
e
a
m
t
m
r
r
if
se
ua
g
nu
Fo
sh
n
r
i
Sq
a
al
AL
c le
Se
n
u
Total time
150
115
75
60
55
15
Percent
31.9
24.5
16.0
12.8
11.7
3.2
Cum %
31.9
56.4
72.3
85.1
96.8
100.0
MACHINE BREAKDOWN
0
Percent
500
3. Control charts
Why We Choose I-MR?
Control charts were made for these processes:
Tablets Production.
Al .
PVC.
Preparation of Machine.
Cleaning of Machine.
Decomposing of Machine.
I-MR Chart of yield%(Tablets)
I-MR Chart of Yield% (PVC)
U C L=103.33
100
_
X=97.92
96
75
110007
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
25
0
100851
LC L=-7.6
110007
U C L=6.654
100155
110034
110059
100853
Batch number
100721
100517
100873
100851
3.0
__
M R=2.037
1.5
M oving Range
U C L=72.17
4.5
0.0
60
40
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
__
M R=22.09
20
LC L=0
110007
0
100851
LC L=0
110007
100155
110034
I-MR Chart of Yield% (AL)
100682
110059
100853
Batch number
100721
100517
100873
100851
Machine cleaning time
120
60
U C L=119.6
90
_
X=53.4
60
30
0
U C L=53.05
Individual Value
Individual Value
100682
80
6.0
M oving Range
_
X=51.1
50
LC L=92.50
92
U C L=109.9
100
Individual V alue
Individual V alue
104
45
30
_
X=23.5
15
0
LC L=-12.9
110007
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
110007
40
__
M R=24.91
20
0
LC L=0
110007
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
100851
110034
100682
110059
100853
Batch number
100721
100517
100873
100851
U C L=36.30
Moving Range
60
100155
40
U C L=81.40
80
M oving Range
LC L=-6.05
100851
30
20
__
M R=11.11
10
0
LC L=0
110007
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
100851
Machine Decomposing time
Machine preparing time
200
_
X=103.5
100
0
-100
Individual V alue
Individual V alue
20.0
U C L=319.2
300
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
12.5
110007
100851
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
100851
0.50
U C L=265.0
200
__
M R=81.1
100
0
LC L=0
110007
100155
110034
100682
110059
100853
Batch number
100721
100517
100873
100851
M oving Range
300
M oving Range
_
U CL=15
X=15
LC
L=15
15.0
10.0
LC L=-112.2
110007
17.5
0.25
__
MCR=0
U
LC
L=0
L=0
0.00
-0.25
-0.50
110007
100155
110034
100682
Note: All Processes Are in Control.
110059
100853
Batch number
100721
100517
100873
100851
4. Capability analysis:
Capability Analysis of yield% (tablets)
LSL
USL
P rocess Data
LS L
95
Target
*
USL
100
Sample M ean
97.916
Sample N
10
StDev (Within) 1.80556
StDev (O v erall) 1.41943
Within
Overall
P otential (Within) C apability
Cp
0.46
C P L 0.54
C P U 0.38
C pk 0.38
O v erall C apability
Pp
PPL
PPU
P pk
C pm
94
O bserv ed P erformance
P P M < LS L 0.00
P P M > U SL 0.00
P P M Total
0.00
Exp.
PPM
PPM
PPM
96
Within P erformance
< LSL
53153.68
> U SL 124205.94
Total
177359.62
98
100
0.59
0.68
0.49
0.49
*
102
Exp. O v erall P erformance
P P M < LSL 19971.14
P P M > U SL 71025.67
P P M Total
90996.80
We note from figure that capability of the process=0.38<1,
so the process is incapable.
Capability Analysis of Yield% (PVC)
LSLUSL
P rocess D ata
LS L
95
Target
*
USL
100
S ample M ean
51.117
S ample N
10
S tD ev (Within)
19.5833
S tD ev (O v erall) 17.142
W ithin
Ov erall
P otential (Within) C apability
Cp
0.04
C PL
-0.75
C PU
0.83
C pk
-0.75
O v erall C apability
Pp
PPL
PPU
P pk
C pm
20
O bserv ed P erformance
P P M < LS L
1000000.00
PPM > USL
0.00
P P M Total
1000000.00
E xp.
PPM
PPM
PPM
40
Within P erformance
< LS L
987481.59
> USL
6277.42
Total
993759.00
60
80
0.05
-0.85
0.95
-0.85
*
100
E xp. O v erall P erformance
P P M < LS L 994765.90
PPM > USL
2174.66
P P M Total
996940.56
We note from figure that capability of the process=0.75<1, so the process is incapable and all of data
points are out of specification limits.
Capability Analysis of Yield% of (AL)
LSLUSL
W ithin
Ov erall
P rocess D ata
LS L
95
Target
*
USL
100
S ample M ean
53.372
S ample N
10
S tD ev (Within)
22.0853
S tD ev (O v erall) 20.5056
P otential (Within) C apability
Cp
0.04
C PL
-0.63
C PU
0.70
C pk
-0.63
O v erall C apability
Pp
PPL
PPU
P pk
C pm
0
O bserv ed P erformance
P P M < LS L
1000000.00
PPM > USL
0.00
P P M Total
1000000.00
E xp.
PPM
PPM
PPM
20
40
Within P erformance
< LS L
970276.53
> USL
17374.63
Total
987651.16
60
80
0.04
-0.68
0.76
-0.68
*
100
E xp. O v erall P erformance
P P M < LS L 978825.54
PPM > USL
11485.69
P P M Total
990311.23
We note from figure that capability =-0.63 <1, so the
process is incapable and all of data points are out of
specification limits.
5. Study of losses
Losses from time.
Product
Tablet Out
Theoretical productivity in
one hour
Lost time
Lost tablet
#of tablet per
sachet
Lost sachet
Cost of one
sachet
Lost cost
91840
36000
2.9
106160
10
10616
7.2
76435.2
139836
43200
0.9
40308
12
3359
5.4
18138.6
154360
36000
2.2
79640
10
7964
1.8
14335.2
83230
36000
10.7
384770
10
38477
10.5
404009
290380
72000
9.1
657860
20
32893
3
98679
36001
25200
5.8
146699
7
20957
18
377226
186260
72000
10.0
719500
20
35975
12
431700
85050
36000
2.3
83070
10
8307
9
74763
7469
25200
1.5
36631
7
5233
18
94194
383560
72000
7.1
510680
20
25534
4.8
122563
NAPREX 500 MG
ERYTHROTAB
ORACAL CHEWABLE
MOBICOL 200 MG
SEREPAM 5 MG
LARICID 500 MG
ORALUTE
SPIRONE
LARICID 500 MG
PHENOTAB 15 MG
Total lost cost (NIS) =1,712,043
Losses from defects (tablets, PVC, AL)
Defects of
Product
(Tablets)
Product
#of tablet per
cost of one
sachet
defect sachet
sachet
lost cost
349
10
35
7.2
251.28
1000
12
83
5.4
450
520
10
52
1.8
93.6
2100
10
210
10.5
2205
3060
20
153
3
459
400
7
57
18
1028.571
3756
20
188
12
2253.6
500
10
50
9
450
100
7
14
18
257.1429
5352
20
268
4.8
1284.48
NAPREX 500 MG
ERYTHROTAB
ORACAL CHEWABLE
MOBICOL 200 MG
SEREPAM 5 MG
LARICID 500 MG
ORALUTE
SPIRONE
LARICID 500 MG
PHENOTAB 15 MG
Total lost cost (NIS) =8732.674
Batch #
Product
Defects of
Al (Kg)
Lost cost
cost of one
Defects of
PVC(Kg)
KG of AL
110007
NAPREX 500 MG
2.06
100155
ERYTHROTAB
12.4
110034
ORACAL CHEWABLE
1.08
100682
MOBICOL 200 MG
SEREPAM 5 MG
3.7
100853
LARICID 500 MG
1.67
100721
ORALUTE
2.38
100517
SPIRONE
0.95
100873
LARICID 500 MG
1.85
PHENOTAB 15 MG
82.77
40.18
498.23
40.18
43.39
40.18
90.41
40.18
148.67
40.18
67.10
14.1
26.6
Lost cost
11.9
167.79
11.9
316.54
11.9
105.91
11.9
163.03
11.9
260.61
11.9
130.9
11.9
243.95
11.9
103.53
11.9
139.23
11.9
238
13.7
21.9
11
40.18
95.63
40.18
38.17
40.18
74.33
40.18
122.55
20.5
8.7
11.7
3.05
20
Total cost
Total cost
of PVC
8.9
2.25
110059
100851
40.18
cost of one KG
1261.25
1869.5
Total cost of all
defects=1261.25+1869.5+8732.674=11863.424
Total losses from time and
defects=1,712,043+11863.424=1,723,907 NIS.
6. Sigma level calculations
We calculated defects per million and long term sigma
level that are followed in the blistering process for three
blistering machines
Defects per million=∑Defects/∑Output of
tablets*1,000,000
Long term sigma level=NORMSINV {1(∑Defects/∑Output of tablets)} + 1.5
Defects per million=11753.89
Long term sigma level=3.8
IMPROVE
Because of:
1. Limited time.
2. The company is satisfied with current quality level so, it didn't accept
applying solutions as design of experiments.
So, we suggest some solutions to improve the process that show in the
following table:
Problem
Cause
Suggested Solution
PVC and Al shiftiness
Defective from supplier
Never use defective raw materials
Camera Problems (extra tablets, breaking tablets).
Labor error, excess temperature, hardness of
Retraining blistering and maintenance employees.
tablet.
Control previous Processs (Mixing and compression).
Unclear batch number.
Labor error.
Retraining blistering employees.
Calibrating Machine problems.
Labor error.
Retraining maintenance employees.
Empty sachets.
Labor error.
Retraining blistering employees,
Sealing Problems.
Mold problems, Excess temperature, Labor
Retraining blistering and maintenance employees,
error, broken rings.
Replace Local molds with original ones, Replace
defective molds, replace rings.
Entering extra tablets under sealing unit.
Labor error.
Retraining blistering employees, put the barrier part to
prevent tablet entering.
Forming
Unsuitable temperature with PVC type, worn
New calibration, replace rings.
rings.
Squashed( Burning)
Inaccurate calibration, unsuitable temperature, Retraining blistering and maintenance employees,
PVC type, labor error.
new calibration.
CONTROL
Specific Control tasks that DMAIC team much
complete include:
Implement three levels of retaining (low, medium, high)
on three employees with same level of productivity and
measure the effect on their performance.
Perform Reliability analysis on all machines in the
company.
Perform Design Of Experiments on critical parameters
of any process in the company.
Put a good criteria for preventive maintenance for all machines in the
company.
Developing a monitoring process to keep track of the changes they have set
out.
Creating a response plan for dealing with problems that may arise.
Helping focus management’s attention on a few critical measures that give
them current information on the outcomes of the project (the Y) and key
process measures, too (the Xs.)
We recommended implementing major concept in
control world; this is (Poka-Yoke).
Poka-Yoke is the transliteration of a Japanese phrase
meaning “to make mistakes impossible"
Common Poka-Yoke implementations include:
Physical features or geometry.
Automated processing, inspection systems.
Limiting controls that don’t allow the process to be
operated at unacceptable levels.
We recommend using simple devices and procedures
that prevent mistakes:
Photo sensors in order to skip the bad sachets.
Checklists to ensure that everything in good status.
Trip switches to stop the machine when the workers
work on fault setting especially in temperature.
Control panel in order to set the process easily.
Fixtures to orient parts.
Conclusion
We can conclude from this project that:
1. Birzeit Pharmaceutical company took the best rate
according to our evaluation so, it was choose for the
project.
2.
long term sigma level for the machines:
UHLMANN UPS 1680=3.8
UHLMANN B 1240=4.44
UHLMANN UPS 300=3.6
Then, UHLMANN B 1240 has the higher sigma level, so
this machine has good quality.
3. The most of lost cost comes from waste time not come
from bad quality, in the contract, the quality of process
and products were good and not costly as waste time so
the company should work on lean manufacturing
system.
4. The results from comparison between three machines
that the performance of UHLMANN B 1240 machine better
than UHLMANN UPS 1680 machine performance which
better than UHLMANN UPS 300 machine performance.
5. The total lost cost resulted from all machines production
process for tablets during 3 months =58,281,053(NIS)
Alaa Alsadi
Ameera Dawoud
Mays Hijjawi
Ons Shawahni